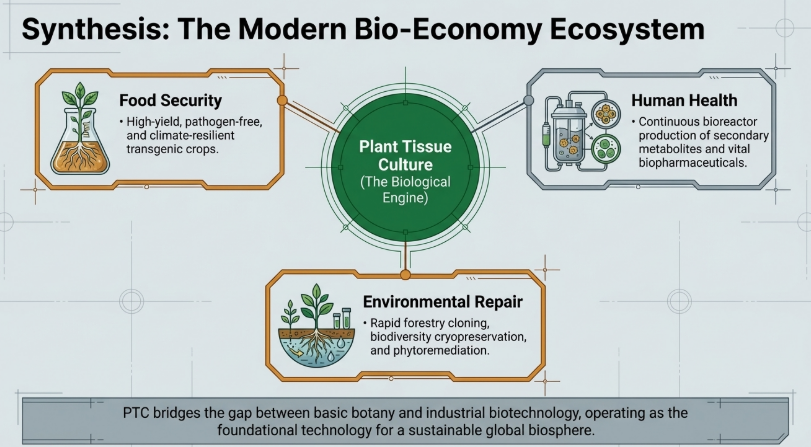
1. Executive Introduction: The Strategic Role of Plant Tissue Culture
Plant Tissue Culture (PTC) is the aseptic in-vitro culture of cells, tissues, and organs under strictly controlled nutritional and environmental conditions. As the global population approaches 10 billion, PTC has transitioned from a laboratory curiosity into a critical strategic asset for solving agricultural and environmental crises. It provides a robust framework for addressing food security and the rapid acceleration of species extinction by enabling the high-volume production of disease-free, “true-to-type” plantlets regardless of external climatic constraints.

In the current biotechnological landscape, PTC serves as the “ground truth” for in-vitro cloning, the synthesis of secondary metabolites, and the isolation of high-purity phytoconstituents. By leveraging small fragments of plant material (explants), the industry can bypass traditional breeding cycles, significantly reducing the “time-to-market” for new cultivars. This shifts agriculture from a seasonal, weather-dependent gamble into a year-round, high-efficiency production system.

The Strategic Value of Micropropagation In vitro cloning, or micropropagation, provides a professional advantage over traditional propagation (seeds or cuttings) through:
- Rapid Multiplication: Producing thousands of identical, disease-free clones from a single microscopic “explant.”
- Temporal Independence: Growth is maintained year-round, entirely independent of seasonal or climatic constraints.
- Genetic Fidelity: Ensuring that all offspring are “true-to-type” clones of a superior mother plant.

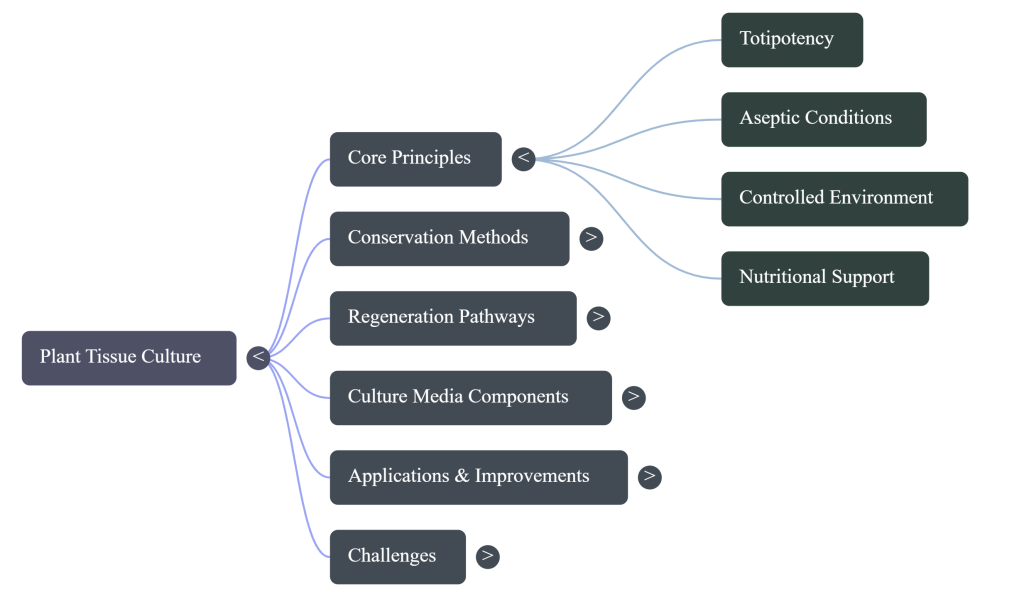
2. The Foundations of Totipotency and Historical Evolution
Totipotency is the biological mandate that allows a single plant cell to express its full genome and regenerate into a complete, functional organism. This ability to “reprogram” differentiated somatic cells back to an embryonic state is the engine of the micropropagation industry.

