1. Introduction: The Strategic Role of Meristem Culture in Global Food Security
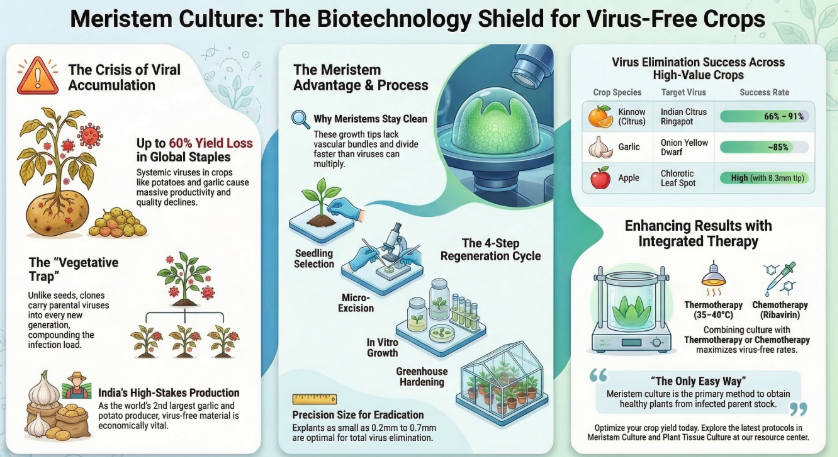
In the 21st century, global agriculture faces a critical convergence of pressures: escalating world food requirements, a shrinking arable land base, and the increasing frequency of biotic stresses exacerbated by climate change. Among these, plant viruses represent a formidable threat to productivity due to the total lack of direct chemical controls. For vegetatively propagated crops—including potato, garlic, banana, and sugarcane—viruses are particularly destructive because they accumulate within the propagating material (cloves, tubers, or suckers) and are passed from generation to generation. This leads to a progressive “viral load” that degenerates the variety’s yield and quality over time.

Meristem Culture has emerged as the premier biotechnological intervention for producing In Vitro Virus-Free Plants. By leveraging the unique physiological properties of the shoot apex, specialists can “clean” infected cultivars, ensuring that the foundational material for commercial agriculture maintains its highest health status. This technique is more than a propagation method; it is a strategic requirement for preserving the genetic integrity and commercial viability of the world’s most essential food and ornamental crops.

| Feature | Traditional Propagation (Seed/Cutting) | High-Throughput Meristem Culture |
|---|---|---|
| Genetic Stability | Low (seeds) / High (cuttings) | Maximum (direct regeneration) |
| Multiplication Rate | Limited by mother plant biomass | Exponential (up to 1700+ per explant) |
| Disease Status | Susceptible to systemic pathogens | Pathogen-free (virus elimination) |
| Recalcitrance | High in aging woody tissue | Overcome by hormone optimization |
| Volume Efficiency | High biomass requirement | Rapid scaling from minimal material |
1.1 The Power of the Tiny: Defining the Meristematic Dome
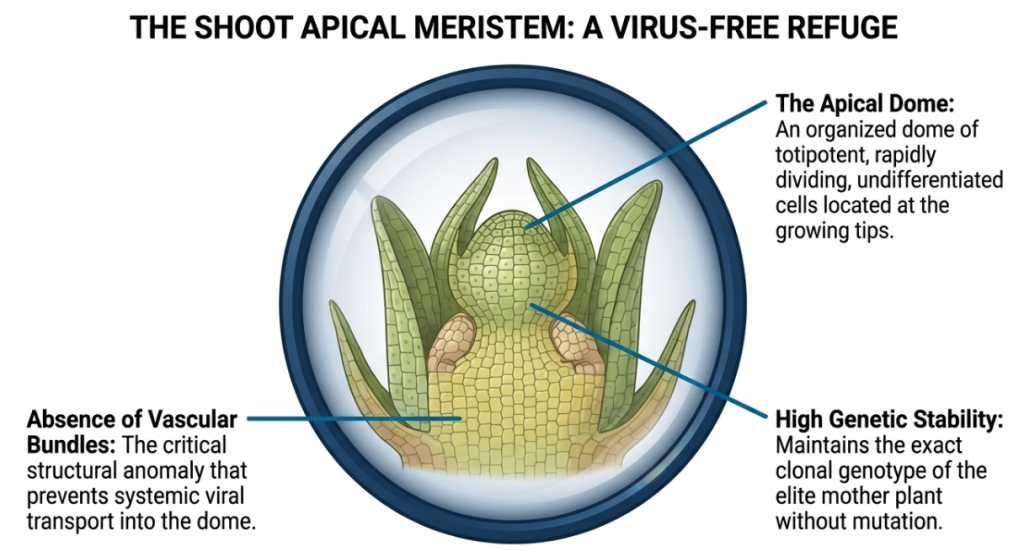
The shoot apical meristem is a tiny, dome-shaped region of totipotent cells located at the tip of a plant’s shoot. Based on the source context, totipotency is the extraordinary biological ability of these cells to divide and differentiate into any type of specialized plant cell, making the meristem the “center of activity” for all developmental programs.
In the laboratory, we distinguish between two primary types of starting materials, or “explants”:
- Meristem Culture: Involves the isolation of the true meristematic dome alone, typically measuring less than 0.1 mm. This is the gold standard for virus elimination.
- Meristem Tip Culture: Comprises the dome plus two to three rudimentary leaf primordia (the small ridges surrounding the dome). In commercial practice, meristem tips (0.1 to 0.5 mm) are preferred as the primordia significantly increase the explant’s survival rate.
The Anatomy of Potential
| Feature | Meristematic Dome / Tip | Shoot Tip Explant |
|---|---|---|
| Typical Size | 0.1 mm to 0.5 mm (up to 0.7 mm in Apple) | 0.5 mm to 5.0 mm |
| Primary Use | Virus Elimination & Germplasm Storage | Rapid Mass Propagation |
| Establishment | High Complexity (difficult to survive) | Lower Complexity (easier to establish) |
2. Biological Fundamentals: Why the Meristem is the Key to Pathogen Eradication
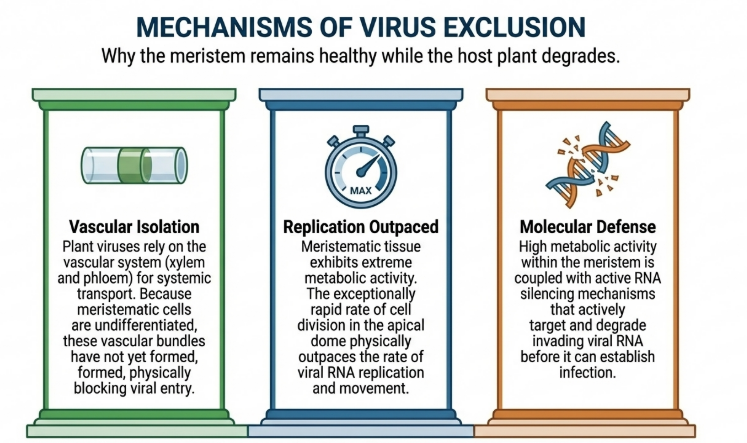
The efficacy of meristem culture is predicated on the unique biological characteristics of the shoot apex. While a virus may systemically infect a host, the concentration of viral particles decreases dramatically toward the younger, undifferentiated growth. In the terminal buds, the viral presence is often described as “feeble,” rendering it unable to maintain pace with the plant’s growth.
