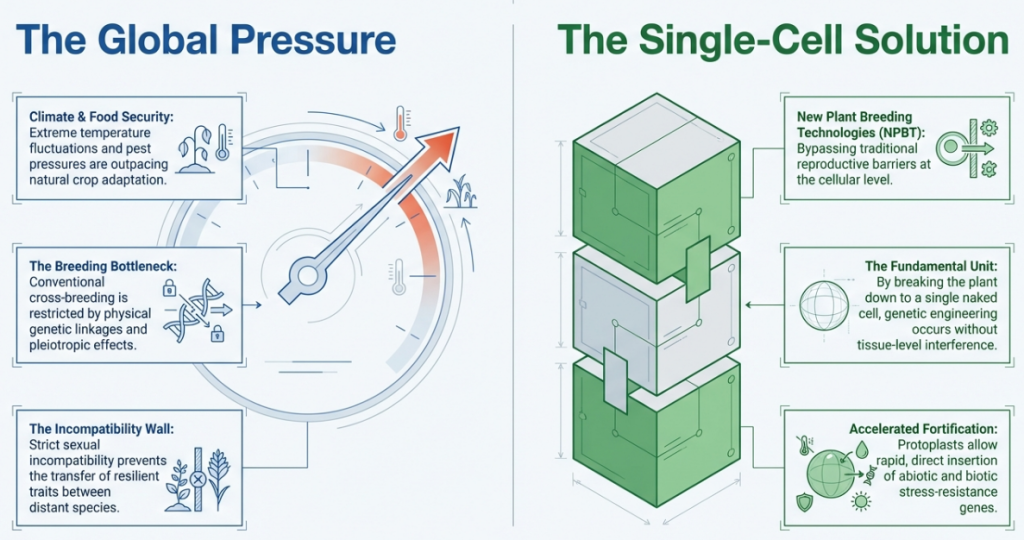
1. Introduction to Protoplast Technology in Modern Agriculture
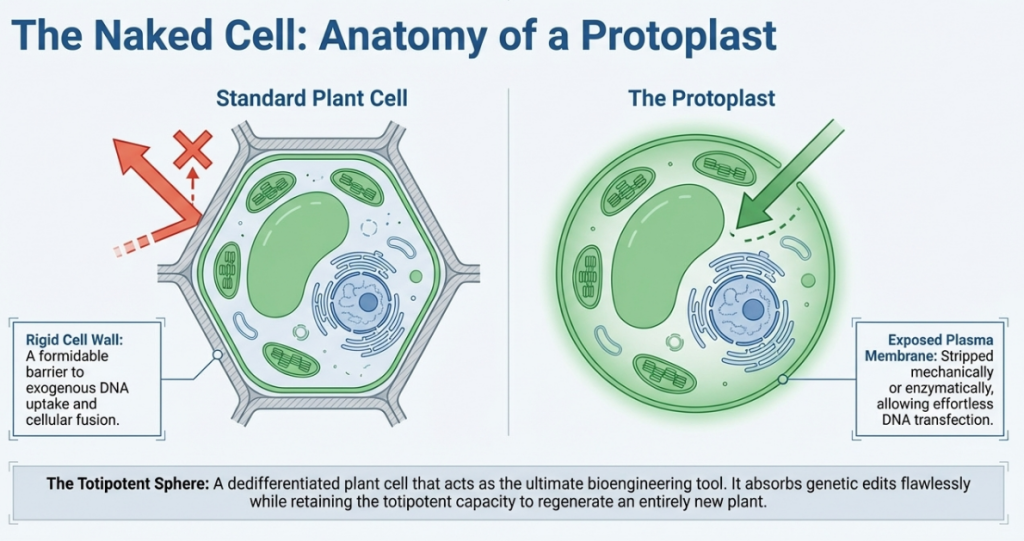
Protoplasts represent the “living matter” of the plant, consisting of individual cells stripped of their rigid cell walls to expose the underlying plasma membrane. In the current era of crop improvement, these individualized cells serve as a foundational platform for accelerating the “design-build-test-learn” cycle, allowing for rapid iterations in genetic research that bypass the multi-year constraints of traditional breeding. By providing direct cellular access for genetic transformation and the precise study of plant physiology, protoplast technology bridges the gap between theoretical molecular biology and strategic agricultural advancement. Mastering the transition from an isolated cell back to a fully functional, regenerated plant is the essential prerequisite for leveraging these systems in advanced breeding programs.

Historically, protoplast technology has evolved from the 19th-century microsurgery of plasmolyzed cells to the 1960 breakthrough of enzymatic digestion. Today, these systems offer distinct advantages over agroinfiltration and stable transformation:
- Unmatched Speed: Phenotypic observation and gene expression data are available within hours, compared to the months required for stable integration.
- High-Throughput Potential: Parallel characterization of thousands of genetic parts (e.g., promoters, UTRs) and CRISPR constructs.
- Transient Expression Flexibility: Rapid screening of genetic circuits and DNA-free editing without the complications of insertion-site variability.
- Broad Species Applicability: A versatile chassis for species recalcitrant to standard Agrobacterium-mediated transformation.
Establishing this platform requires a sophisticated pipeline, beginning with the precision optimization of isolation and purification.
Key Milestones in Protoplast History
- 1880: Hanstein introduces the term “protoplast” to describe the living matter within the cell walls.
- 1892: Klercker achieves the first isolation using microsurgery on plasmolyzed cells.
- 1960: Cocking develops the gold-standard enzymatic method for cell wall digestion, using enzymes like cellulase and macerozyme.
Why Researchers Use Protoplasts
- Genetic Transformation: Protoplasts can easily take up foreign DNA or CRISPR-Cas ribonucleoprotein (RNP) complexes via PEG-mediated transfection or electroporation, enabling transgene-free genome editing.
- Somatic Hybridization: Scientists can fuse protoplasts from different varieties or species to create “somatic hybrids,” bypassing sexual compatibility barriers to transfer traits like disease resistance or cytoplasmic male sterility.
- Rapid Gene Testing: Protoplasts allow for high-throughput characterization of genetic parts (promoters, UTRs) within hours, providing a much faster alternative to the months required for stable plant transformation.
2. Principles of Protoplast Isolation: Maximizing Yield and Viability
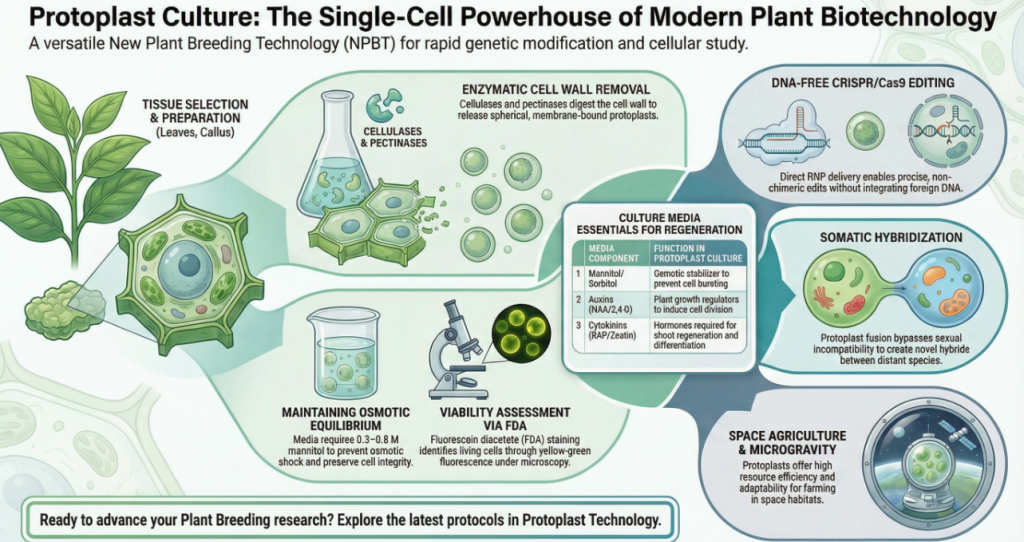
Isolation efficiency is the foundational bottleneck for all downstream protoplast applications. Because processes such as CRISPR/Cas-mediated genome editing and somatic hybridization require high-quality cellular starting material, the optimization of enzymatic digestion is non-negotiable. Isolation success is governed by a precise synergy of enzyme concentrations, osmotic stabilization, and tissue age.
| Species | Tissue Source | Enzyme Composition | Optimal Incubation Conditions |
|---|---|---|---|
| Brassica oleracea | Young leaf blades | 0.5% Cellulase, 0.1% Pectolyase | 16h @ 26°C (30 rpm shaker) |
| Arabidopsis (PSB-D) | Suspension culture | 1% Cellulase RS, 0.05% Pectinase, 0.2% Driselase | 1.5–2h @ 27°C (130 rpm shaker) |
| Tobacco (BY-2) | Suspension culture | 1% Cellulase RS, 0.05% Pectinase, 0.2% Driselase | 2.5–3h @ 25°C (130 rpm shaker) |
| Rice | Suspension culture | 4% Cellulase RS, 0.2% Pectinase, 0.8% Driselase | 3–5h @ 30°C (Dark, no shaking) |
Isolation Yield and Viability For B. oleracea, optimized digestion protocols yield an average of 2.5 × 10⁶ cells per gram of fresh weight (FW), with a mean viability of 88.2% as confirmed by fluorescein diacetate (FDA) staining.
The Strategic Necessity of High Density Maintaining a working density of 2–5 × 10⁵ cells/ml is critical for success. This high density is essential because:
- Paracrine Signaling: It ensures the balanced release of endogenous growth factors and phytosulfokines necessary for cellular communication.
- Mitotic Re-entry: It provides the physiological environment required for cells to overcome the arrest associated with wall removal and re-enter the mitotic cycle.
- Metabolic Stability: Densities outside this range typically result in developmental inhibition due to the localized overproduction of inhibitory metabolites.
The Protoplast Reagent Toolkit
| Reagent Name | Biological Target/Role | The “Learner’s Insight” (Primary Benefit) |
|---|---|---|
| Cellulase (0.5% Onozuka R-10) | Cellulose fibers in the cell wall. | Systematically dissolves the rigid scaffolding that gives the cell its fixed shape. |
| Pectolyase (0.1% Y-23) | Pectin in the middle lamella. | Cleaves the biological “glue” holding cells together, ensuring tissue dissociation. |
| Mannitol (0.4 M – 0.5 M) | Osmotic stabilizer. | Creates an isotonic environment to prevent immediate cell lysis once the wall is removed. |
| MES (5 mM) | pH Stabilization. | Maintains a stable pH (5.7–5.8) during the 16-hour digestion, preventing metabolic shock. |
The “Delicate Balance” of Mannitol The concentration of mannitol is your most critical variable. Within a walled cell, turgor pressure is maintained against the cellulose structure. Once that wall is removed, the cell is highly susceptible to osmotic shock. Mannitol acts as a buffer; if the concentration is too low, the internal pressure causes the cell to burst (lysis). Conversely, if it is too high, the cell shrivels (plasmolysis). Precise tuning keeps the protoplast viable and round, which is a prerequisite for mitotic re-entry.

2.1 Strategic Selection: Source Materials for Transformation
Selecting your source material is a strategic decision that balances growth speed against imaging requirements. As a researcher, you must choose between “unlimited” lab cultures and greenhouse-grown tissue.
| Criteria | Suspension Cultures | Leaf Sheaths / Mesophyll |
|---|---|---|
| Growth Speed | Fast-growing; provides a constant, year-round supply of cells. | Dependent on seedling growth (e.g., 4 weeks for Brassica). |
| Imaging Compatibility | Excellent. Low autofluorescence allows for clear monitoring of GFP/fluorescent tags. | Challenging. Red chloroplast fluorescence in mesophyll often interferes with signals. |
| Availability | Requires maintenance of stable lab lines like Tobacco BY-2 or Arabidopsis PSB-D. | Requires growth chamber space; material is often “fresher” but less uniform. |
| Specific Species Example | Used for rapid testing of genetic parts (cis-regulatory elements). | Oryza sativa (Rice) leaf sheaths are used because rice leaves have prohibitive wax and silica layers. |
2.2 From Single Cell to Whole Plant: The Regeneration Bottleneck
Getting DNA into a cell is only half the battle. To create an edited crop, you must “rebuild” the plant from a single cell. This is the regeneration bottleneck, and it is notoriously genotype-dependent.