Several properties make meristematic tissue uniquely suited for pathogen eradication:
- Rapid Cell Division: Meristematic cells divide at rates that often exceed viral replication and systemic movement.
- Vascular Isolation: The absence of fully developed vascular tissue (xylem and phloem) in the extreme tip restricts the transport of virions into the meristematic dome.
- Totipotentiality: As first hypothesized by Haberlandt in 1902, every plant cell contains the “total potential” or genetic blueprint of the entire organism. Meristematic cells are in an ideal state of division and differentiation to regenerate into a complete, functional plant.
In a lab environment, we distinguish between “Meristem Culture” and Meristem Tip Culture (MTC). A true apical meristem is only 50 to 80 microns in diameter. However, because such microscopic explants suffer from extremely low survival rates, practitioners typically utilize a “Meristem Tip”—approximately 250 microns in size. In crops like the dahlia, this tip includes the terminal dome, the most recently formed leaf primordia, and a small amount of sub-jacent tissue. This size—roughly that of a printed period—is a calculated strategic compromise between maximizing virus elimination and ensuring the survival of the regenerated plantlet.
2.1 The Size-Regeneration Paradox: Analytical Evaluation of Explant Geometries
The inverse relationship between explant size and phytosanitary purity governs the success of viral elimination.
- 0.2–0.3 mm Range: This geometry provides the highest probability of total virus exclusion. However, in Malus, it carries a 48.91% desiccation risk and a high frequency of callusing rather than shoot proliferation.
- 0.6–0.7 mm Range: These larger shoot tips exhibit the highest establishment rates (36.36%). However, they are significantly prone to contamination, phenolic exudation (browning), and a high risk of viral retention, particularly for persistent titers like ACLSV.
- Optimal 0.3–0.6 mm Window: This range constitutes the strategic phytosanitary threshold for Malus. It balances the physiological requirements for regeneration with the exclusion of the viral complex.
Data indicates a significant seasonal influence on viability; explants harvested during summer or active vegetative growth demonstrate a 62.35% survival rate, compared to significantly lower rates for dormant buds.

Elimination Efficiency vs. Explant Size
Data-driven evaluation of indexing results (OD 405 nm) reveals critical efficiency gaps:
- Apple Chlorotic Leaf Spot Virus (ACLSV): Remains the most difficult pathogen to eliminate. At the 0.5–0.6 mm range, the efficiency is only 50% (1/2). At the 0.6–0.7 mm range, elimination efficiency drops to 0% (0/3).
- Apple Stem Grooving Virus (ASGV) and Apple Mosaic Virus (ApMV): Successfully eradicated (100% efficiency) at the 0.6 mm threshold when combined with 4-week thermotherapy.
- Apple stem pitting virus (ASPV): Demonstrates negative results (100% elimination) across the 0.3–0.6 mm range.
Shoots exhibiting OD values ≥0.201 are classified as positive and must be immediately culled to prevent the accumulation of titers in the multiplication phase.
3. The Economic Impact: Evaluating Viral Threats in Vegetatively Propagated Crops
The systemic accumulation of viruses is the “chief limiting factor” in the production of high-value clonal crops. For species that lack viable seed development—such as garlic, which is strictly propagated through cloves—MTC is the only viable path for variety improvement. As Roland A. Mildner noted regarding dahlia production, “all other problems… are insignificant in comparison” to virus diseases. Without MTC, elite clones would eventually succumb to viral load and face commercial extinction.
Table 1: Analysis of Crop Yield Losses Due to Viral Pathogens
| Crop Name | Primary Viral Diseases | Estimated Yield Loss (%) |
|---|---|---|
| Banana | Banana Bunchy Top Virus (BBTV) | Up to 100% |
| Apple | ACLSV, ASGV, ASPV, ApMV | 30% – 100% |
| Potato | Potato Viruses X, Y, S, and M | 20% – 60% |
| Garlic | OYDV, LYSV, GCLV, Allexiviruses | 25% – 54% |
| Sugarcane | SCMV, SStMV, Yellow Leaf Virus | 10% – 50% |
Because viruses like Dahlia Mosaic Virus (DMV) can be transmitted through seed more frequently than historical botanical assumptions suggested (as confirmed by 1976–1978 data), the reliance on clonal “cleaning” through MTC is an absolute economic necessity.
4. Technical Protocol: Standardizing the In Vitro Environment
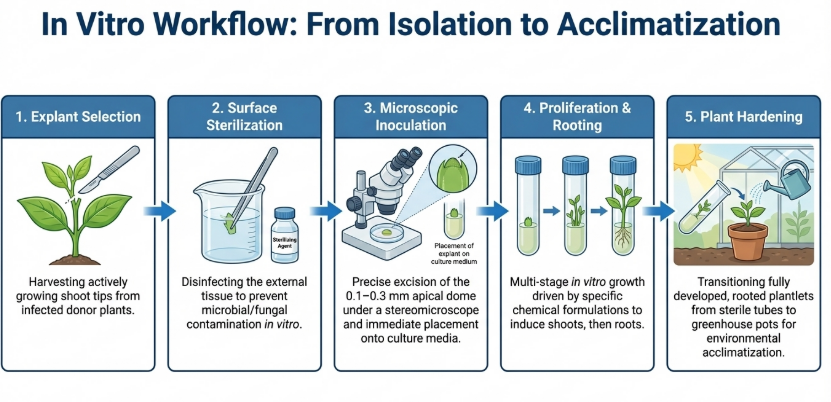
Regenerating a virus-free plant from a microscopic tip requires a meticulously controlled environment. The transition from mother plant to hardened plantlet involves precise chemical and physical parameters.
Explant Preparation and Sterilization
Initial sterilization is vital to remove fungal and bacterial contaminants.
- Surfactants: Material is first agitated in 75% Ethanol or detergents like Tween-20 to reduce surface tension.
- Sterilants: The material is soaked in 6% Calcium Hypochlorite or Sodium Hypochlorite (Clorox) for approximately 10–15 minutes, followed by three to four rinses in sterile distilled water.

The Medium: Murashige and Skoog (MS)
The MS medium is the global standard, providing macro-nutrients (concentrations >0.5 mM) and micro-nutrients (concentrations <0.5 mM). A definitive lab protocol requires the following inorganic salts:
- Nitrates: Ammonium Nitrate (NH4NO3), Potassium Nitrate (KNO3).
- Sulfates: Magnesium Sulfate (MgSO4⋅7H2O), Manganous Sulfate (MnSO4⋅H2O), Zinc Sulfate (ZnSO4⋅7H2O), Cupric Sulfate (CuSO4⋅5H2O).
- Halides: Calcium Chloride (CaCl2⋅2H2O), Potassium Iodide (KI), Cobalt Chloride (CoCl2⋅6H2O).
- PO4/BO3/MoO4: Potassium Phosphate (KH2PO4), Boric Acid (H3BO3), Sodium Molybdate (Na2MoO4⋅2H2O).
- Iron Source: Ferrous Sulfate (FeSO4⋅7H2O) and Disodium EDTA (Na2EDTA).
Organic Supplements and pH: The medium is supplemented with Sucrose (30 g/L), Myo-inositol (100 mg/L), Thiamine HCl (0.4 mg/L), and L-tyrosine (100 mg/L).
- pH Regulation: For garlic, pH must be adjusted to 5.77–5.82. For dahlia, a target of 5.0 ± 0.1 is required.
Growth Regulators: Shoot and Root Induction
The ratio of Cytokinins to Auxins determines the success of regeneration.
- Initiation/Multiplication: In garlic, a combination of TDZ (1.0 mg/L) and NAA (0.15 mg/L) has shown superior efficacy for regeneration.
- Rooting Induction: Early dahlia protocols (Morel and Martin, 1952) utilized complex grafting onto seedlings. Modern techniques eliminate this by reducing Kinetin to 1/2, 1/4, or 1/8 of its initial concentration to induce adventitious rooting in the etiolated explants.
4.1 Biochemical Optimization: Media Formulations and Hormonal Calibration
Overcoming the recalcitrance of woody germplasm requires high-precision media selection. Murashige and Skoog (MS) medium is the “Gold Standard” for Duboisia and Malus due to its high concentration of NH4NO3, which is four times higher than in WPM or QL media. However, MS can prove toxic to mature woody tissue, requiring salt reduction to half-strength during specific phases.
Hormonal Synergy and Genotypic Specificity
PGRs are synthetic hormones used to dictate plant architecture. By manipulating the ratio of cytokinins to auxins, we can control whether a cell mass becomes a forest of shoots or a network of roots.
- Benzyladenine (BA): A powerful cytokinin and the primary engine for shoot multiplication and proliferation. However, practitioners must note genotypic specificity: while BA drives growth in D. myoporoides, it can be lethal to species like Duboisia hopwoodii, which often requires Kinetin to survive and proliferate without 100% mortality.
- Indole-3-butyric acid (IBA): The essential auxin for root induction. It signals the shoot base to transition into a functional root system.
- Gibberellic Acid (GA3): Vital for shoot elongation. Without GA3, cultures often remain in a stunted “rosette” form, making individual shoots difficult to separate and root.

Hormonal calibration must account for extreme genotypic variation. While BA (1.0 mg/L) is effective for Malus and D. myoporoides, it is lethal for D. hopwoodii.
| Growth Phase | Regulator Combination | Observed Impact |
|---|---|---|
| Establishment | BA (1.0 mg/L) + IBA (0.05 mg/L) + GA3 (0.1 mg/L) | 56.62% establishment in Malus; BA causes 100% mortality in D. hopwoodii. |
| Recalcitrant Induction | Kinetin (1–10 mg/L) | Mandatory for D. hopwoodii to prevent necrotic browning and callus overgrowth. |
| Multiplication | Low BA (0.5 mg/L) + IBA (0.08 mg/L) | Achieves 2-to-4-fold multiplication; reduces apical dominance. |
| Rooting | IBA (1–10 g/L) Pulse Treatment | 5-second basal dip; essential for substrate-based induction. |
BA and IBA: Concentration Effects
| Hormone | High Concentration Effects | Low Concentration Effects |
|---|---|---|
| Benzyladenine (BA) | 1.0 mg/l: Optimal for initial establishment and survival of the tiny explant. | 0.5 mg/l: Superior for proliferation (increasing the total shoot count). |
| Indole-3-butyric acid (IBA) | Increases root quantity but risks callus formation (unorganized cell lumps). | Results in cleaner, more functional root systems with less abnormal tissue. |
The Risk of Callus: The improper balance of hormones—such as adding NAA or maintaining excessive BA levels—leads to “callus overgrowth.” This mass of unorganized cells not only hinders organ development but also increases the risk of somaclonal variation, undermining the goal of clonal propagation.
Learning Insight: The Meristem Trade-off
- Small Explants (0.2–0.3 mm): The “Gold Standard” for virus elimination, but they face a staggering 48.91% desiccation rate.
- Large Explants (0.6–0.7 mm): Boast significantly higher survival and establishment rates but are far more likely to carry systemic viruses into the new culture.
The Recipe for Success: MS vs. WPM
| Feature | Murashige and Skoog (MS) | Woody Plant Medium (WPM) / QL |
|---|---|---|
| Nutrient Density | High Salt: Features high concentrations of macro and micronutrients. | Lower Salt: Designed with reduced macronutrient content for sensitive species. |
| Nitrogen Source | Relies on high levels of NH4NO3 and KNO3. | Contains significantly lower NH4NO3 (often 1/4th the concentration of MS). |
| Primary Use | Standard for high growth, biomass, and rapid shoot proliferation. | Preferred for delicate induction in woody or recalcitrant species. |
| Toxicity Risk | Can be toxic to mature woody explants due to excessive salt levels. | Offers a gentler environment for tissues sensitive to high osmotic pressure. |
5. Advanced Eradication Strategies: Synergistic Therapies
While MTC alone is powerful, certain high-copy-number viruses require additive therapies to achieve 100% elimination.
- Thermotherapy: Exposing mother plants to temperatures between 35°C and 40°C slows viral multiplication, expanding the “virus-free zone” at the shoot tip.
- Chemotherapy: The inclusion of antiviral compounds like Ribavirin or Virazole (e.g., 35–40 mg/L) in the MS medium inhibits viral RNA synthesis.
- Cryotherapy: This advanced technique uses ultra-low temperatures to selectively destroy infected cells, leaving only the healthy meristematic cells to regenerate.
- Combined Approach: For persistent apple viruses (ASPV), efficiency reaches 95% when Ribavirin is coupled with heat therapy.

6. Environmental Challenges: Contamination and Phenolics
Navigating the in vitro environment requires managing three primary physiological threats:
- Microbial Contamination: Bacteria and fungi thrive in tissue culture media. Larger explants offer more surface area for “hitchhiking” microbes; for instance, while 0.2 mm tips may see 31% contamination, 0.7 mm tips can exceed 48%.
- Phenolic Exudation (Browning): When tissue is cut during excision, the tissue wounding triggers the release of phenols. These oxidize, turning the medium brown and creating a toxic zone that can kill the explant. We combat this using Polyvinylpyrrolidone (PVP) or Ascorbic Acid as chemical shields.
- Desiccation: The smallest meristems (0.2 mm) lack a protective cuticle. Without the ability to retain moisture, they often “dry out” and die before they can establish a connection with the medium.
Once a healthy shoot is established and multiplied, the final challenge is the transition to the terrestrial world.
The Three Rules of Successful Tissue Culture
- Match the Medium to the Species: Use high-salt MS for vigorous growth, but switch to low-salt WPM or QL (which has 1/4th the nitrogen) if the species shows signs of salt toxicity or recalcitrance.
- Balance the Hormone Ratio: Use higher BA concentrations for initial establishment, but lower them for proliferation. Be wary of BA lethality in sensitive species (e.g., D. hopwoodii), and favor Kinetin when necessary.
- Manage Explant Size: Balance the “Meristem Trade-off.” Smaller tips (0.2–0.3 mm) are required for virus-free stock but face a ~49% desiccation risk. Use larger tips for mass propagation where virus status is already verified.

7. The Final Leap: Substrates and Acclimatisation
The transition from a high-humidity sterile tube to soil is the most vulnerable point in the pipeline. Our research shows that the rooting substrate is the deciding factor for survival.
- Agar-based Rooting: Traditional, yet often results in poor root hairs and excessive callusing.
- Substrate-based Rooting (Jiffy cubes / Rockwool): This method is vastly superior. Rockwool, specifically, was found to rescue the rooting success of the recalcitrant D. hopwoodii, improving rates from a meager 10% in Jiffy cubes to 70% due to its superior oxygen diffusion and water retention.