Optimized Digestion and Purification Sequences
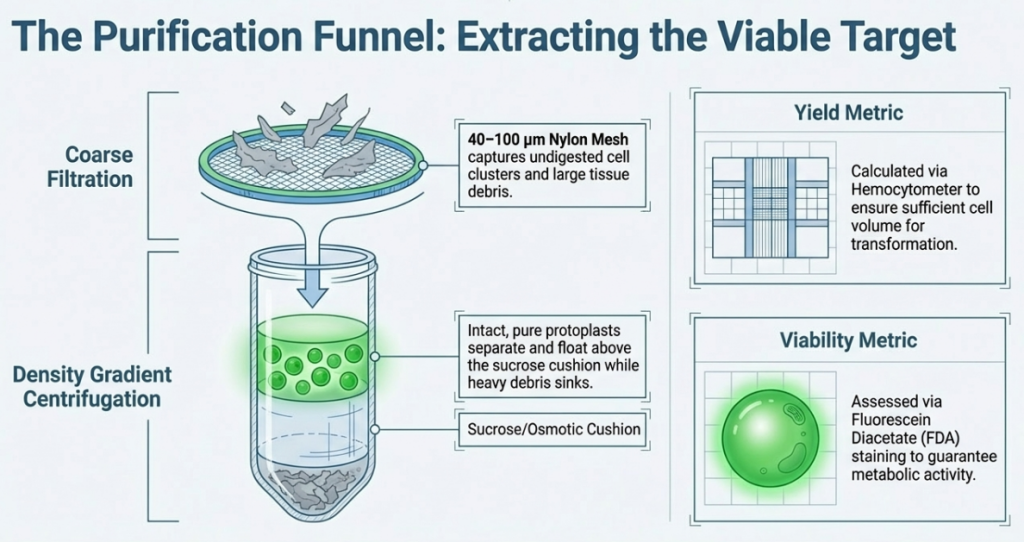
Recovering undamaged protoplasts requires minimizing mechanical shear forces and ensuring high-purity separation from vascular debris.
Digestion and Purification Workflow
- Incubation: Digestion occurs in the dark. Suspension cells are shaken at 130 rpm (1.5–3 h), while leaf tissues are maintained at 30–50 rpm (4–16 h).
- Filtration: The mixture is passed through a 50 µm or 100 µm nylon mesh to remove undigested tissue.
- Density Gradient Centrifugation:
- Floating Taxa (Arabidopsis/Tobacco): After initial pelleting, cells are resuspended in Sucrose/MES and overlaid with W5. During centrifugation (1200 rpm, 10 min), viable protoplasts float to the interphase.
- Pelleting Taxa (Rice): Protoplasts are typically recovered as a pellet in W5 or R2S medium.
- Washing Cycles: Traces of enzymes are removed via centrifugation at 80 x g to 200 x g.
- CRITICAL PARAMETER: Always disable the centrifuge brake. Rotor braking causes a vortex that disrupts the delicate cell pellet or interphase layer.
Handling and Aspiration Nuance
To maintain maximal viability, use only 1-ml top-cut pipet tips for all cell transfers to prevent shearing. For floating layers, use a peristaltic pump with a sharpened glass needle. Insert the needle through the protoplast layer to the bottom of the tube to aspirate the underlying solution, rather than attempting to pipette the cells from the top.
The Four Stages of Regeneration
Acclimatization: Moving plantlets from the pampered in vitro environment to ex vitro soil.
Cell Wall Resynthesis: The cell must first rebuild its “cage.” We confirm the completion of this rebuilding phase using Calcofluor White staining; a blue fluorescent glow under UV light indicates that cellulose is once again protecting the cell.
Mitotic Division: The single cell re-enters the cell cycle to form a multicellular microcallus.
Organogenesis: We manipulate the Cytokinin/Auxin ratio to induce shoots. Pro-Tip: My lab has found that for Brassica, a medium with 1% sucrose + 2% mannitol stimulates more effective shoot regeneration than the standard 3% sucrose.
3. Advanced Protoplast Culture: Immobilization and Microcallus Formation
The strategic transition from isolated cells to dividing colonies requires a stable physical environment that prevents cell aggregation. Aggregation in traditional liquid culture often leads to the localized overproduction of toxic metabolites, which induces premature cell death.
Advanced Immobilization: The Alginate-Layer Technique
Physical separation in a semi-solid matrix is critical to prevent the cell aggregation that leads to toxic metabolite overproduction and inhibited development.
Thin Alginate Layer (TAL) Protocol
- Mixing: Purified protoplasts are adjusted to 8 x 10⁵ cells/ml and mixed 1:1 with a 2.8% sodium alginate solution.
- Gelation: 400 µl of the mixture is spread onto calcium-agar medium. Allow one hour for gelation at room temperature.
- Cultivation: Layers are submerged in 4 ml of liquid media (CPPO1, Bras4, or Bras5) supplemented with 200 mg/l Cefotaxime. Media must be renewed every 10 days.
- Release: For downstream organogenesis, microcalli are released from the matrix using a 20 mM sodium citrate solution for one hour, followed by a wash in CPPD2 medium.
Osmotic Stabilization: While glucose is a primary energy source, the addition of 2% mannitol to the regeneration medium (Medium P) has been shown to stimulate shoot formation in Brassica more effectively than sucrose alone.
Developmental Timeline (B. oleracea Protocol)
- Day 1: Initial viability assessment via FDA staining.
- Day 2–3: Commencement of cell wall resynthesis; cellulose deposition is monitored using Calcofluor White M2R.
- Day 5: Mitotic re-entry and the first observed cytokinesis (plating efficiency).
- Day 30: Formation of visible microcalli (≥ 0.5 mm) ready for release from the alginate matrix via sodium citrate treatment.
Despite these optimized conditions, the “dominant role of genotype” remains the primary determinant of plating efficiency, necessitating cultivar-specific refinements for recalcitrant germplasm.
4. The Regeneration Frontier: Overcoming Recalcitrance
The “protoplast-to-plant” regeneration pathway is the ultimate frontier for producing stable transformants and somatic hybrids. However, many genotypes exhibit extreme recalcitrance, failing to develop beyond the callus stage regardless of morphological appearance.
Media Composition and PGR Dynamics
The transition to whole plants is governed by the Cytokinin/Auxin (C/A) ratio. Data indicates that a balanced 1:1 ratio is significantly more effective for Brassica than a high 5:1 ratio. The success of “Medium P” is attributed to its specific hormonal profile:
- Hormonal Selection: Medium P utilizes 2iP (Cytokinin) and NAA (Auxin), which has proven far superior to failed formulations using Zeatin and IAA.
- Growth Regulators: Low-concentration Gibberellic Acid (GA3 ≤ 0.02 mg l⁻¹) surprisingly promotes shoot formation.
- Osmotic Stabilization: Supplementation with 2% mannitol alongside 1% sucrose stimulates more effective shoot regeneration than sucrose-only media.
Genotypic Failures: The Case of ‘Haco’ Even when morphological indicators are positive, genotype remains the absolute arbiter of success. For example, the Red Cabbage cultivar ‘Haco’ successfully formed dense, green callus—a typical indicator of regenerative potential—yet failed to produce shoots on all tested media. This highlights the necessity of overcoming internal genetic barriers that persist even when primary culture requirements are met.
The Molecular Network Hierarchy
To overcome recalcitrance in elite commercial varieties, R&D programs must manipulate the hierarchical network of somatic embryogenesis master regulators:
1. WUS (WUSCHEL): The apex regulator required for activating the transcription factor network.
2. LEC1 & LEC2 (LEAFY COTYLEDON): Activated by WUS to drive embryonic development.
3. BBM (BABYBOOM) & AGL15: These factors interact with the LEC network to trigger endogenous auxin production, essentially looping the cell back into a regenerative state.
Case Study: Accelerating Improvement in Brassica oleracea and Collard Varieties
Brassica oleracea (cabbage, broccoli, kale) represents a high-value commercial segment, yet many varieties like collard (var. viridis) remain genetically untapped. These crops are currently threatened by multigenic challenges like fusarium yellows and black rot. Protoplast fusion and somatic hybridization offer a unique advantage here, enabling the transfer of complex traits—such as broad-spectrum resistance and cytoplasmic male sterility—that are difficult to achieve through traditional breeding.
Proof of Concept: Successful ‘Vates’ Collard Regeneration The first successful protoplast-to-plant regeneration of the ‘Vates’ collard cultivar has been achieved. Beyond basic breeding, this success establishes collard as a high-potential platform for molecular farming. The ability to regenerate ‘Vates’ allows it to serve as a source of recombinant antigens and pharmaceutical proteins, such as B5 vaccine candidates against smallpox. Flow cytometry of these regenerants confirmed a high rate of diploidy (79.5%), ensuring genetic stability and fertility for commercial production.
5. Strategic Applications: Synthetic Biology and Transient Transformation
Protoplasts serve as a high-throughput platform for rapid gene testing, providing data within hours—compared to the months required for stable transformation.

5.1 The Mechanism of Entry: Physical vs. Chemical Methods
To move DNA across the plasma membrane, we generally rely on either mechanical force or chemical facilitation.
Physical Method: Electroporation
Mechanism of Action: Protoplasts and DNA are placed in a conductive buffer and subjected to a high-voltage electrical pulse (typically 130V). This creates temporary, microscopic pores in the plasma membrane, allowing charged DNA to enter the cell.
- Primary Benefit: High efficiency and speed. It is a “clean” mechanical method that works well with Arabidopsis and Tobacco cells.
- Potential Drawback: High risk of cell damage.
- Pro-Tip: The electrical pulse must be precisely calibrated, usually between 25 to 50 msec. Crucially, never pulse the cells twice; doing so will result in irreversible membrane rupture and cell death.
Student Takeaway: Electroporation is the “power move.” It requires expensive equipment (like a Gene Pulser) but offers high efficiency for transient expression if your pulse parameters are perfect.
Chemical Method: PEG-Mediated Transformation
Mechanism of Action: This method utilizes Polyethylene Glycol (PEG) and high concentrations of calcium (calcium-induced uptake) to alter membrane properties and facilitate DNA entry.
- Primary Benefit: Extremely low cost and multi-sample handling. Unlike electroporation, which requires queuing at a machine, PEG reactions are tube-based, allowing you to process dozens of constructs simultaneously.
- Efficiency Benchmark: Under optimal conditions (1×10⁷ cells/ml and 10–40µg DNA), leaf sheath protoplast transformation can reach a staggering 90% efficiency.
- Potential Drawback: PEG is time-sensitive; prolonged incubation is toxic to the “naked” cells.
Student Takeaway: PEG is the “accessible” method. It is the preferred choice for most labs because it is inexpensive, handles high throughput, and is effective across a wide range of species, including rice and cabbage.

Transformation Methodology Comparison
| Feature | PEG-Mediated Transformation | Electroporation |
| Required Equipment | High-purity PEG 4000; Sucrose/MES stabilization buffers | Electroporation apparatus; 0.4-cm gap cuvettes |
| Suitability | Rice leaf sheaths, Arabidopsis, Tobacco | suspension-cultured Arabidopsis and Tobacco |
| Key Parameters | Centrifugation without deceleration/braking | Single pulse of 130V and 1000 mF (25-50 msec) |
| Key Advantages | Low cost; scalable for multiple simultaneous samples | High efficiency for suspension cultures |
Lab-on-a-Chip and Microfluidics The integration of “Lab-on-a-Chip” technology allows for the encapsulation of single protoplasts in microdroplets. This miniaturization facilitates high-throughput sorting and screening of CRISPR/Cas libraries. By creating closed, controlled environments, researchers can screen for specific metabolites or gene expression profiles before selecting individual high-performing cells for regeneration.
Applications of Transient Expression
- Subcellular Localization: Mapping protein trafficking using markers like aleurain-GFP (vacuolar) and mRFP-AtVSR2 (prevacuolar).
- STARR-seq: Characterizing thousands of native and synthetic cis-regulatory elements in parallel to identify the “regulatory logic” of the plant genome.