The Acclimatisation Protocol
For the critical first 7–14 days, maintaining near-100% humidity via misting is non-negotiable.
- Seed Trays with Domes: In trials with D. myoporoides, this method achieved a 100% survival rate.
- Ziploc Bags: While portable, these only achieved 60% survival, as they offer inferior gas exchange and humidity control compared to seed trays.
8. Case Studies:

Dahlia: The Recovery of Elite Clones
During programs conducted in 1974 and 1975, specialized protocols were developed to recover diseased dahlia stock. Key successes included the variety Reve Rose (Pink Dream). Further research based on records from 1976–1978 (published in 1980) definitively established that dahlia viruses are seed-transmitted, emphasizing the role of MTC in maintaining healthy clones. Varieties like Juanita, Kidd’s Climax, and Golden Heart remain prime candidates for these programs due to their superb exhibition quality.
Garlic: Regeneration Response in Short-Day Varieties
Trials focusing on varieties G-41, G-282, and G-323 revealed significant differences based on clove dormancy:
- Sprouted Cloves: Being metabolically active, these responded faster to initiation, with an average regeneration of 91.6%.
- Fresh Cloves: Showed a slightly higher overall survival of 95% once dormancy was broken.
- Variety Survival: The variety G-282 demonstrated the highest regeneration frequency at 96.8%.

Apple (‘Oregon Spur-II’)
Success in apple culture is dictated by seasonality and bud position. Explants collected in Spring and Summer from distal positions on actively growing shoots yield the best survival (62.35%). While 0.6–0.7 mm tips are best for establishment, researchers must compromise with the 0.3–0.6 mm range to successfully eliminate ACLSV and other latent viruses.
Duboisia (Corkwood)
Duboisia is prized for pharmaceutical scopolamine production. Our research shows that tissue culture requirements are highly species-specific:
- D. myoporoides and D. leichhardtii: These species thrive on the cytokinin BA and prefer Jiffy cubes for rooting.
- D. hopwoodii: This species is more specific, requiring Kinetin for survival (as BA can cause mortality) and Rockwool substrates for successful rooting due to its need for higher oxygen and water diffusion.
9. Validation and Certification: Ensuring Pathogen-Free Status
MTC regeneration is only the first step; rigorous indexing is mandatory to confirm the absence of pathogens.
- ELISA (Serological): Rapidly detects viral proteins but may miss minute concentrations.
- RT-PCR (Molecular): The most sensitive indexing tool, capable of detecting minute amounts of viral RNA that ELISA might overlook.
- Bio-assay: Inoculating sensitive “indicator plants” with the sap of the recovered dahlia or garlic to monitor for visual symptoms.
Current constraints include the lack of official government certification for ornamental crops like dahlia. In these instances, a “blue-ribbon panel” of specialists is necessary to verify the health of stock before commercial deployment.
10. Conclusion: The Future of In Vitro Crop Improvement

Meristem culture is the cornerstone of modern plant biotechnology, offering a definitive solution for preserving heritage varieties and ensuring the commercial stability of clonal crops. By integrating MTC with advanced therapies—thermotherapy, chemotherapy, and cryotherapy—we can overcome even persistent pathogens like Allexiviruses. Continued experimentation with complex, synergistic treatments will be essential to refine regeneration frequencies and secure the future of global food and floral systems.

Image Summary



Questions/Answers
1. How do thermotherapy and meristem culture combine to eliminate viruses?
Thermotherapy and meristem culture combine to eliminate viruses by using heat to suppress viral activity while utilizing the unique anatomical properties of the plant’s growth tip to isolate healthy cells. This synergistic approach is significantly more effective than using either technique alone, particularly for viruses that are capable of invading the meristematic region.
The two techniques function together through the following mechanisms:
1. Expanding the Virus-Free Zone
While apical meristems are often naturally virus-free, some viruses can still colonize them. Thermotherapy—exposing the host plant to high temperatures, typically between 35°C and 42°C for several weeks—slows down or stops viral replication and movement. This heat treatment expands the pathogen-free area at the shoot tip, creating a larger “clean” zone from which tissue can be taken.
2. Facilitating Larger, More Viable Explants
A major challenge in meristem culture is that smaller explants have a higher chance of being virus-free but a lower chance of surviving the regeneration process. By combining the treatments, researchers can excise relatively larger meristem tips (e.g., 0.5 mm to 1.0 mm) that remain virus-free due to the heat treatment. These larger explants have significantly higher survival and regeneration rates than the tiny domes (often <0.2 mm) required when meristem culture is used alone.
3. Synergistic Biological Defense
The combination leverages both physical isolation and physiological suppression:
- Vascular Disconnection: Viruses primarily move through the plant via developed vascular tissues (phloem and xylem). Meristematic domes lack these connections, acting as a physical barrier to viral entry.
- Rapid Cell Division: Meristematic cells divide so rapidly that they often outpace viral multiplication, especially when the virus is further slowed by heat.
- RNA Silencing: High temperatures can activate and enhance the plant’s own defense system through virus-induced gene silencing (VIGS), which targets and degrades viral RNA in the shoot tips.
4. Application and Limitations
This combined approach has successfully eliminated devastating viruses in numerous crops, including sugarcane, garlic, potato, apple, banana, and yam. However, the success of the treatment is highly dependent on the plant genotype and the virus species. For example, some pathogens like Allexiviruses in garlic or certain viroids in citrus are highly heat-tolerant, making them difficult to eradicate even with combined thermotherapy and meristem culture.
2. Explain the role of hormones in establishing virus-free apple cultures.
In establishing virus-free apple cultures, hormones—specifically plant growth regulators (PGRs)—play a fundamental role in the regeneration of excised meristematic tissues into healthy, whole plantlets. Meristem culture is preferred because the apical dome often lacks a vascular connection to the rest of the plant, which prevents systemic viruses from easily colonizing that region.
The role of hormones in this process can be categorized into three main stages:
1. Culture Initiation and Establishment
Because excised meristems are extremely small (often <1 mm), they frequently lack the necessary internal resources to grow independently.
- Endogenous Hormones: The inclusion of one or two leaf primordia with the meristem tip is often essential, as these young leaves produce the endogenous auxins and cytokinins required for early development.
- Establishment Media: For the apple cultivar ‘Oregon Spur-II’, a combination of BA (1.0 mg/l), IBA (0.05 mg/l), and GA3 (0.1 mg/l) was found to be effective for the initial establishment of explants.
- Avoiding Callusing: Scientists must balance hormone concentrations carefully; for example, combining BA with NAA in some apple cultures failed to promote shoot growth and instead led to undesirable callus formation.
2. Shoot Proliferation and Multiplication
Once established, the goal shifts to maximizing the number of virus-free shoots through axillary branching.
- Breaking Apical Dominance: Cytokinins, most commonly Benzyladenine (BA), are applied to overcome apical dominance, stimulating the branching of lateral buds.
- Genotype Specificity: Apple genotypes respond differently to various PGR combinations. While BA is the most effective cytokinin for many woody species, researchers noted that for some apple rootstocks, the number of shoots per explant actually increased when BA and GA3 concentrations were decreased in the multiplication medium.
- Counteracting Cytokinins: Auxins may be added to multiplication media to nullify the suppressive effects that high cytokinin levels can have on shoot elongation.
3. Root Induction
The final stage requires a distinct shift in hormonal balance to facilitate the transition to a soil-based environment.
- Auxin Requirement: For adventitious root formation, shoots are transferred to a rooting medium typically supplemented with auxins such as NAA, IBA, or IAA.
- Pulse Treatments: In “hard-to-root” apple rootstocks, a specific strategy is used where shoots are exposed to a high-auxin “root initiation medium” for 4 to 8 days and then transferred to an auxin-free medium to prevent callus growth and allow roots to elongate.
- Activated Charcoal: To ensure successful rooting, activated charcoal is sometimes added to the medium to absorb residual cytokinins that might otherwise inhibit root development.
Direct Link to Virus Elimination
Beyond regeneration, hormones may be involved in the natural resistance of the meristem to viral invasion. It is hypothesized that the high concentration of auxins naturally present in the apical dome may be one of the biological factors that inactivates or prevents the multiplication of viruses in that specific region. Combined with other treatments like thermotherapy or chemotherapy (such as the addition of ribavirin to the medium), these hormonal protocols ensure that a high percentage of regenerated apple shoots are entirely free from pathogens like Apple chlorotic leaf spot virus (ACLSV) and Apple stem pitting virus (ASPV).
3. What defines the optimal meristem size for successful virus elimination?
The optimal meristem size for successful virus elimination is defined as a compromise or balance between two inversely related factors: the frequency of virus elimination and the capacity of the explant to survive and regenerate into a whole plant.
1. The Inverse Relationship
The size of the excised tissue directly influences the outcome of the procedure:
- Smaller Explants: Because viruses are unevenly distributed and their concentration (titer) typically decreases as you approach the apical dome, smaller explants have a significantly higher probability of being virus-free. However, they are extremely delicate, lose water rapidly during dissection, and have a much lower survival and regeneration rate.
- Larger Explants: These have a higher success rate for in vitro establishment and rapid growth because they contain more nutrient reserves and more primordial leaf structures to produce essential hormones. However, as the size increases, so does the risk that viral particles have already invaded the vascular or sub-meristematic tissues included in the cutting.
2. General Size Benchmarks
While the exact “optimal” size depends on the plant species and the specific virus being targeted, several general benchmarks exist:
- 0.2 mm to 0.5 mm: This range is most frequently cited as the standard for producing virus-free plants while maintaining acceptable survival rates.
- Below 0.2 mm: Often necessary for difficult-to-eliminate pathogens, such as the Apple mosaic virus, which can be detected in meristems larger than 0.2 mm.
- 0.5 mm to 1.0 mm: These larger explants are typically used when meristem culture is coupled with thermotherapy.
- 3 mm to 20 mm: If the objective is rapid mass propagation rather than virus elimination, these much larger “shoot tips” are preferred for their ease of handling and high survival.
3. Species-Specific Variations
Optimal sizes vary across different crops due to their unique anatomy and host-virus interactions:
- Potato: A size of 100 μm (0.1 mm) has been shown to be highly effective for eliminating Potato virus Y (PVY).
- Apple: Explants between 0.3 mm and 0.6 mm successfully eliminated four major viruses, though some at the 0.5 mm–0.6 mm range remained infected with Apple chlorotic leaf spot virus (ACLSV).
- Sugarcane: Meristems of 0.1 mm to 0.2 mm are used to eliminate viruses, while slightly larger ones (0.2 mm to 0.3 mm) can eliminate phytoplasmas.
- Dahlia: Early successful experiments used sections of approximately 0.25 mm (250 microns).
4. Impact of Coupled Therapies
The use of thermotherapy (heat treatment) significantly shifts what is considered an “optimal” size. By exposing the donor plant to high temperatures, viral replication is slowed and the pathogen-free zone at the shoot tip is expanded. This allows researchers to excise relatively larger meristems (e.g., 0.5 mm to 1.0 mm) that are both virus-free and robust enough to survive the regeneration process.
How does RNA silencing change at high temperatures in plants?
At high temperatures, the process of RNA silencing in plants is significantly enhanced, acting as a critical component of the host’s defense system against viruses and viroids. This temperature-dependent mechanism plays a vital role in inhibiting viral activity and is a key factor in why certain techniques, such as thermotherapy, are effective at eliminating pathogens.
The primary changes to RNA silencing at elevated temperatures include:
- Activation of Defense Systems: High temperatures can activate the plant’s own defense system through a process known as virus-induced gene silencing (VIGS). This activation helps prevent viral infections from establishing or spreading within the plant.
- Degradation of Viral RNA: Studies have shown that during thermotherapy (heat treatment), RNA silencing is enhanced to the point where it leads to the active degradation of viral RNA. This has been specifically observed in the shoot tips of plants like raspberry.
- Pathogen Exclusion from Meristems: Meristematic tissues are often naturally virus-free due to their high metabolic rate and rapid cell division. However, the effectiveness of RNA silencing mechanisms in these regions provides an additional “physiological and molecular defense” that further reduces virus replication.
- Synergy with Thermotherapy: High temperatures (typically 35°C to 42°C) are believed to augment pathogen-free areas in the plant not just by inhibiting virus movement, but by leveraging this enhanced RNA silencing to degrade the virus in those critical growing zones.
While it is well-established that RNA silencing-mediated defense is temperature-dependent, researchers are still investigating exactly how thermotherapy might specifically utilize this mechanism to exclude persistent pathogens like certain viroids from meristematic tissues.
4. Why do smaller meristems have higher virus elimination rates?
Smaller meristems have higher virus elimination rates because they consist primarily of the apical dome, which is frequently free of viral particles due to its unique anatomical and physiological properties.
The primary reasons for this higher efficiency include:
- Lack of Vascular Connection: Viruses typically move systemically through a plant via the phloem and xylem. Because the meristematic region lacks developed vascular tissues, viral particles are physically restricted from entering the apical dome. Excising a smaller explant (often <1 mm or even 0.1 mm) ensures that only this “clean” terminal region is taken, effectively excluding infected tissue.
- Uneven Viral Distribution: Virus titer is not uniform throughout a plant and generally decreases as the meristematic dome is approached. For instance, studies on clover showed that explants smaller than 0.6 mm resulted in 100% virus-free plants, whereas larger explants (2.5–3.0 mm) only achieved a 6% elimination rate.
- Rapid Cell Division: Meristematic cells divide and grow at an extremely fast rate. This rapid growth often outpaces viral replication and cell-to-cell movement, creating a pathogen-free zone at the very tip of the shoot.
- Biological Defense Mechanisms: Meristems possess high metabolic activity and effective RNA silencing mechanisms that prevent or degrade viral RNA, acting as a molecular barrier against infection. Furthermore, the high concentration of endogenous hormones (specifically auxins) naturally present in the apical dome may inhibit or inactivate viral particles.
While smaller explants offer the highest probability of obtaining virus-free plants, they present a significant biological compromise. Smaller meristems have a much lower regenerative capacity and survival rate in culture because they lack the internal resources and leaf primordia needed to grow independently. Scientists must therefore balance explant size to find an optimal point where virus elimination is maximized without losing the ability to regenerate a whole plant.
5. How does activated charcoal affect root development in Duboisia?
Based on the sources, the effect of activated charcoal (AC) on root development in Duboisia is species-specific, meaning it benefits some species while hindering others.
Research conducted on three Duboisia species revealed the following impacts:
- Positive Impact on D. myoporoides: The addition of activated charcoal to modified Murashige and Skoog (MS) medium (referred to as MSCh) significantly improved outcomes for this species. When combined with a pulse treatment of IBA (1–10 g L⁻¹), the use of activated charcoal resulted in a maximum rooting rate of 50% and supported good shoot quality.