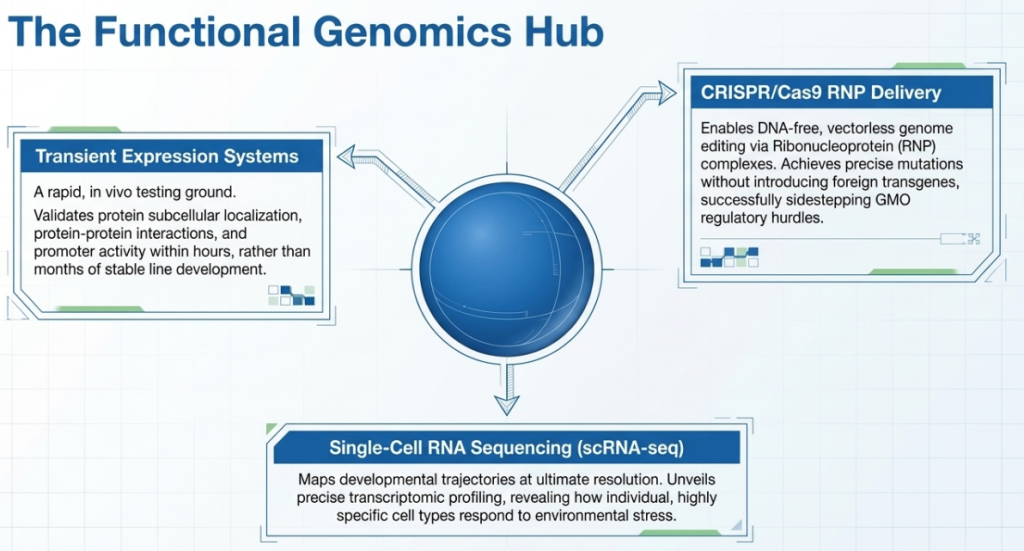
Table: Optimal Conditions for Cereal Protoplast Transformation
| Parameter | Recommended Specification |
|---|---|
| Protoplast Density | 1 × 10⁷ cells/ml |
| Transformation Efficiency | ~90% (Validated in Rice) |
| DNA Concentration | 10 – 40 µg per 500 µl aliquot |
| Centrifugation Note | Use swinging-bucket rotors without deceleration to preserve the fragile floating layer. |
DNA Quality and Ratios
- Ratios: Use 10–40 µg of plasmid DNA for 2–5 x 10⁶ cells. Exceeding this threshold increases cellular toxicity without improving efficiency.
- DNA Preparation: While commercial kits (e.g., Qiagen Maxi) are suitable for leaf-derived protoplasts, they paradoxically result in very low efficiency for suspension-cell transformations. For high-stakes suspension assays, CsCl-gradient centrifugation remains the gold standard for DNA purification.
Quality Assurance: Characterization and Performance Metrics
Standardized metrics ensure that experimental results are representative and reproducible.
Post-Isolation Assessment Checklist
- Yield Calculation: Use a Fuchs Rosenthal or hemocytometer; expect approx. 2.5 x 10⁶ cells/g FW for healthy Brassica leaf tissue.
- Viability Assays: Fluorescein Diacetate (FDA) staining should reveal >80% viability in high-quality preparations.
- Cell Wall Resynthesis: Tracked via Calcofluor White M2R. In Brassica, 80–90% of cells should show cellulose reconstruction by 72 hours.
- Plating Efficiency: Measured at Day 5 and Day 15 to validate mitotic vigor.
Ploidy Analysis and Genomic Stability
Flow cytometry is required to monitor the risk of tetraploidization (due to spontaneous fusion) and mixoploidy (due to mitotic instability in callus). In the context of the “design–build–test–learn” cycle, ploidy instability ruins the “test” and “learn” phases by introducing non-representative genomic data, ultimately undermining the reproducibility of synthetic biology circuits and stable regeneration efforts.
This unified workflow provides the scalability necessary for advanced applications, from the production of pharmaceutical antigens to high-efficiency genome editing across multiple plant taxa.
6. Conclusion: Actionable Directives for R&D Adoption
Protoplast platforms are no longer a niche tool; they are a prerequisite for competitive excellence in the age of synthetic biology.
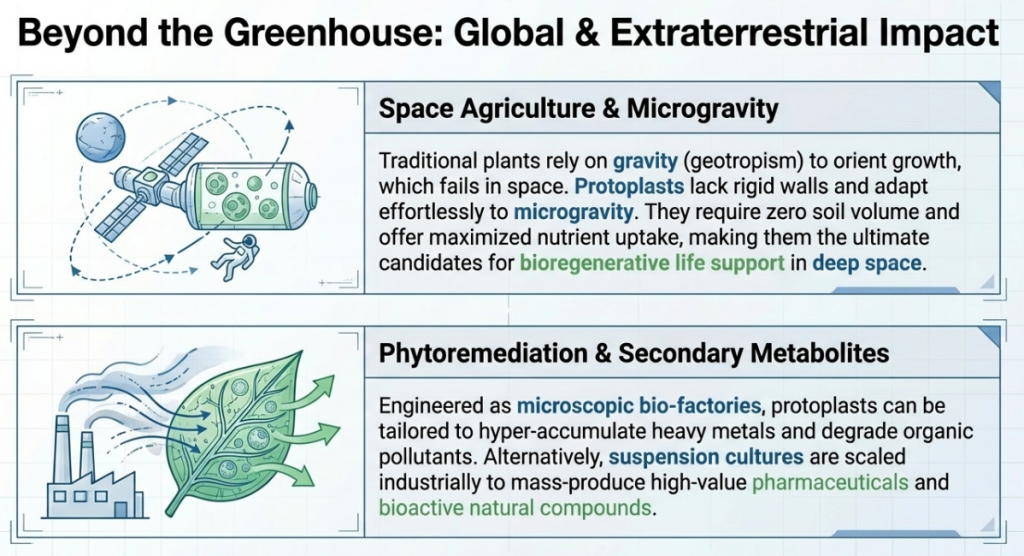
To secure commercial resilience and technological leadership, the R&D roadmap must mandate the following:
Utilization of Protoplast Fusion: Leverage somatic hybridization for the transfer of multigenic traits, such as disease resistance, that are currently unmanageable through traditional crossing.
Investment in Genotype-Independent Regeneration: Mandate the use of thin alginate-layer embedding and the manipulation of the WUS/BBM molecular hierarchy to ensure elite germplasm is not excluded due to recalcitrance.
Adoption of RNP-Based Editing: Utilize RNP-based activation of BBM/WUS to stimulate transgene-free regeneration, ensuring a clear path to market through favorable regulatory frameworks.

7. Technical Appendix: Media and Solutions Reference
| Solution Name | Primary Components | Strategic Application |
|---|---|---|
| W5 Solution | 154 mM NaCl, 125 mM CaCl₂, 5 mM KCl, 2 mM MES, 5 mM Glucose; pH 5.8 | Protoplast purification and washing; post-isolation stabilization |
| CPPO1 Medium | Kao & Michayluk salts, 0.4 M Glucose, Casein enzymatic hydrolysate, 2,4-D, NAA, BA | Protoplast culture and initial growth stage |
| MMg Solution | 4 mM MES, 0.5 M Mannitol, 15 mM MgCl₂; pH 5.7 | Preparation for PEG-mediated transformation |
| CPPD2/P Medium | MS salts, Mannitol, 1.0 mg/l NAA, 1.0 mg/l 2iP, 0.02 mg/l GA3, Casein enzymatic hydrolysate, Zeatin (add post-autoclave) | Callus maturation and induction of shoot organogenesis |
| ESC Enzyme | 0.5% Cellulase R-10, 0.1% Pectolyase Y-23, Mannitol, CaCl₂, 5 mM MES | Enzymatic cell wall digestion for Brassica mesophyll |
| PEG Solution | 40% (w/v) PEG 4000, 0.3 M Mannitol, 0.1 M CaCl₂ | Chemical induction of plasma membrane permeability for DNA uptake |
Image Summary


Questions/Answers
1. What is the protoplast-to-plant regeneration protocol for Brassica oleracea?
The protoplast-to-plant regeneration protocol for Brassica oleracea (which includes varieties such as cabbage, broccoli, kale, and collard) involves several critical stages, ranging from tissue selection to final plant acclimatization. While protocols vary slightly by cultivar, a recently optimized and robust method for several B. oleracea varieties is detailed below.
1. Source Tissue and Pre-treatment
- Optimal Source: The most effective source of protoplasts for B. oleracea is typically young leaf blades from 4-week-old plants grown in vitro. Alternatively, 1-week-old hypocotyls or cotyledons may be used, though leaf mesophyll often yields a higher number of cells.
- Preparation: Leaf material is cut into fine pieces and pre-treated in a plasmolysis solution for one hour. This step is crucial to shrink the cytoplasm away from the cell wall, protecting the plasma membrane from damage during enzymatic digestion.
2. Protoplast Isolation and Enzymolysis
- Enzymatic Digestion: The pre-treated tissue is incubated in an enzyme solution (such as ESC solution) for approximately 16 hours at 26°C in the dark. These solutions typically contain a mixture of cell-wall-degrading enzymes like cellulase and pectinase (e.g., Macerozyme or Pectolyase).
- Shaking: Incubation is performed on a gyratory shaker at a low speed (e.g., 30 rpm) to facilitate the release of the protoplasts.
3. Purification and Immobilization
- Purification: After digestion, the suspension is filtered through a nylon mesh (100 µm) and centrifuged to pellet the cells. Gradient centrifugation using a sucrose/MES solution overlaid with a W5 solution is employed to isolate undamaged, viable protoplasts from the interphase.
- Immobilization: High-quality protoplasts are embedded in thin calcium-alginate layers. Alginate embedding is preferred over liquid culture because it prevents cell aggregation, which can lead to the production of toxic metabolites and inhibited growth.
- Density: The working density is typically adjusted to approximately 8 × 10⁵ protoplasts per ml.
4. Protoplast Culture and Microcallus Formation
- Incubation: Embedded protoplasts are cultured in the dark at 24°C in specific media (such as CPPO1, Bras4, or Bras5) designed to support cell wall resynthesis and the first mitotic divisions.
- Microcallus Release: Protoplast-derived cells undergo continuous divisions, forming microcalli visible to the naked eye after about 30 days. These microcalli are then released from the alginate matrix using a sodium citrate solution.
5. Shoot and Root Regeneration
- Shoot Induction: The released microcalli are plated onto solid regeneration media. Research indicates that Medium P—supplemented with 1 mg l⁻¹ NAA, 1 mg l⁻¹ 2iP, and low concentrations of GA₃ (0.02 mg l⁻¹)—is highly effective for shoot formation.
- Carbon Source: Interestingly, while sucrose is standard, a combination of reduced sucrose (1%) and mannitol (2%) in the regeneration medium has been shown to stimulate more effective shoot regeneration in many B. oleracea cultivars.
- Rooting: Shoots typically produce roots as they develop on the regeneration media.
6. Acclimatization
- Environment: Rooted plantlets are transferred to a substrate (like coconut substrate) and placed in climatic chambers at approximately 19°C.
- Humidity Control: Initial air humidity is maintained at 90% and gradually reduced to 70% over a two-week period to allow the plants to harden for ex vitro conditions.
Key Influencing Factors
- Genotype Dependence: The efficiency of this protocol is highly dependent on the genetic makeup of the cultivar; for instance, varieties like ‘Kalibos’ red cabbage and ‘Vates’ collard show significantly higher regeneration rates than others.
- PSK Application: The addition of Phytosulfokine-α (PSK) to the culture medium has been found to stimulate cell divisions and enhance regeneration in several B. oleracea accessions.
2. How do electroporation and PEG methods compare for DNA transformation?
Both PEG (polyethylene glycol) and electroporation are widely used methods for DNA transformation in plant protoplasts, each presenting distinct mechanical mechanisms, costs, and levels of technical complexity. PEG-mediated transformation is the more commonly adopted technique due to its simplicity, low cost, and minimal requirement for specialized instrumentation.