- Negative Impact on D. leichhardtii and D. hopwoodii: In contrast, the inclusion of activated charcoal in the rooting medium had an adverse effect on these two species. For D. leichhardtii, rooting success was only 20% in standard MS medium and failed entirely in the charcoal-supplemented MSCh medium. For D. hopwoodii, the charcoal medium failed to induce any roots at all.
General Mechanisms of Activated Charcoal
While the specific results for Duboisia varied by species, the sources provide general theoretical reasons why activated charcoal is often used in plant tissue culture to assist rooting:
- Absorption of Inhibitors: AC can absorb toxic metabolic waste products or growth inhibitors released by the plant tissues into the medium.
- Cytokinin Removal: AC is known to absorb residual cytokinins from the multiplication stage, which can otherwise inhibit the formation and growth of roots.
- Light Shading: AC provides a dark environment (shading) for the base of the microshoot, which can be beneficial as light sometimes inhibits in vitro root growth.
Despite these general benefits, the sources emphasize that for Duboisia, the actual rooting percentage decreased for two out of the three species when charcoal was added, demonstrating the necessity of species-specific protocols. Ultimately, substrate-based rooting (using Jiffy cubes or rockwool) was found to be far more effective for Duboisia than any agar-based media, including those containing activated charcoal.
6. How does heat treatment improve survival of large explants?
Heat treatment (thermotherapy) improves the survival of explants by expanding the virus-free zone at the shoot tip, which allows researchers to excise larger, more robust pieces of tissue that have a much higher capacity for regeneration.
The mechanism by which this improves survival involves resolving the fundamental biological “compromise” of meristem culture:
1. Resolving the Size vs. Survival Conflict
Without heat treatment, viruses are often found very close to the growing tip, meaning only an extremely small meristem (typically <0.2 mm) is likely to be virus-free. However, these tiny explants lack the nutrient reserves and primordial leaf structures necessary to produce essential growth hormones (auxins and cytokinins), resulting in very low survival and regeneration rates.
2. Expanding the “Clean” Zone
Thermotherapy—subjecting the plant to temperatures typically between 35°C and 42°C—slows down or stops viral replication and inhibits the movement of virus particles into new growth. This creates a larger pathogen-negative area at the shoot apex than would naturally occur.
3. Facilitating Larger, More Viable Explants
Because the heat treatment has “cleaned” a deeper section of the shoot tip, researchers can excise relatively larger explants (often 0.5 mm to 1.0 mm, or even up to 2.5 cm in certain species like sweet potato) that remain entirely virus-free.
The survival of these larger explants is superior for several reasons:
- Nutrient Reserves: Larger explants contain more internal resources to sustain the tissue during the initial establishment phase in vitro.
- Hormonal Independence: They include more leaf primordia, which are the primary sites for the endogenous production of the hormones required for shoot and root development.
- Reduced Trauma Sensitivity: Larger pieces of tissue generally adapt more readily to the transition to culture media and recover faster from the physical trauma of excision.
By using heat to make a larger area of the plant healthy, scientists can take a larger “cutting” that is biologically better equipped to survive the transition to a test tube while still achieving the primary goal of virus elimination.
7. What is the impact of genotype on virus eradication?
The genotype of a plant is a major factor that directly influences the efficiency and success of virus eradication techniques, such as meristem culture and thermotherapy. Because no two genotypes respond identically to a given set of culture conditions, protocols must often be empirically determined and optimized for individual species and cultivars.
The impact of genotype on virus eradication manifests in several critical ways:
1. Variation in Virus Elimination Efficiency
Even within the same species, different genotypes exhibit widely varying rates of successful sanitization:
- Strawberry: In a study on Chilean strawberry accessions, meristem culture resulted in 100% virus-free plants for the ‘Contulmo’ accession, but only 78% efficiency for the ‘Purén’ accession when targeting the Strawberry mild yellow edge virus.
- Yam: Research on four yam varieties showed that while D. rotundata varieties reached 100% elimination of the Yam mosaic virus through thermotherapy, the D. alata variety showed lower efficiency. This suggests that certain species or varieties may possess more virus resistance genes or developed systemic immunity.
- Garlic: The garlic variety ‘Bhima Purple’ failed to be cleared of Allexiviruses using thermotherapy, whereas a previous study using a different genotype (G-1) was successful.
2. Tolerance to Therapeutic Stress
The effectiveness of combined treatments like thermotherapy depends on the genotype’s ability to survive harsh conditions:
- Heat Tolerance: In potatoes, different cultivars showed significant differences in their tolerance to heat treatment (37°C to 40°C). Cultivars that were more heat-tolerant also produced the largest number of virus-free plants.
- Phytotoxicity: Some genotypes are more sensitive to the phytotoxic effects of chemotherapy agents like ribavirin or high temperatures, which can lead to desiccation, browning, or death of the explant, thereby reducing the total number of recovered virus-free plantlets.
3. Morphogenetic Response and Regeneration
Genotype determines the survival and regeneration capacity of the excised meristem:
- Survival Rates: A study of 11 commercial peach genotypes showed significant survival differences on a common medium, with only three cultivars achieving a survival rate higher than 50%.
- Endogenous Hormones: Genotypic differences in the levels of endogenous phytohormones influence how a plant responds to external growth regulators. For example, in Duboisia, two species preferred the hormone BA for multiplication, while the same hormone caused mortality in a third species, which instead required kinetin.
4. Interaction with Initial Viral Titer
The success of eradication is often linked to the initial viral load (titer) within the specific genotype. If a particular variety naturally accumulates a very high copy number of a virus, standard eradication protocols may fail to achieve complete clearance, as seen with Allexiviruses in certain garlic genotypes.
8. Does RNA silencing work against all plant viruses?
RNA silencing is described in the sources as a broad-spectrum antiviral defense system that plays a critical role in protecting plants against many viruses and viroids. While it is a primary physiological and molecular defense mechanism, its success is often temperature-dependent and its effectiveness against specific pathogens can vary based on the host-virus relationship.
Key aspects regarding the reach of RNA silencing include:
- Broad-Spectrum Nature: Research indicates that components of the silencing pathway, such as RDR6, have a broad-spectrum antiviral defense role. This mechanism is a key reason why many viruses are unable to invade the meristematic region.
- Action Against Diverse Pathogens: RNA silencing has been shown to work against both RNA viruses, such as Potato virus X, and viroids, such as the Potato spindle tuber viroid (PSTVd).
- Temperature Sensitivity: The defense mediated by RNA silencing is significantly enhanced at high temperatures. During thermotherapy, this enhanced state can lead to the active degradation of viral RNA in growing tips, which has been specifically observed in raspberry.
- Meristematic Defense: Meristem tissues possess highly effective RNA silencing mechanisms that act as a “physiological and molecular defense,” further reducing virus replication in these critical zones.
While the sources characterize RNA silencing as a fundamental defense against a wide range of viral threats, they do not explicitly state that it is universally effective against all 2,000+ known species of plant viruses. Its success in eliminating a virus often depends on leveraging high temperatures to activate or augment these natural silencing processes.
9. How does the size of a meristem affect virus elimination?
The size of the excised meristem is the primary factor influencing the success of producing healthy, virus-free plants. There is a fundamental inverse relationship between the size of the explant and the efficiency of virus elimination: smaller explants have a significantly higher rate of virus removal but a much lower capacity for survival and regeneration.