Mechanical and Methodological Differences
- Mechanism of Action: PEG acts by physically binding to cell membranes, disturbing the phospholipid bilayer and destabilizing the cell surface potential to facilitate DNA entry. Conversely, electroporation utilizes brief, high-voltage electrical pulses to temporarily induce microscopic pores in the plasma membrane through which exogenous DNA can enter.
- Equipment and Accessibility: PEG methods use inexpensive supplies and are easily adapted to various plant species and tissue sources without needing specialized devices. Electroporation requires expensive, specialized equipment to generate and control the electrical pulses.
- Sample Throughput: The PEG method is favored for its ease in handling multiple samples simultaneously, whereas electroporation often involves individual cuvette processing.
Efficiency and Cell Viability
- Transformation Rates: Success rates for PEG typically range between 30% and 50%, though rates as high as 80% to 90% have been reported in wheat, rice, and petunia. For electroporation, demonstrated efficiencies for delivering components like CRISPR/Cas9 range from 20% to 30%. In specific studies on cabbage, electroporation was found to be nearly twice as efficient as PEG, though both maintained relatively low overall rates (3.4% vs 1.8%).
- Cellular Impact: PEG can be toxic to cells, and its concentration must be carefully balanced; as PEG concentration increases, cell viability often decreases. Electroporation avoids chemical toxicity but requires meticulous standardization of voltage, pulse length, and pulse number because sub-optimal conditions can cause severe cellular damage or death.
Comparative Advantages and Disadvantages
| Feature | PEG-Mediated Transformation | Electroporation |
|---|---|---|
| Primary Advantage | Simple, low-cost, and accessible. | Controlled environment and high repeatability. |
| Primary Disadvantage | Chemical toxicity and potential for reduced cell aggregation. | High equipment cost and potential for electrical damage. |
| Optimization | Focuses on PEG concentration and incubation time. | Focuses on pulse voltage, frequency, and duration. |
Ultimately, while PEG is the method of choice for many laboratories due to its convenience and speed, electroporation is considered a high-performance alternative that provides a more mechanically controlled delivery environment when specialized equipment is available.
3. Why is collard regeneration significant for modern plant breeding technology?
Collard regeneration, particularly from protoplasts, is a critical breakthrough because it serves as the essential gateway for applying New Plant Breeding Technologies (NPBTs) to this valuable but often overlooked variety of Brassica oleracea. While techniques for other Brassica species existed, the first successful protocol for regenerating entire plants from collard protoplasts was only recently established.
The significance of this technology for modern breeding includes the following key areas:
1. Facilitating Precision Genome Editing
Regeneration is a fundamental requirement for genome editing tools like CRISPR/Cas9. Because the process of cell wall removal allows for the direct uptake of genetic material, researchers can edit genes at the single-cell level. Successful regeneration then allows these edited single cells to develop into whole, fertile plants that are genetically uniform, avoiding the common problem of chimerism found in other transformation methods.
2. Restoring and Broadening Genetic Diversity
The genetic diversity of collard has been significantly depleted over time due to the widespread use of commercial hybrids. Protoplast regeneration enables somatic hybridization (fusing the cells of different species), which allows breeders to:
- Bypass sexual incompatibility barriers to introduce beneficial traits from wild relatives or distant Brassica species.
- Transfer polygenic traits that are difficult to move through conventional breeding.
- Broaden the overall genetic pool available for future Brassica breeding.
3. Improving Disease Resistance and Agronomic Traits
Modern collard cultivars are highly susceptible to Fusarium yellow and black rot, which cause severe damage in infested soils. An established regeneration protocol allows for the use of biotechnological methods to introduce resistance to these and other bacterial and fungal diseases. It also facilitates the improvement of other multigenic traits like yield capacity and stress tolerance.
4. Enabling Transgene-Free Modifications
Protoplast regeneration is highly effective for “transgene-free” editing, where the plant’s genome is modified without integrating foreign DNA. This is significant because:
- It can shorten breeding timelines by achieving desired outcomes in a single clonal generation.
- It helps mitigate regulatory hurdles and public concerns associated with Genetically Modified Organisms (GMOs).
5. Pharmaceutical and Biotechnological Potential
Collard is being explored as a platform for molecular farming to produce recombinant antigens and pharmaceutical proteins. A robust regeneration system allows protoplasts to serve as reliable explants for both stable and transient transformation, which could facilitate the production of vaccines, such as those for smallpox.
4. What are the advantages of using alginate embedding over liquid culture?
Using alginate embedding for protoplast culture offers several significant advantages over liquid culture, primarily by providing a stable, semi-solid environment that physically separates individual cells.
The key advantages include:
- Prevention of Cell Aggregation: Protoplasts in liquid media are prone to aggregating. This aggregation can lead to the overproduction of toxic metabolites and non-homogeneous callus formation. In some cases, mass cell death from aggregation causes toxin buildup that affects the survival rate of the entire culture. Alginate embedding ensures physical separation, preventing these issues.
- Enhanced Viability and Efficiency: Protoplasts embedded in hydrogels, particularly calcium alginate, typically display greater cell survival, proliferation, and plating efficiency. Research in Brassica oleracea specifically found that alginate consistently enhanced cell division rates compared to other methods.
- Suitability for Heat-Sensitive Cells: Unlike agar or agarose, which must be heated to their melting point before they can be used for embedding, alginate gelling is induced by calcium ions at room temperature. This makes it a “non-heating option” that is highly favorable for fragile, heat-sensitive protoplasts.
- Individual Cell Tracking: Liquid media is generally not suitable for experiments requiring the tracking of individual cells. Solidifiers like alginate allow for the traceability of cells, which is essential for certain imaging and developmental studies.
- Reduced Chimerism: Because embedding ensures that regenerated plants are derived from single, physically separated cells, it significantly reduces the rates of plantlet chimerism often seen in liquid cultures where cell aggregates might form a single shoot.
- Protection from Physical Damage: Transferring an alginate gel layer onto regeneration media, rather than moving individual microcalli, minimizes physical damage to the developing tissues, which can further enhance regeneration efficiency.
- Simplified Handling: Alginate embedding can simplify the plating process and facilitate the exchange of media by allowing liquid nutrients to surround the gel without disturbing the fixed protoplasts.
5. How do genotype and medium composition affect shoot formation?
The efficiency of shoot formation from protoplasts is primarily governed by a combination of genotype-specific responses and the precise balance of hormones, carbon sources, and osmoticums within the culture media.
The Role of Genotype
Genotype is widely recognized as a dominant and often persistent influence on the success of plant regeneration protocols. Even within the same species, different cultivars or ecotypes exhibit widely varying regenerative capacities under identical conditions.
- Cultivar-Specific Success: In studies of Brassica oleracea, the cultivar ‘Kalibos’ showed the highest rate of shoot formation (6.6%), whereas ‘Red’ did not exceed 0.1%, and ‘Haco’ failed to regenerate shoots entirely, despite producing potentially regenerative green callus.
- Ecotype Variation: In Arabidopsis thaliana, the ecotype Ws-2 demonstrated high regeneration efficiency, while Col-0, No-0, and HR-10 were relatively ineffective regardless of varied media compositions.
- Hormonal Background: Different genotypes have unique endogenous hormonal balances. For example, the tomato cultivar ‘Micro-Tom’ requires a distinct medium compared to the ‘Red Setter’ cultivar, likely due to its altered levels of gibberellins and brassinosteroids.
Medium Composition and Phytohormones
The selection and ratio of plant growth regulators (PGRs) in the regeneration medium are critical for inducing organogenesis.
- Cytokinin-to-Auxin (C/A) Ratio: While a high C/A ratio is traditionally considered necessary for shoot induction, this requirement varies by species. In B. oleracea, a 1:1 ratio (Medium P) was found to be nearly ten times more efficient for shoot formation than a 5:1 ratio. Conversely, in tomato (Micro-Tom), cytokinin-rich media like Sl-SIM-T are essential for optimal regeneration.
- Gibberellins (GA3): Although GA3 is often omitted from regeneration media because it can hinder shoot and root formation, low concentrations (0.02 mg/l) have been shown to actually promote shoot formation in Brassica species.
- Other PGRs: Strong cytokinin analogues like thidiazuron (TDZ) have been successfully used to stimulate multiple shoot formation in recalcitrant species like pea.
Carbon Sources and Osmotic Regulation
The primary energy source and the osmotic pressure of the medium also significantly impact shoot regeneration efficiency.
- Sugar Combinations: While sucrose (1–3%) is the standard carbon source, specific combinations can enhance results. In B. oleracea, a medium supplemented with reduced sucrose (1%) and mannitol (2%) stimulated more effective shoot regeneration than the standard 3% sucrose.
- Osmotic Balance: Maintaining proper osmolarity (often using mannitol or sorbitol) is crucial to protect the structural integrity of the protoplast during the initial stages of culture and cell wall resynthesis. A gradual reduction of osmotic pressure as division progresses is often necessary to promote continuous proliferation.
Callus Quality and Medium Sequence
The developmental trajectory of shoots is heavily dependent on the quality of the intermediate callus. Well-structured, dense green callus is typically associated with high regeneration potential, whereas white, loose, or browning callus often fails to differentiate. Successful protocols often involve a sequence of specialized media—starting with a protoplast culture medium to support cell wall reconstitution, followed by a callus-inducing medium to develop pluripotent tissue capable of organogenesis.
6. How do PEG and electroporation affect cell viability?
Both PEG (polyethylene glycol) and electroporation significantly affect cell viability through distinct chemical and mechanical mechanisms, often requiring a delicate balance between transformation efficiency and the survival of fragile protoplasts.
Effects of PEG-Mediated Transformation
PEG affects viability primarily through chemical toxicity and physical disruption of the cell membrane.
- Mechanism of Action: PEG physically binds to cell membranes, disturbing the phospholipid bilayer and destabilizing the cell surface potential to allow DNA entry. This process can be inherently stressful for cells without cell walls.
- Concentration-Dependent Viability: Research indicates that as PEG concentration increases, the fraction of viable cells tends to decrease. For example, in tomato protoplasts, a 40% PEG concentration resulted in only approximately 20% healthy cells.
- Incubation Time: Prolonged exposure to PEG solutions (e.g., over 30 minutes) can result in severe protoplast destruction and reduced cell division.
- Post-Treatment Recovery: Because the debris from cells broken during treatment is detrimental to survivors in liquid culture, it is often necessary to embed PEG-treated protoplasts in a semi-solid matrix like alginate to ensure successful regeneration. Washing transfected cells twice with a stabilizing buffer helps mitigate PEG’s chemical toxicity.
Effects of Electroporation
Electroporation affects viability through mechanical and electrical damage caused by high-voltage pulses.
- Mechanism of Action: This method uses brief, high-voltage electrical pulses to temporarily induce microscopic pores in the plasma membrane. While it avoids chemical toxicity, it risks physical rupture of the cell.
- Sensitivity to Pulse Parameters: Cell survival is highly dependent on the meticulous standardization of voltage, pulse length, and pulse number. Sub-optimal conditions can cause immediate cellular death.
- Impact of Voltage and Pulses:
- Voltage: High electric field voltages (e.g., over 1 kV/cm) significantly decrease survival and division rates.
- Pulse Number: Research on gentian protoplasts found that a single pulse resulted in nine-fold higher viability than two pulses.
- Pulse Length: The duration of the pulse is critical; while a 20 μs pulse showed 70% viability, a 5 ms pulse was found to completely kill the protoplasts.
- Buffer Influence: The composition of the electroporation buffer (including pH and salt concentrations like KCl and MgCl2) significantly impacts the ability of the cells to survive the electrical discharge.
Comparative Impact on Viability
| Feature | PEG-Mediated Transformation | Electroporation |
|---|---|---|
| Type of Damage | Chemical toxicity and osmotic shock. | Mechanical damage and electrical rupture. |
| Key Factor | PEG concentration and incubation duration. | Voltage, pulse duration, and pulse number. |
| Mitigation Strategy | Immediate washing and embedding. | Meticulous parameter optimization. |
While electroporation can sometimes be nearly twice as efficient as PEG (as seen in cabbage), both methods typically result in relatively low overall survival and transformation rates compared to non-treated controls. Regardless of the method, the removal of the cell wall makes protoplasts exceptionally vulnerable to environmental changes and mechanical handling.
7. Which method is better for CRISPR/Cas9 delivery?
Determining the “better” method for CRISPR/Cas9 delivery depends on your specific goals, such as maximizing efficiency versus minimizing cost and technical complexity. The sources primarily compare PEG-mediated transformation and electroporation for delivering CRISPR components into plant protoplasts.
PEG-Mediated Transformation: The Most Accessible Method
PEG (polyethylene glycol) is generally considered the method of choice for many laboratories due to its accessibility and high throughput.
- Advantages: It is simple, low-cost, and requires no specialized equipment. It allows for the simultaneous processing of many samples.
- Efficiency: Success rates typically range between 30% and 50%. In tomato studies, PEG-mediated protoplast transfection was found to be approximately 12 times more efficient for gene editing than Agrobacterium-mediated transformation.
- Outcome: It has been used successfully to deliver CRISPR/Cas9 ribonucleoproteins (RNPs) in Solanum species (potato and tomato), yielding transgene-free edited plants.
Electroporation: A High-Performance Alternative
Electroporation is often cited as a more mechanically controlled delivery method that can achieve higher performance if specialized equipment is available.
- Advantages: It provides high repeatability and avoids the chemical toxicity sometimes associated with PEG.
- Efficiency: Demonstrated transfection efficiencies for CRISPR/Cas9 range from 20% to 30%. Notably, in an optimized study on cabbage protoplasts, electroporation was nearly twice as efficient as PEG delivery (3.4% vs. 1.8%).
- Disadvantages: It requires expensive, specialized hardware and meticulous standardization of voltage and pulse duration to avoid severe cellular damage.
Ribonucleoprotein (RNP) vs. Plasmid DNA Delivery
Regardless of the mechanical delivery method (PEG or electroporation), delivering CRISPR components as pre-assembled RNPs is increasingly considered superior to using plasmid DNA.
- Transgene-Free Results: RNP delivery is entirely “DNA-free,” circumventing the risk of random transgene integration into the host genome.
- Higher Precision: Because the Cas9 protein and guide RNA are already assembled, editing can occur faster, and the RNP complex degrades quickly within the cell, which significantly reduces off-target effects.
- Universal Application: RNPs eliminate the need for host-specific codon optimization or the construction of complex binary vectors.
8. Why Protoplast Delivery is Preferred Over Other Techniques
Protoplast-based delivery is considered better than traditional methods like Agrobacterium or particle bombardment for specific breeding goals:
- Avoiding Chimerism: Plants regenerated from a single transformed protoplast are genetically uniform, avoiding the common problem of chimeras (where only parts of the plant are edited) seen in multicellular explant transformation.
- Overcoming Host Range: Protoplast methods bypass the lack of host susceptibility to Agrobacterium, which is a major bottleneck in many monocots.
- Higher Edit Concentration: Techniques like PEG allow for the delivery of an “overwhelming amount” of editing complexes, resulting in substantially higher editing efficiency compared to Agrobacterium-mediated methods.
9. How does the genotype affect shoot formation efficiency?
The genotype is considered a dominant and often persistent influence on the success and efficiency of shoot formation in protoplast-to-plant regeneration. Even within the same species, different cultivars, ecotypes, or landraces exhibit widely varying regenerative capacities when subjected to the same culture conditions.
The impact of genotype on shoot formation efficiency is characterized by the following factors:
1. Significant Variation Within Species
Research consistently shows that shoot formation efficiency is highly cultivar-dependent:
- Brassica oleracea: In a study of seven cultivars, ‘Kalibos’ red cabbage demonstrated the highest shoot formation rate at 6.6%, while the cultivar ‘Red’ did not exceed 0.1%. Most notably, the cultivar ‘Haco’ failed to regenerate shoots entirely, despite producing dense green callus that appeared potentially regenerative.
- Arabidopsis thaliana: When comparing four ecotypes, Ws-2 showed high regeneration efficiency, whereas Col-0, No-0, and HR-10 landraces were relatively ineffective despite various optimizations to the media and tissue culture methods.
- Tomato (Solanum lycopersicum): Different cultivars require distinct media. For instance, the cultivar ‘Micro-Tom’ requires a different culture medium than ‘Red Setter,’ likely due to its unique endogenous hormonal balances, such as lower brassinosteroid and gibberellin levels.
2. Genetic Recalcitrance
A major bottleneck in new plant breeding technologies is that many important genotypes are recalcitrant, meaning they are extremely difficult to regenerate from single cells. Studies have reported that even with optimized protocols, a large portion of accessions within a species may fail to produce shoots. For example, in one evaluation of ten Brassica accessions, four failed to regenerate any shoots.
3. Dissociation Between Division and Regeneration
A high rate of initial protoplast division does not always guarantee high shoot formation efficiency. In Cyclamen species, it was observed that C. graecum had the highest protoplast division rates but performed the worst in actual plant regeneration. Similarly, in some B. oleracea cultivars, frequent initial cell divisions did not enhance microcallus formation, possibly due to the robust production of reactive oxygen species or phenolics during those early stages, which can lead to later growth inhibition.
4. Biological and Molecular Drivers
The efficiency differences between genotypes are driven by several underlying biological factors:
- Endogenous Hormonal Balance: Each genotype has its own sensitivity to plant growth regulators (auxins and cytokinins) and its own internal hormonal state.
- Cellular Competence: Success depends on “genotype-specific cellular competence,” which is the inherent ability of a cell to respond to signals and enter a regenerative pathway.
- Epigenetic Factors: Stochastic reactivation of stem cell regulators, which are typically under epigenetic silencing in somatic cells, is required for regeneration. The ease with which these regulators are reactivated varies by genotype.
- Stressor Sensitivity: Different genotypes react differently to the stress of cell wall removal (protoplasting), which triggers alterations in gene expression that can either stimulate or hinder the regeneration process.
10. How are protoplasts used for transgene-free genome editing?
Protoplasts are essential for transgene-free genome editing because their lack of a cell wall allows for the direct delivery of gene-editing components into the cell without the need for genomic integration. This approach allows researchers to achieve precise genetic outcomes in a single clonal generation while avoiding the regulatory hurdles and public concerns associated with transgenic crops.
Core Mechanisms for Transgene-Free Editing
- Ribonucleoprotein (RNP) Delivery: The most effective “DNA-free” method involves introducing pre-assembled CRISPR/Cas9 RNP complexes (consisting of the Cas9 protein and guide RNA) directly into the protoplast. Because these RNPs function transiently and degrade quickly within the cell, they induce the desired mutation without leaving any foreign DNA behind.
- Transient Expression: CRISPR components can also be delivered as mRNA or plasmid DNA. While plasmids carry a small risk of random integration, they are often used because they are highly accessible to researchers and can still yield transgene-free plants if the edit occurs before any genomic insertion.
- Direct Delivery Methods: The most common techniques for facilitating this uptake are PEG-mediated transformation (using polyethylene glycol to destabilize the membrane) and electroporation (using electrical pulses to create temporary pores).
The Path to Whole Plants
Once a protoplast is successfully edited, it must be regenerated into a whole plant through a multi-step tissue culture process.
- Avoiding Chimerism: Traditional transformation methods involving multicellular tissues often produce “chimeric” plants where only some cells are edited. Because a protoplast-derived plant originates from a single edited cell, the resulting plant is genetically uniform and the edit is stably inherited by the next generation.
- Selection and Fixation: Edited single cells can be rapidly screened, and those with the desired genotype are induced to form a callus, which then differentiates into shoots and roots.
Significance for Modern Breeding
- Speed and Efficiency: This technology can bypass the need for years of backcrossing required in conventional breeding to restore an “elite” genetic background after introducing a new trait.
- Overcoming Biological Barriers: Protoplast systems allow for the editing of species that are not susceptible to Agrobacterium—a common bottleneck for many monocot crops like wheat and maize.
- Application in Complex Crops: It is particularly valuable for sterile, slow-growing, or highly heterozygous perennial crops like grapevine, citrus, and potato, where removing transgenes through breeding is difficult or impossible.
Proven Success in Crops
Transgene-free, protoplast-based editing has been successfully applied to:
- Potato: Knocking out genes like StEDS1 and StPAD4 to improve resistance to pathogenic fungi.
- Citrus: Editing the CsLOB1 gene to produce plants resistant to citrus canker, which were subsequently approved as non-regulated by the USDA.
- Grapevine: Targeting the VviMlo6 gene to develop lines resistant to powdery mildew.
- Tobacco and Tomato: Replacing disease-susceptibility regions or knocking out viral resistance inhibitors.
11. What are the benefits of using calcium-alginate for immobilization?
Using calcium-alginate for the immobilization (embedding) of plant protoplasts offers several significant advantages over traditional liquid culture methods, primarily by providing a stable, semi-solid environment that physically separates individual cells.
The key benefits identified in the sources include:
1. Prevention of Cell Aggregation and Toxicity
Protoplasts cultured in liquid media are prone to aggregating, which can lead to the overproduction of toxic metabolites and the formation of non-homogeneous callus. Alginate embedding ensures physical separation, which prevents these issues and mitigates the accumulation of toxic substances released from dying cells that might otherwise inhibit the growth of neighboring cells.
2. Enhanced Viability and Plating Efficiency
Protoplasts embedded in hydrogels, particularly calcium-alginate, consistently display greater cell survival, proliferation, and plating efficiency compared to other culture methods. In studies of Brassica oleracea, for example, the use of alginate was found to consistently enhance cell division and plating efficiency across various cultivars.
3. Suitability for Heat-Sensitive Cells
Unlike solidifiers such as agar or agarose, which must be heated to their melting point before they can be used for embedding, alginate gelling is induced by calcium ions at room temperature. This “non-heating” process is highly favorable for fragile, heat-sensitive protoplasts that might be damaged by the temperatures required for other matrices.
4. Traceability and Reduced Chimerism
- Individual Cell Tracking: Solid media like alginate are more appropriate for imaging studies because they allow for the traceability of individual cells, whereas liquid media do not support tracking single-cell development.
- Genetically Uniform Plants: Because embedding ensures that regenerated plants are derived from single, physically separated cells, it significantly reduces the rates of plantlet chimerism often seen in liquid cultures where cell aggregates might form a single shoot.
5. Protection from Physical Damage and Simplified Handling
- Minimized Damage: Transferring an entire alginate gel layer onto regeneration media, rather than moving individual microcalli, minimizes physical damage to the developing tissues, which can further enhance shoot regeneration efficiency.
- Easier Nutrient Exchange: Alginate embedding simplifies the plating process and facilitates the exchange of media by allowing liquid nutrients to surround the gel and pass through to the cells without disturbing the fixed protoplasts.
12. How do you count and test protoplast viability?
The most prevalent methods for counting and testing the viability of isolated protoplasts involve the use of a hemocytometer and fluorescein diacetate (FDA) staining, respectively.
Counting Protoplasts
- Methodology: Protoplast yield is reliably estimated by cell counting using a hemocytometer (often specified as a Fuchs Rosenthal chamber) under an inverted microscope.
- Yield Calculation: The yield is typically presented as the number of protoplasts per gram of fresh weight (FW) of the source tissue.
- Procedure: After purification and centrifugation, the protoplast pellet is resuspended in a known volume of stabilizing solution (such as mannitol). A small aliquot is then loaded into the hemocytometer for counting.
- High-Throughput Alternatives: In some contexts, a flow cytometer may be used for rapid, automated counting and assessment.
Testing Protoplast Viability
The quality of isolated protoplasts is critical, as only viable cells can successfully undergo transformations or regenerate into whole plants.
- FDA Staining: This is the standard technique for assessing health. Fluorescein diacetate (FDA) is a non-fluorescent compound that permeates cell membranes. In living cells, enzymes degrade FDA into yellow-green fluorescent compounds. Non-viable protoplasts lack this enzymatic activity and remain dark.
- Procedure: A working solution of FDA is added to the protoplast culture (e.g., 100 µl of solution left for 15 minutes in the dark). The ratio of fluorescent cells to the total number of cells is observed under a fluorescence microscope.
- Calculations: For statistical significance, observations are typically performed on a minimum of 400 to 500 cells per treatment.
- Alternative Dyes: Other stains used for determining viability include phenosafranine and 5-carboxyfluorescein diacetate.