1. Why Smaller Meristems Are More Effective
The effectiveness of using small meristematic sections (typically 0.1 mm to 0.5 mm) for virus elimination is based on the biological and anatomical properties of the shoot tip:
- Uneven Viral Distribution: Virus titer is not uniform throughout the plant and typically decreases (or disappears entirely) as the apical dome is approached.
- Lack of Vascular Connection: Viruses primarily move systemically through the plant’s phloem and xylem. Because vascular tissues are not yet formed in the terminal region of the shoot meristem, viral particles are physically restricted from entering the apical dome.
- Rapid Cell Division: Meristematic cells divide so rapidly that they often outpace viral multiplication and cell-to-cell movement, creating a pathogen-free zone at the very tip.
- Physiological Defenses: The meristem possesses high metabolic activity and effective RNA silencing mechanisms that prevent or degrade viral RNA. Additionally, high natural concentrations of auxins in the dome may inhibit viral replication.
2. The “Biological Compromise”
While the smallest possible explant offers the highest chance of obtaining a virus-free plant, scientists must balance this against the need for the tissue to survive:
- Small Explants (<0.2 mm): These are often required for difficult pathogens, such as the Apple mosaic virus, which can be detected in meristems larger than 0.2 mm. However, these tiny explants lack the nutrient reserves and primordial leaf structures (which produce essential growth hormones) needed for regeneration, leading to high mortality rates.
- Larger Explants (>0.5 mm): These have a much higher chance of establishing in culture and growing into whole plants. However, the risk of viral infection increases as the size of the tissue increases, as it is more likely to include infected vascular or sub-meristematic cells.
3. Optimal Sizes by Crop
Optimal meristem size is often species-specific and depends on the target pathogen:
- Potato: A size of 100 μm (0.1 mm) is highly effective for eliminating Potato virus Y (PVY).
- Sugarcane: Meristems of 0.1 mm to 0.2 mm are used to eliminate viruses, whereas slightly larger ones (0.2 mm to 0.3 mm) are effective against phytoplasmas.
- Apple: Explants between 0.3 mm and 0.6 mm can eliminate multiple viruses, but those at the larger end (0.5–0.6 mm) sometimes remain infected with Apple chlorotic leaf spot virus (ACLSV).
- Banana: Tips between 0.2 mm and 0.5 mm most frequently yield virus-free plants.
4. Impact of Combined Therapies
The use of thermotherapy (heat treatment) significantly shifts the definition of an “optimal” size. High temperatures suppress viral replication and movement, effectively expanding the virus-free zone at the shoot tip. This allows researchers to excise relatively larger, more robust meristems (e.g., 0.5 mm to 1.0 mm) that are both healthy and better equipped to survive the transition to in vitro culture.
10. How does rooting success differ between agar and substrate-based methods?
Rooting success is generally significantly higher when using substrate-based methods compared to traditional agar-based methods, particularly for recalcitrant or woody plant species.
1. Rooting Rates and Efficiency
Data from the sources, specifically concerning the genus Duboisia, demonstrate a stark contrast in success rates between the two methods:
- Duboisia myoporoides: Achieved 100% rooting in Jiffy cubes (substrate) compared to a maximum of 50% in modified agar medium.
- Duboisia leichhardtii: Showed a dramatic increase from 20% success on agar to 100% success in Jiffy cubes.
- Duboisia hopwoodii: Success rose from 50% (with poor quality) on agar to 70% when using rockwool as a substrate.
In addition to higher percentages, substrate-based rooting often accelerates the propagation process, which is critical for commercial production.
2. Physiological Factors for Success
The superior performance of substrates like Jiffy cubes (coir-based) and rockwool is attributed to several physiological advantages:
- Improved Aeration and Oxygen Diffusion: Agar can lead to poor aeration and a slow rate of diffusion for toxic metabolites released by growing tissues. Rockwool, by contrast, has better oxygen diffusion, which is essential for root formation in certain species.
- Water Retention: Substrates like rockwool and Jiffy cubes can retain high levels of water while still ensuring proper aeration, providing a more balanced environment for the delicate microshoots.
- Nutrient Availability: Jiffy cubes contain balanced levels of mineral nutrients that support plantlet vigor.
- Reduced Inhibition: Agar is not a completely inert material and may contain growth inhibitors that result in poor root growth in sensitive species.
3. Impact on Plant Quality and Survival
Plants rooted in substrates tend to exhibit better overall shoot quality and higher vigor than those rooted on agar. This increased vigor is directly linked to higher acclimatization success when moving plants from in vitro to greenhouse environments. For example, the success of Duboisia acclimatization was attributed to the vigorous root systems and abundant root hairs that developed specifically in substrate-based environments.
4. Methodological Adaptations for Agar
To counteract the limitations of agar, researchers sometimes use specific treatments to mimic the benefits of substrates:
- Activated Charcoal (AC): Supplementing agar with AC can improve results by absorbing toxic substances or residual cytokinins that inhibit rooting.
- Pulse Treatments: Dipping shoots in a high-concentration auxin solution for a few seconds before placing them on agar (rather than incorporating the hormone into the agar itself) can sometimes reduce callus formation and improve root induction.
11. How do researchers determine the best hormone levels for meristems?
Researchers determine the best hormone levels for meristem culture through empirical study and optimization, as there is no universal medium that works for all plants. Because different species and even specific cultivars within a species respond differently to various combinations, scientists must systematically test varying concentrations of plant growth regulators (PGRs) across different stages of development.
The process of determining these optimal levels involves several critical factors:
1. Stage-Specific Experimentation
Researchers optimize hormone levels for three distinct phases of the meristem culture pipeline:
- Culture Initiation: In some species, initiation can occur on hormone-free media, suggesting sufficient endogenous (internal) hormones are already present. For others, low levels of cytokinins are added to support early growth. In apple, a combination of BA (1.0 mg/l), IBA (0.05 mg/l), and GA3 (0.1 mg/l) was found most effective for establishment.
- Shoot Multiplication/Proliferation: The goal here is to maximize the number of shoots through axillary branching. Researchers typically apply high concentrations of cytokinins (such as BA or Kinetin) to break apical dominance.
- Root Induction: Once shoots are developed, researchers transfer them to a medium supplemented with auxins (like NAA, IBA, or IAA) to promote adventitious root formation.
2. Balancing the Auxin/Cytokinin Ratio
Determining the best level often depends on the critical balance between different classes of hormones. For example, in yam, a higher proportion of cytokinins relative to auxins promotes more shoots but can reduce shoot length, whereas a lower ratio encourages shoot elongation. Similarly, in dahlia, researchers found that rooting success depended on a specific balance between Kinetin and IAA, though the exact “best” ratio had to be established through multiple trials varying Kinetin to 1/2, 1/4, or even 1/8 of its initial level.
3. Accounting for Genotype and Endogenous Levels
Researchers must consider the plant’s internal hormone levels, which vary by genotype, age, and tissue type. Tissues taken from young, actively growing shoots often have higher natural hormone levels and are easier to establish than those from dormant buds. In Duboisia, researchers discovered that two species preferred the cytokinin BA, while the same hormone caused mortality in a third species, which instead required Kinetin.
4. Evaluation of Quantitative and Qualitative Metrics
To identify the “best” levels, researchers use standardized evaluation criteria to compare the results of different hormone treatments:
- Survival and Regeneration Rates: Measuring the percentage of explants that survive the initial transition to culture.