Visual and Physical Indicators of Viability
Beyond staining, researchers use several physical and morphological markers to gauge protoplast health:
- Shape: Healthy, intact protoplasts are characterized by a perfectly round (spherical) shape. Broken or dysfunctional cells appear as debris or cell fragments.
- Buoyancy: During purification via centrifugation, protoplasts in good condition will typically float to the top layer.
- Autofluorescence: Under certain microscope filters (like FITC), dead protoplasts may display intense yellow-range fluorescence, whereas living suspension or root-derived protoplasts exhibit only faint blue autofluorescence.
- Cell Wall Resynthesis: The ability of a protoplast to rebuild its cell wall—tracked using calcofluor white staining—is a strong indicator of metabolic activity and potential for mitotic division.
13. What are the common causes of low protoplast viability?
Common causes of low protoplast viability span the entire process from source tissue selection and isolation to transformation and subsequent culture. The sources identify the following primary factors:
1. Isolation and Enzymolysis Stress
- Extended Incubation Time: Prolonged exposure to cell-wall-degrading enzymes is a major cause of cell death. While digestion is necessary to release protoplasts, exceeding optimal durations (e.g., more than 8 hours for tomato or 6 hours for jasmine callus) leads to protoplast breakage and dysfunction.
- Enzyme Concentration and Composition: High concentrations of enzymes like cellulase or the presence of impurities in crude enzyme extracts can be toxic. Some enzymes act as biochemical sorters, where specific compositions may interact negatively with certain cell cycle phases.
- Mechanical Damage: Because they lack a cell wall, protoplasts are extremely fragile. Excessive mechanical stress during maceration, vigorous pipetting, or high-speed centrifugation (especially sudden braking) can physically rupture the plasma membrane.
2. Osmotic and Chemical Shocks
- Improper Osmolarity: Protoplasts require a isotonic environment to maintain structural integrity. Improper osmotic pressure during isolation or culture can cause them to burst or shrivel.
- Osmotic Shock: Rapid changes in the concentration of osmotic stabilizers (like mannitol or sorbitol) when transferring cells between solutions can lead to lethal osmotic shock.
3. Transformation Procedures
- PEG Toxicity: Polyethylene glycol, while an effective fusogen, is inherently chemically toxic. Viability tends to decrease as PEG concentration increases; for instance, at 40% PEG, healthy cells may drop to only 20%. Exposure exceeding 30 minutes can cause severe protoplast destruction.
- Electroporation Damage: High-voltage electrical pulses can cause irreversible mechanical and electrical damage. Parameters such as high electric field voltage (over 1 kV/cm), long pulse durations (e.g., 5 ms), or multiple pulses have been shown to significantly decrease survival or result in total cell death.
4. Source Tissue and Biological Factors
- Tissue Age and Health: Protoplasts derived from older, senescing, or stressed tissues generally exhibit much lower viability and regenerative capacity than those from young, vigorously growing tissues.
- Donor Plant Growth Conditions: Environmental factors during the growth of donor plants, such as improper light, temperature, or nutrient supply, directly influence the quality and survival rate of isolated protoplasts.
- Genotype Dependence: Viability and the ability to survive the “stress of protoplasting” are highly genotype-specific. Some cultivars or ecotypes are naturally more recalcitrant and prone to death during the procedure regardless of optimization.
5. Culture Environment
- Toxic Accumulation (Browning): The accumulation of phenolics and reactive oxygen species (ROS) can lead to oxidative browning of the media, which inhibits growth and eventually kills the protoplasts.
- Cell Aggregation: In liquid cultures, protoplasts are prone to aggregating. This can cause a local buildup of toxic metabolites released from dying cells, which then inhibits the survival of neighboring healthy cells.
- Overcrowding: Plating at an excessively high density can result in competition for nutrients and accelerated toxin buildup, leading to mass culture collapse.
- Inappropriate Light and Temperature: Extremes in temperature or exposure to high light intensities (which can cause bleaching in some species) are detrimental during the early stages of cell wall resynthesis.
14. What causes the browning or failure of some callus clumps?
The browning and subsequent failure of callus clumps in plant protoplast cultures are primarily driven by the accumulation of toxic substances and the biological response of specific genotypes to stress.
1. Accumulation of Phenolics and Oxidative Stress
The most common cause of browning is the accumulation of phenolics within the culture medium. Phenolic compounds can undergo oxidation—often catalyzed by enzymes like polyphenol oxidase—leading to oxidative browning, which inhibits further growth and cell division. This process is frequently linked to the production of reactive oxygen species (ROS) during the initial stages of culture. Extensive or frequent cell divisions in the early stages can trigger robust ROS and phenolic production, resulting in oxidative stress-related inhibition of growth at later stages.
2. Hormonal Imbalance
The concentration of plant growth regulators (PGRs) in the medium significantly impacts callus health. Specifically, high concentrations of auxin have been found to frequently induce callus browning, which is associated with lower shoot regeneration rates. While auxins are necessary for callus initiation, excessive levels can be detrimental to the long-term proliferation and differentiation of the tissue.
3. Cell Aggregation and Overcrowding
Physical factors during culture play a major role in clump failure:
- Aggregation in Liquid Media: Protoplasts cultured in liquid media are prone to aggregating. This aggregation leads to a local buildup of toxic metabolites released from dying cells, which can poison the surrounding healthy cells and cause mass culture collapse.
- Overcrowding: Plating protoplasts at excessively high densities leads to competition for nutrients and accelerates the buildup of toxins, which negatively affects the growth of the entire plate.
4. Genotype-Specific Recalcitrance
The tendency for callus to brown is often genotype-dependent. Some cultivars or species are naturally more susceptible to “lethal browning” after subculturing regardless of the media used. In studies of Brassica oleracea, for example, some cultivars failed to develop beyond the green callus stage or turned brown and ceased development entirely due to their specific genetic makeup.
5. Media and Environmental Management
Failure to regularly refresh the culture medium can lead to the depletion of nutrients and the unchecked accumulation of the toxic byproducts mentioned above. To mitigate these issues, researchers often use additives such as:
- Polyvinylpyrrolidone (PVP): To adsorb and counteract phenolics.
- Activated Charcoal: To adsorb inhibitory elements and growth inhibitors that cause browning.
- Antioxidants: Such as ascorbic acid or L-cysteine, to mitigate the inhibitory effects of ROS.
15. What are the common ploidy levels in regenerated plantlets?
While the primary goal of most regeneration protocols is to maintain the original genetic complement of the donor plant, regenerated plantlets frequently exhibit a range of ploidy levels due to somaclonal variation and the inherent instabilities of the tissue culture process.
The common ploidy levels and the factors influencing them include:
1. Diploid (2x)
In many optimized protocols, the majority of regenerated plants remain diploid, matching the ploidy of the source tissue.
- Brassica oleracea: A study of 176 regenerants across five cultivars found that 79.5% were diploid.
- Solanum peruvianum: Research on this wild tomato species showed no evidence of chromosome instability, with regenerants maintaining their diploid state.
- Urochloa ruziziensis: Using young embryogenic calli (two months old or less) is recommended to ensure the generation of fertile diploid plants.
2. Tetraploid (4x) and Polyploid
The shift from diploid to tetraploid is a frequent occurrence in protoplast regeneration.
- Spontaneous Polyploidization: In B. oleracea, 18.2% of regenerants were identified as tetraploids. This is often attributed to spontaneous fusion of two protoplasts during the isolation or early culture phase.
- Species-Specific Tendencies: In Gentiana decumbens, 100% of regenerated plants were found to be tetraploid, despite being derived from diploid protoplasts.
- Higher Levels: Long-term cultures of ruzigrass have been known to produce octoploids (8x) in addition to tetraploids.
- Synthetic Hybrids: Somatic hybridization is often used specifically to create higher ploidy levels, such as citrus autotetraploids or allohexaploid Brassica.
3. Mixoploid (2x–4x)
Mixoploidy occurs when a single plant contains cells with different chromosome numbers.
- In B. oleracea studies, 2.3% of regenerants exhibited mixed ploidy.
- This condition is likely caused by in vitro-induced disruption of cytokinesis and subsequent fusion of daughter nuclei during the proliferation of the callus, leading to a mitotically unstable tissue.
4. Aneuploid and Aberrant Complements
Aneuploidy (an abnormal number of chromosomes that is not a multiple of the haploid set) and other rearrangements can also occur.
- Potato (Solanum tuberosum): Protoplast regeneration in autotetraploid potato often induces widespread aneuploidy and genome rearrangements. This may be due to the higher rate of chromosome mis-segregation during mitosis in polyploid species compared to diploids.
- Arabidopsis: Cultures maintained for long durations in callus media can yield plants with aberrant chromosome complements.
Drivers of Ploidy Variation
- Systemic Endopolyploidy: Some tissues, such as Arabidopsis mesophyll or root vascular meristems, possess a natural range of ploidy levels (endopolyploidy), which can be reflected in the resulting regenerants.
- Culture Conditions and Stress: Environmental stressors like salt stress have been shown to induce polyploidy in carrot regenerants. Additionally, high levels of 2,4-D in callus media can reduce the efficiency of normal shoot differentiation and lead to chromosomal aberrations.
- Growth Advantage: It is hypothesized that in some cultures, tetraploid cells may divide faster than diploid cells, allowing them to eventually dominate the population of regenerated shoots.
- Endoreduplication: This process, where DNA is amplified without subsequent mitosis, is a known cause of increased DNA content in callus cells, which can then lead to polyploid shoots.
16. How does enzyme concentration impact protoplast isolation yield?
Enzyme concentration is one of the most critical factors influencing the yield and quality of isolated protoplasts. The relationship between concentration and yield is generally characterized by an optimal peak, where concentrations that are either too low or too high result in significantly reduced yields.
1. Optimal Concentration Ranges
The specific concentration required for maximum yield varies significantly depending on the plant species and the type of source tissue.
- General Standards: For many species, such as tomato and eggplant, a cellulase concentration of 1.5% to 2% (w/v) is typically recommended.
- Species-Specific Optima:
- Arabidopsis: Digestion of proliferating root tissues with 2.0% to 2.5% cellulase for 3–5 hours was found to be optimal for protoplast release.
- Stevia: Research on stevia leaves found that 2% cellulase provided the highest yield. Notably, both decreasing the concentration to 1% and increasing it to 3% resulted in a significant drop in yield and viability.
- Jasmine: The highest yield from jasmine callus was achieved using 1.5% cellulase.
- Potato: A concentration of 1% cellulase was shown to be effective for leaf and tuber tissues.
2. Consequences of Sub-optimal Concentrations
- Insufficient Concentration: If the enzyme concentration is too low, the cell wall is not fully degraded, leading to a failure to release the protoplasmic mass and a resulting low yield.
- Excessive Concentration: High concentrations can be chemically toxic to the cell. Overly concentrated enzymes can cause protoplast breakage, dysfunction, and immediate cellular damage, which reduces the count of viable, intact cells.
3. Interaction with Tissue Composition
The required concentration is directly influenced by the biochemical composition of the cell wall in the source material.
- Cellulose Density: Tissues with cellulose-rich cell walls, such as those in older tomato or eggplant tissues, often necessitate higher concentrations of cellulase to achieve complete digestion.
- Tissue Age: Younger, actively growing tissues (like 4-week-old leaf blades) generally have thinner cell walls that are more susceptible to enzymatic digestion, often allowing for lower concentrations or shorter incubation times compared to mature tissues.
4. Synergy and “Biochemical Sorting”
Yield is often improved not just by increasing a single enzyme’s concentration, but by using an optimized mixture of enzymes (cellulase, pectinase, and sometimes hemicellulase or macerozyme).
- Step-wise Digestion: Pectinase typically targets the middle lamella to separate adjacent cells, allowing the cellulase to more effectively degrade individual cell walls and release protoplasts.
- Cell Cycle Influence: Some research suggests that certain enzymes act as “biochemical sorters,” interacting differently with cells at various stages of the cell cycle. Therefore, the specific balance of enzyme concentrations can determine the morphogenetic potential (ability to divide later) of the isolated population.
5. Yield vs. Quality Balance
While higher concentrations might speed up the release of protoplasts, they may also act as pathogenic elicitors. A concentration that provides the absolute highest yield of isolated cells may not always yield the highest quality cells for subsequent regeneration into whole plants. Meticulous standardization is required to find the concentration that maximizes both quantity and viability.
17. What is the role of mannitol in stabilizing protoplasts?
Mannitol plays a critical role as an osmotic stabilizer (or osmoticum) throughout the entire process of protoplast isolation, culture, and transformation. Because protoplasts lack a rigid cell wall, they are extremely fragile and susceptible to bursting or shriveling due to improper osmotic pressure.
The specific roles of mannitol include:
1. Facilitating Isolation (Pre-treatment and Enzymolysis)
- Pre-plasmolysis: Before cell wall digestion, tissue is often treated with a mannitol solution (typically 0.4 M to 0.6 M). This induces plasmolysis, shrinking the cytoplasm away from the cell wall. This protection prevents damage to the plasma membrane during enzymatic digestion and improves the yield of intact cells.
- Maintaining Osmolality: During the hours-long enzyme treatment, mannitol is included in the buffer to balance the internal pressure of the cell. This prevents osmotic shock, which would otherwise lead to the rupture of the protoplasts as their walls are removed.
2. Preserving Structural and Functional Integrity
- Structural Support: Mannitol acts as a metabolically inert sugar that provides physical support to the cell membrane. It maintains the spherical shape of the protoplasts in liquid or semi-solid environments.
- Stabilizing Solutions: It is a standard component in washing and storage solutions (such as MMg solution) used to remove residual enzymes and keep cells viable before they are used for experiments.
3. Supporting Transformation and Culture
- Transformation Buffers: In both PEG-mediated and electroporation methods, mannitol is used to stabilize the cells during the entry of exogenous DNA. It helps the cells declump and regain their shape after the stress of chemical or electrical treatment.
- Initial Culture Support: During the first stage of culture, when the cell wall has not yet been rebuilt, mannitol provides the necessary osmotic pressure to ensure protoplast vigor.
- Gradual Weaning: As protoplasts begin to resynthesize their cell walls and enter mitotic division, their need for high external osmotic pressure decreases. Protocols often involve a gradual reduction of mannitol concentration to promote continuous cell proliferation and microcallus formation.
4. Thermal and Environmental Stability
- Temperature Buffering: Because wall-less protoplasts are highly temperature-sensitive, mannitol is added as a stabilizing agent to help prevent rupture during handling.
- Metabolic Role: While mannitol is often considered physiologically inactive as an energy source compared to sucrose or glucose, its presence in regeneration media has been shown in some species, like Brassica oleracea, to stimulate more effective shoot regeneration when used in combination with other sugars.
18. How do you optimize PEG concentration for specific cultivars?
Optimizing PEG (polyethylene glycol) concentration for specific cultivars requires a careful balancing act between maximizing transformation efficiency and maintaining high cell viability. Because different genotypes exhibit widely varying sensitivities to the chemical toxicity and osmotic stress of PEG, a one-size-fits-all approach is generally ineffective.
Based on the sources, the optimization process involves the following strategies:
1. Establish a Basal Range and Test Incrementally
While the common range for PEG concentration in plant protoplast protocols is 10% to 40%, specific cultivars may require much lower levels to survive.
- Case Studies: In eggplant protoplasts, transformation efficiency was found to increase up to 40% PEG and then decrease slightly, with an optimal efficiency of 53% at the 40% mark. Conversely, in potato leaf protoplasts, 12.5% PEG was found to be superior to 20% PEG for subsequent callus formation, even though both treatments significantly reduced growth compared to untreated controls.
- Standard Starting Point: Many protocols for Arabidopsis and Solanum species utilize a 20% to 40% PEG 4000 or 6000 solution as a starting point for optimization.
2. Monitor the Efficiency-Toxicity Trade-off
Optimization must prioritize the intended outcome: transient expression (which often tolerates higher PEG for better efficiency) versus stable regeneration (which requires high cell survival).
- Viability Testing: Use fluorescein diacetate (FDA) staining to assess health after PEG exposure. In tomato protoplasts, increasing PEG from 10% to 40% caused a sharp decline in the fraction of viable cells; at 40% PEG, only about 20% of cells remained healthy.
- Visual Indicators: If PEG-treated protoplasts appear damaged or fail to “declump” and regain their spherical shape after dilution, the PEG concentration is likely too high and should be reduced.
3. Account for Genotype-Specific Recalcitrance
The success of PEG-mediated transformation is highly genotype-dependent.
- Endogenous Balance: Cultivars differ in their internal hormonal state and chromatin accessibility, which influences how they respond to the “stress of protoplasting” and chemical fusogens.
- Regenerative Capacity: Some cultivars may show high transformation efficiency but fail to regenerate into whole plants because the PEG treatment, even if successful at delivering DNA, inhibits the subsequent mitotic divisions required for callus formation.
4. Optimize Incubation Duration and Molecular Weight
PEG concentration cannot be optimized in isolation; it must be paired with specific durations and molecular weights.
- Exposure Time: Standard incubation typically lasts 10 to 30 minutes. Exceeding 30 minutes of PEG exposure often results in severe protoplast destruction regardless of the concentration.
- Molecular Weight: Higher molecular weight PEG (e.g., 6000 vs. 4000) can lead to higher fusion or transformation frequencies but may result in tighter cell adhesion that makes recovery of viable fused cells more difficult.
5. Post-Treatment Recovery
To mitigate the toxicity of the optimized PEG concentration, transfected cells should be washed twice with a stabilizing buffer (often containing salts like NaCl, CaCl2, and KCl) before being plated or embedded in an alginate matrix. Embedding is particularly recommended for PEG-treated cells because the debris from broken cells is detrimental to the continued liquid culture of survivors.
19. What additives help prevent callus browning?
The accumulation of phenolics and reactive oxygen species (ROS) during culture often leads to oxidative browning, which inhibits growth and can cause mass cell death. Several additives are used in isolation and culture media to counteract this process:
1. Adsorbing Agents
- Polyvinylpyrrolidone (PVP): This is a widely used additive that adsorbs phenolic compounds, preventing them from oxidizing and browning the media. It is frequently included in isolation buffers and regeneration media at concentrations ranging from 0.5% to 2.0%.
- Polyvinylpolypyrrolidone (PVPP): A highly cross-linked version of PVP that has been found to prevent browning even more effectively than standard PVP in certain species, such as guar.
- Activated Charcoal: This additive is used for its ability to adsorb inhibitory elements, including phenolics and growth inhibitors that cause callus discoloration. However, its concentration must be carefully optimized; for example, while 0.02% (w/v) was ideal for chrysanthemum, a higher concentration of 0.1% caused the cells to die.
2. Antioxidants
- General Antioxidants: Supplements such as ascorbic acid, citric acid, reduced glutathione, and L-cysteine are used to mitigate the inhibitory effects of ROS.
- Ascorbic Acid: In oil palm cultures, the addition of 200 mg/L ascorbic acid significantly reduced discoloration and promoted the development of healthy embryogenic calli.
3. Enzyme Inhibitors
- 2-aminoindane-2-phosphonic acid (AIP): This is a reversible inhibitor of phenylalanine ammonia lyase (PAL), the enzyme required for polyphenol production. While early addition can increase protoplast survival and sustained division, extended use should be avoided as it can eventually hinder shoot and root growth.
4. Other Chemical Regulators
- Polyamines: Compounds like putrescine, spermidine, and spermine can regulate oxidative stress and increase antioxidant activity. In cabbage cultures, the exogenous application of putrescine was specifically linked to higher frequencies of shoot organogenesis.
- Silver Nitrate (AgNO3): An inhibitor of ethylene action, silver nitrate has been shown to increase callus formation and regeneration efficiency in several Brassica and rice species.
- Cefotaxime and Timentin: While primarily used as antibiotics to prevent contamination, these substances have been observed to increase regeneration efficiency in species like carrot and hydrangea.
20. How do temperature and light affect regeneration?
Temperature and light are fundamental environmental factors that directly influence every stage of the protoplast-to-plant regeneration process, from the quality of the starting material to the final acclimatization of the plant.
1. Impact on Initial Protoplast Culture and Division
During the early stages of culture, when protoplasts are rebuilding their cell walls and initiating mitotic divisions, their sensitivity to light and temperature is at its highest.
- Preference for Darkness: Protoplasts generally prefer darkness and room temperature (typically around 24–25°C) during the first stages of culture. For species like lettuce, light can be lethal, bleaching and killing protoplasts within three days, while dark culture allows for sustained division. Conversely, Arabidopsis cotyledon protoplasts appear less sensitive, showing no significant difference in growth rates between light and dark conditions.
- Temperature Stability: Maintaining a consistent temperature is crucial for the success of microcallus formation. For cabbage, moving cultures from 25°C in the dark to 23°C in the light after only seven days resulted in very few cell divisions compared to those kept at a constant 25°C in the dark for 15 days.
- Cell Wall Regeneration: On-chip studies have shown that the absence of light can strongly decrease the elongation process in moss protoplasts, though growth restarts quickly once illumination returns.
2. Role in Shoot and Root Morphogenesis
Once the protoplasts have developed into microcalli, light becomes a necessary stimulus for inducing organogenesis.
- Inducing Green Callus: Light is typically required for callus to expand and turn green, a prerequisite for shoot differentiation in many species. For Brassica oleracea and tomato, shoot induction media are maintained under a 16-hour photoperiod at approximately 24–25°C.
- LED Quality: The quality of the light source significantly affects morphogenesis. Monochromatic LEDs are often more efficient than conventional fluorescent lights. For example, red LEDs are efficient for callus and shoot proliferation in several ornamentals, while combinations of red and blue LEDs enhance plant development by increasing net photosynthesis.
- Thermal Stress on PLBs: In orchids, protocorm-like body (PLB) regeneration is highly sensitive to temperature; any deviation—higher or lower—from the optimum acts as a stress factor that can inhibit organogenesis.
3. Influence on Source Tissue and Isolation
The environmental conditions of the donor plant directly determine the “competence” of the resulting protoplasts.
- Photoperiod of Donor Plants: In Arabidopsis, plants grown under a short-day photoperiod (10 h) produced protoplasts with a fourfold higher plating efficiency than those grown under long-day (16 h) conditions.
- Isolation Conditions: Most enzymolysis (digestion) protocols are conducted in the dark to avoid photoinhibition and the production of reactive oxygen species (ROS) in cells containing chloroplasts. However, some species, like geranium, actually show higher yield and viability when digestion occurs in the light.
- Minimizing Shocks: It is generally recommended to use isolation temperatures close to the growth conditions of the source material to minimize thermal shocks that could damage fragile wall-less cells.
4. Environmental Stress and Genetic Regulation
Light and temperature also serve as regulatory cues for specific breeding traits.
- Male Sterility: In environment-sensitive genic male sterile (EGMS) lines, temperature and photoperiod are used to control fertility. For instance, a thermosensitive rapeseed line is fertile at temperatures above 20°C but undergoes altered gene expression leading to sterility at different temperatures.
- Acclimatization Hardening: The final stage of regeneration requires lower temperatures (e.g., 19°C for cabbage) and higher light intensities to harden the plantlets for ex vitro conditions. During this phase, light intensity and humidity are gradually adjusted to help the plants transition from the protected in vitro environment to soil.
21. How does FDA staining reveal protoplast metabolic activity?
Fluorescein diacetate (FDA) staining reveals protoplast metabolic activity by utilizing the ability of living cells to enzymatically convert a non-fluorescent compound into a fluorescent one. This process serves as a standard technique for assessing the health and vitality of isolated protoplasts.
The mechanism involves the following steps:
- Membrane Permeation: FDA is a non-polar, non-fluorescent compound that has the unique ability to permeate the intact plasma membrane of a protoplast.
- Enzymatic Degradation: Once inside a living, metabolically active cell, intracellular enzymes (specifically esterases) degrade or hydrolyze the FDA.
- Fluorescence Production: This enzymatic reaction converts the FDA into fluorescein, a polar, yellow-green fluorescent compound.
- Visual Identification: Because fluorescein is polar, it is trapped within the cell membrane of viable protoplasts. When observed under a fluorescence microscope using a suitable filter set (such as FITC), these active cells emit a bright green fluorescence.