- Shoot and Root Metrics: Counting the average number of shoots or roots produced per explant and measuring their lengths.
- Quality Scoring Systems: Using standardized scales (e.g., 1 to 5) to rate the morphological and physiological health of the plantlets, looking for signs of vigor or undesirable traits like callusing or leaf chlorosis.
- Avoiding Phytotoxicity: Testing different concentrations to ensure the hormones do not cause damage, such as desiccation or browning of the tiny, delicate meristem.
5. Interaction with Environmental Factors
Optimization also considers how hormone levels interact with the culture environment. Researchers have observed that requirements for hormones can shift depending on light intensity, temperature, and even the salt concentration of the basal medium. For instance, in strawberry, the optimal light intensity for shoot proliferation varies by genotype and must be standardized alongside the hormone regime.
12. Can RNA silencing eliminate viruses without using heat treatment?
Yes, RNA silencing functions as a natural defense mechanism in plant meristems to reduce virus replication and prevent accumulation, even without external heat treatment. However, its effectiveness for complete virus elimination is highly temperature-dependent, and it is most commonly leveraged in conjunction with heat treatment to be successful.
The relationship between RNA silencing and heat treatment involves several key factors:
- Natural Meristematic Defense: RNA silencing is part of the “physiological and molecular defense” that helps keep meristematic tissues naturally free of viruses. Along with rapid cell division and a lack of vascular connection, RNA silencing mechanisms in these regions act as a barrier to viral invasion. For example, the exclusion of the potato spindle tuber viroid (PSTVd) from the meristem is attributed to this mechanism.
- Enhancement by High Temperatures: While the process occurs naturally, research indicates that viral RNA silencing is significantly enhanced at high temperatures. During thermotherapy (heat treatment), this enhanced state can lead to the active degradation of viral RNA in growing tips, a process specifically observed in raspberry.
- Activation of Host Defenses: High temperatures can activate the plant’s own defense system through virus-induced gene silencing (VIGS). This activation helps prevent viral infections from establishing or spreading into the clean zones of the shoot tip.
- Temperature Sensitivity: Because the defense mediated by RNA silencing is temperature-dependent, standard ambient conditions may not always provide a strong enough response to eliminate persistent or heat-tolerant pathogens.
In summary, while RNA silencing is always “working” as a biological barrier in the meristem, it is typically used as a tool for intentional virus elimination by applying heat to augment and trigger a more robust silencing response.
References
Benke, A. P., Krishna, R., Khandagale, K., Gawande, S., Shelke, P., Dukare, S., Dhumal, S., Singh, M., & Mahajan, V. (2023). Efficient Elimination of Viruses from Garlic Using a Combination of Shoot Meristem Culture, Thermotherapy, and Chemical Treatment. Pathogens, 12(1). https://doi.org/10.3390/pathogens12010129
Demirel, E. (2025). Virus-Free Plant Production Using Meristem Culture in the Pepper (Capsicum annuum L.) Plant: Basic Principles, Applications and Current Approaches. In Plant Protection: Innovation and Sustainable Strategies (p. 56).
Gogile, A., Markos, T., Kebede, M., Kidanemariam, D., & Abraham, A. (2024). Elimination of yam mosaic virus from yam using an optimized combination of meristem culture and thermotherapy. Australasian Plant Pathology, 53(2), 185–197. https://doi.org/10.1007/s13313-024-00965-9
Grout, B. W. W. (n.d.). Meristem-Tip Culture for Propagation and Virus Elimination.
Karasawa, M. M. G., Botega, V. T., Pinto, J. E. B. P., Lédo, F. J. S., Pereira, A. Vander, & Pinto, J. C. (2023). Effects of thermotherapy and meristem culture on forage production and nutrition value in elephant grass cultivars. Agronomy Science and Biotechnology, 9, 1–24. https://doi.org/10.33158/asb.r176.v9.2023
Krishna, R., Ansari, W. A., Khandagale, K., Benke, A. P., Soumia, P. S., Manjunathagowda, D. C., Gawande, S. J., Ade, A. B., Mokat, D. N., & Singh, M. (2022a). Meristem culture: A potential technique for in vitro virus-free plants production in vegetatively propagated crops. In Advances in Plant Tissue Culture (pp. 329–344).
Krishna, R., Ansari, W. A., Khandagale, K., Benke, A. P., Soumia, P. S., Manjunathagowda, D. C., Gawande, S. J., Ade, A. B., Mokat, D. N., & Singh, M. (2022b). Meristem culture: A potential technique for in vitro virus-free plants production in vegetatively propagated crops. In Advances in Plant Tissue Culture: Current Developments and Future Trends (pp. 325–343). Elsevier. https://doi.org/10.1016/B978-0-323-90795-8.00017-5
Lassois, L., Lepoivre, P., Swennen, R., van den Houwe, I., & Panis, B. (2012). Thermotherapy, Chemotherapy, and Meristem Culture in Banana (pp. 419–433). https://doi.org/10.1007/978-1-62703-074-8_32
Leticia, T., Silvina, S. M., Ángel, S. M., & Alberto, B. P. (2024). Meristem culture is a quite efficient method for the eradication of Potato Virus Y (PVY) from calibrachoa ‘Pampa Salmon INTA.’ Plant Cell, Tissue and Organ Culture, 159(1). https://doi.org/10.1007/s11240-024-02887-2
Manjunathagowda, D. C., Benke, A. P., Mahajan, V., & Singh, M. (2022). Standardization of Meristem Tip Culture in Short Day Garlic Varieties. International Journal of Agriculture and Biology, 27(6), 399–404. https://doi.org/10.17957/IJAB/15.1940
Nehra, N. S., & Kartha, K. K. (1994). Meristem and Shoot Tip Culture: Requirements and Applications*. In Plant Cell and Tissue Culture (pp. 37–70).
Quiroz, K. A., Berríos, M., Carrasco, B., Retamales, J. B., Caligari, P. D. S., & García-Gonzáles, R. (2017). Meristem culture and subsequent micropropagation of Chilean strawberry (Fragaria chiloensis (L.) Duch.). Biological Research, 50(1). https://doi.org/10.1186/s40659-017-0125-8
Sarropoulou, V., Grigoriadou, K., Maliogka, V. I., Sassalou, C. L., & Ziogas, V. (2024). The Elimination of Viroids through In Vitro Thermotherapy and a Meristem Tip Culture from a New Limonime Hybrid (Citrus x limon var. limon (L.) Burm. f. x Citrus latifolia var. latifolia). BioTech, 13(3). https://doi.org/10.3390/biotech13030037
Vivek, M., & Modgil, M. (2018). Elimination of viruses through thermotherapy and meristem culture in apple cultivar ‘Oregon Spur-II.’ VirusDisease, 29(1), 75–82. https://doi.org/10.1007/s13337-018-0437-5
Wang, P.-J., & Charles, A. (1991). Micropropagation Through Meristem Culture. In Biotechnology in Agriculture and Forestry (Vol. 17).
Weland, G. G. (n.d.). Meristem Tip Culture. Xue, Y., Hiti-Bandaralage, J. C. A., Jambuthenne, D. T., Zhao, Z., & Mitter, N. (2023). Micropropagation of Duboisia Species via Shoot Tip Meristem. Horticulturae, 9(12). https://doi.org/10.3390/horticulturae9121313