- Indicator of Non-Viability: Protoplasts that are dead or have severely compromised metabolism lack the necessary enzymatic activity to degrade the FDA. Consequently, these cells do not emit fluorescence and remain dark under the microscope.
Researchers typically estimate viability by calculating the ratio of fluorescent (green) protoplasts to the total number of cells observed. This assessment is often performed shortly after isolation to ensure the quality of the population before proceeding with genetic transformations or regeneration protocols.
22. What is the effect of culture medium on microcallus formation?
The composition and management of the culture medium are identified as key factors ensuring the viability of protoplasts and the successful transition from single cells to microcalli. The effect of the medium is characterized by the precise balance of nutrients, plant growth regulators (PGRs), osmotic stabilizers, and anti-browning additives.
1. Influence of Plant Growth Regulators (PGRs)
The ratio and concentration of auxins and cytokinins are critical for promoting the initial mitotic divisions that lead to microcallus formation.
- Genotype-Specific Requirements: Optimal PGR combinations vary significantly by species. For example, cytokinin-rich media are recommended for tomato (Sl-PIM-hCk and Sl-CIM-hCk) and Kalanchoe, as high auxin levels can induce callus browning. Conversely, some species, like Populus, require a higher auxin-to-cytokinin ratio for effective microcallus formation.
- Detrimental Effects of High Concentrations: Excessive concentrations of PGRs, particularly 2,4-D, can have a slightly detrimental effect on microcallus proliferation.
2. Osmotic Stabilization and Carbon Sources
The energy source and osmotic pressure of the medium must be carefully managed to maintain the structural integrity of wall-less protoplasts.
- Initial Stabilization: In the first stage of culture, osmotic stabilizers like mannitol, sorbitol, or myo-inositol prevent osmotic shock. For cabbage cotyledon protoplasts, myo-inositol has been found superior to mannitol as an osmotic regulator.
- Gradual Osmotic Reduction: Protoplast proliferation media are often sub-optimal for sustained microcallus growth. A gradual reduction of osmotic pressure (weaning the cells off stabilizers) is necessary to promote continuous proliferation as the cell wall is resynthesized.
- Carbon Sources: Energy sources like sucrose and glucose directly affect the division rate and colony generation. In chrysanthemum, 1% sucrose was found more effective for rapid microcallus formation than 2% sucrose.
3. Prevention of Browning and Toxin Buildup
A major challenge in microcallus formation is the accumulation of toxic metabolites and phenolics, which cause the medium to brown and inhibit growth.
- Adsorbing Agents: Additives such as Polyvinylpyrrolidone (PVP) and activated charcoal are frequently included to adsorb phenolics and growth inhibitors.
- Antioxidants: Supplements like ascorbic acid, citric acid, and L-cysteine mitigate the inhibitory effects of reactive oxygen species (ROS) produced during extensive cell division.
- Media Refreshment: Regular dilution or refreshment of the culture medium is essential to provide fresh nutrients and remove accumulated toxins.
4. Physical State of the Medium
The choice between liquid and semi-solid culture significantly impacts development.
- Liquid Media: Some species, such as Arabidopsis, divide earlier and reach higher frequencies in liquid MSAR1 medium. However, liquid culture can lead to cell aggregation, resulting in non-homogeneous callus and the buildup of toxic substances from dying cells.
- Embedding/Solid Media: Embedding protoplasts in a semi-solid matrix like calcium alginate provides physical separation, which prevents aggregation and has been shown to consistently enhance plating efficiency and microcallus formation in Brassica oleracea.
5. Medium Composition Comparisons
Studies in B. oleracea demonstrated that different media formulations result in varying prolificacy. For instance, the CPPO1 medium produced a significantly higher number of microcalli per dish compared to Bras4 or Bras5 media. Similarly, in tomato ‘Micro-Tom’, high-cytokinin media showed significantly higher callus formation efficiency than high-auxin alternatives.
23. How do cellulase and pectinase differ in cell wall digestion?
Cellulase and pectinase serve distinct but complementary roles in plant cell wall digestion, primarily differing in their molecular targets and the mechanical effect they have on plant tissue.
Primary Molecular Targets
- Cellulase: This enzyme complex targets cellulose, which is the primary structural component of the plant cell wall. It functions by hydrolyzing high-molecular-weight cellulose molecules into lower-molecular-weight molecules, eventually breaking them down into simple monosaccharides.
- Pectinase: This enzyme targets pectin, a polysaccharide found predominantly in the middle lamella—the layer that “glues” adjacent plant cells together. It catalyzes the hydrolysis of pectin into β-galacturonide, facilitating its detachment from individual cells.
Mechanical Effects on Tissue
- Cellulase: Its primary role is the actual degradation of the cell wall itself to liberate the protoplasmic mass. Without cellulase, the structural integrity of the cell remains intact, and the protoplast cannot be released.
- Pectinase: Its primary role is the separation of adjacent cells from one another. By digesting the middle lamella, pectinase turns solid tissue into a suspension of individual cells, which significantly increases the surface area available for cellulase to act upon.
Combined and Sequential Action
In standard protoplast isolation protocols, it is common practice to blend these enzymes for a synergistic effect:
- Pectinase is utilized initially (or simultaneously at the start) to degrade pectin and separate the cells.
- Cellulase then performs the sustained action of degrading the individual cell walls to release the protoplasts.
- Some complex mixtures, like macerozyme, contain both cellulase and pectinase activities, along with hemicellulase, to ensure more thorough digestion of various cell wall components.
Specialized Biological Roles
Interestingly, research suggests that pectinases can act as “biochemical sorters“. Different types of pectinases may interact specifically with cells that are in different phases of the cell cycle. Consequently, the choice of pectinase can influence the morphogenetic potential (the ability to divide and regenerate later) of the isolated protoplasts during the culture stage.
24. How long should protoplasts be incubated during electroporation recovery?
Immediately following the high-voltage electrical pulse, protoplasts should be incubated for 20 to 30 minutes at room temperature for recovery. During this period, the cells should remain undisturbed.
Following this initial brief recovery, the process continues as follows:
- Media Addition: After the 20- to 30-minute recovery, protoplast culture medium is added to the cuvette, and the suspension is transferred to a petri dish.
- Expression Incubation: The protoplasts are then incubated for a longer duration of 6 to 18 hours to allow for gene expression and protein production.
- Temperature Specifics: The temperature for this longer incubation depends on the species: 27°C for Arabidopsis cells and 26°C for tobacco BY-2 cells.
This recovery phase is critical because electroporation temporarily creates pores in the cell membrane to allow DNA entry, and sub-optimal conditions or failure to allow for recovery can lead to severe cellular damage or death.
References
Ahmed, M. A. A., Miao, M., Pratsinakis, E. D., Zhang, H., Wang, W., Yuan, Y., Lyu, M., Iftikhar, J., Yousef, A. F., Madesis, P., & Wu, B. (2021). Protoplast isolation, fusion, culture and transformation in the woody plant jasminum spp. Agriculture (Switzerland), 11(8). https://doi.org/10.3390/agriculture11080699
Bugakova, D., Loessberg-Zahl, J., & Segerink, L. I. (2026). Protoplasts on-chip: State-of-the-art and current progress. Sensors and Actuators A: Physical, 397, 117252. https://doi.org/10.1016/j.sna.2025.117252
Chen, K., Chen, J., Pi, X., Huang, L. J., & Li, N. (2023). Isolation, Purification, and Application of Protoplasts and Transient Expression Systems in Plants. In International Journal of Molecular Sciences (Vol. 24, Number 23). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/ijms242316892
Hsieh, J. A., Wu, F., Yang, D., Wu, A., Liu, C., Chen, C., Lin, S., Lin, Y. J., & Lin, C. (2026). Protoplast‐Based Functional Genomics and Genome Editing: Progress, Challenges and Applications. Plant, Cell & Environment. https://doi.org/10.1111/pce.70375
Jeong, Y. Y., Noh, Y. S., Kim, S. W., & Seo, P. J. (2024). Efficient regeneration of protoplasts from Solanum lycopersicum cultivar Micro-Tom. In Biology Methods and Protocols (Vol. 9, Number 1). Oxford University Press. https://doi.org/10.1093/biomethods/bpae008
Král, D., Šenkyřík, J. B., & Ondřej, V. (2025). Early protoplast culture and partial regeneration in Cannabis sativa: gene expression dynamics of proliferation and stress response. Frontiers in Plant Science, 16. https://doi.org/10.3389/fpls.2025.1609413
Mathur, J., Koncz, C., & Szabados, L. (1995). Plant Cell Reports A simple method for isolation, fiquid culture, transformation and regeneration of Arabidopsis thaliana protoplasts. In Plant Cell Reports (Vol. 14).
Mehbub, H., Akter, A., Akter, M. A., Mandal, M. S. H., Hoque, M. A., Tuleja, M., & Mehraj, H. (2022). Tissue Culture in Ornamentals: Cultivation Factors, Propagation Techniques, and Its Application. In Plants (Vol. 11, Number 23). MDPI. https://doi.org/10.3390/plants11233208
Muguerza, M. B., Gondo, T., Ishigaki, G., Shimamoto, Y., Umami, N., Nitthaisong, P., Rahman, M. M., & Akashi, R. (2022). Tissue Culture and Somatic Embryogenesis in Warm-Season Grasses—Current Status and Its Applications: A Review. Plants, 11(9). https://doi.org/10.3390/plants11091263
Mukundan, N. S., Satyamoorthy, K., & Babu, V. S. (2025). Advancing plant protoplasts: innovative techniques and future prospects. In Plant Biotechnology Reports (Vol. 19, Number 6, pp. 659–678). Springer. https://doi.org/10.1007/s11816-025-00957-1
Ovcharenko, O. O., Rudas, V. A., & Kuchuk, M. V. (2023). Protoplast Fusion for Cellular Engineering of the Brassicaceae. Cytology and Genetics, 57(5), 432–450. https://doi.org/10.3103/S0095452723050043
POLAT, Ş. (2025). Protoplast Fusion as a Breeding Tool for Berries: A Review of Prospects and Current Limitations. Agricultural and Food Technologies, 3(1), 131–141. https://doi.org/10.65888/icraft.3.1.14
Ranaware, A. S., Kunchge, N. S., Lele, S. S., & Ochatt, S. J. (2023). Protoplast Technology and Somatic Hybridisation in the Family Apiaceae. In Plants (Vol. 12, Number 5). MDPI. https://doi.org/10.3390/plants12051060
Reed, K. M., & Bargmann, B. O. R. (2021). Protoplast Regeneration and Its Use in New Plant Breeding Technologies. In Frontiers in Genome Editing (Vol. 3). Frontiers Media S.A. https://doi.org/10.3389/fgeed.2021.734951
Reyna-Llorens, I., Ferro-Costa, M., & Burgess, S. J. (2023). Plant protoplasts in the age of synthetic biology. In Journal of Experimental Botany (Vol. 74, Number 13, pp. 3821–3832). Oxford University Press. https://doi.org/10.1093/jxb/erad172
Shen, J., Fu, J., Ma, J., Wang, X., Gao, C., Zhuang, C. X., Wan, J., & Jiang, L. (2014). Isolation, culture, and transienttransformation of plant protoplasts. Current Protocols in Cell Biology, (SUPPL.63). https://doi.org/10.1002/0471143030.cb0208s63
Stelmach-Wityk, K., Szymonik, K., Grzebelus, E., & Kiełkowska, A. (2024). Development of an optimized protocol for protoplast-to-plant regeneration of selected varieties of Brassica oleracea L. BMC Plant Biology, 24(1). https://doi.org/10.1186/s12870-024-06005-4
Wiszniewska, A., & Pindel, A. (2010). Protoplast culture utilization in studies on legume crops. In Acta Agriculturae Scandinavica Section B: Soil and Plant Science (Vol. 60, Number 5, pp. 389–399). https://doi.org/10.1080/09064710903179545
Yang, S. H., Kim, S. W., Lee, S., & Koo, Y. (2024). Optimized protocols for protoplast isolation, transfection, and regeneration in the Solanum genus for the CRISPR/Cas-mediated transgene-free genome editing. In Applied Biological Chemistry (Vol. 67, Number 1). Springer Science and Business Media B.V. https://doi.org/10.1186/s13765-024-00870-1