The technical bedrock of our roadmap remains plant totipotency. This concept, first proposed by Gottlieb Haberlandt in 1902, was grounded in the foundational cell theory of Schleiden (1838) and Schwann (1839). While it took over half a century for F.C. Steward (1958) to experimentally demonstrate this by regenerating whole plants from carrot phloem segments, modern biotechnology has moved beyond mere regeneration. We are now in the era of active cellular reprogramming, where we leverage the innate potential of a single cell to drive industrial-scale production.
Key Milestones in PTC Evolution:
- 1902 – Gottlieb Haberlandt: The “father of plant tissue culture” first proposed the concept of totipotency. While he successfully cultured isolated somatic cells, he failed to witness cell division—a limitation of the media of his era.
- 1958 – F.C. Steward: Provided the definitive experimental proof of totipotency by regenerating whole carrot plants from differentiated phloem segments.
- 1950s – The Orchid Industry: The first major commercial pivot, using PTC to move orchids from a rare luxury to a mass-market commodity.
- 1972 – Carlson et al.: Created the first somatic hybrid (Nicotiana glauca x N. langsdorffii) via protoplast fusion, proving that PTC could bypass sexual incompatibility.
Haberlandt’s hypothesis provided the intellectual capital for today’s multi-billion dollar floriculture and forestry sectors. His vision allows us to manifest the latent genetic potential of a single cell into thousands of identical copies, provided the laboratory environment is perfectly optimized.
3. The In-Vitro Environment: Culture Media and Environmental Control
The culture medium is the “life support system” for the explant, delivering the chemical signals required to trigger specific developmental pathways. To maintain an inductive environment conducive to this reprogramming, the following five environmental factors must be meticulously controlled:
- Nutrient Composition: A precise balance of macronutrients (N, P, K, Mg, S) and micronutrients.
- pH Levels: Typically adjusted between 5.5 and 5.8 to ensure optimal nutrient solubility and growth regulator activity.
- Temperature: Maintained within a steady range, generally between 22°C and 28°C.
- Light Regimes: Set between 50 and 100 μmol m⁻² s⁻¹ with a standard 16h/8h photoperiod.
- Gaseous and Physical Support: Managing O₂/CO₂ exchange and utilizing gelling agents like agar or gellan gum for structural stability.
While these physical parameters establish the stage for growth, the specific developmental trajectory of the cell is directed by exogenous chemical signals that trigger internal morphogenic pathways.

| Media Type | Characteristics | Strategic Applications |
|---|---|---|
| Murashige & Skoog (MS) | High nitrogen (ammonium/nitrate); standard baseline. | The “universal” choice for most herbaceous species and initial R&D. |
| B5 / BABI | B5: Low ammonium. BABI: Increased calcium; separates nitrates from ammonium in different salts. | BABI allows for high-precision customization of nitrogen sources to overcome genotype-specific recalcitrance. |
| WPM / DKW | Woody Plant Medium; DKW features significantly higher sulfate concentrations. | Optimized for tree species like Teak; high sulfate supports specific woody genotype metabolism. |
Hormonal Regulation and the PGR “Ground Truth” Developmental outcomes are governed by the Auxin-Cytokinin Balance:
- High Cytokinin (e.g., BA, TDZ): Strategic impact on shoot regeneration and axillary proliferation.
- High Auxin (e.g., NAA, 2,4-D, PIC): Strategic impact on root formation and embryogenic callus induction.
- Synthetic Potency Note: Practitioners must recognize that synthetic auxins like 2,4-D and PIC are orders of magnitude more potent than natural IAA. Consequently, precise mg/L measurements are the industry standard for ensuring reproducible ROI.
The “So What?” Layer: Media optimization is a primary cost-saving lever. Precise pH control (5.4–5.8) and environmental sterility prevent the catastrophic loss of germplasm, while fine-tuned nitrogen ratios in media like BABI reduce the time required for successful regeneration, directly lowering operational overhead.
Comparison of Primary Plant Growth Regulators
| Regulator Type | Primary Biological Function | Specific Examples |
|---|---|---|
| Auxins | Stimulates cell elongation; promotes adventitious root formation. | 2,4-D, NAA, IAA, IBA, Picloram (PIC) |
| Cytokinins | Promotes cell division; induces shoot organogenesis and axillary proliferation. | BA (BAP), Kinetin, Zeatin, Thidiazuron (TDZ) |
The “So What?” of Hormone Ratios The interaction between Auxins and Cytokinins serves as a developmental “switch.” By manipulating this ratio, researchers can steer tissue toward specific outcomes:
- High Auxin : Low Cytokinin = Promotion of root development (Rhizogenesis).
- Low Auxin : High Cytokinin = Promotion of shoot development (Caulogenesis).
- Balanced Ratio = The formation of Callus, an unorganized mass of parenchymatous tissue that serves as a platform for further regeneration or genetic transformation.
As these chemical gradients take effect, the plant tissue follows physical pathways toward complete regeneration.
3.1 Preparing MS Medium
The most widely utilized formulation in the world is Murashige and Skoog (MS) medium. While highly effective for a broad range of species, a Senior Biotechnologist must note that MS medium is characterized by high levels of ammonium, which can occasionally be toxic to sensitive species. In such cases, BABI medium (a modified B5 formulation) is an excellent alternative starting point for optimization.
Composition of Murashige and Skoog (MS) Medium
| Category | Key Components | Purpose |
|---|---|---|
| Macronutrients | KNO3, NH4NO3, MgSO4, CaCl2, KH2PO4 | Fundamental building blocks for biomass and cell structure. |
| Micronutrients | Iron (FeEDTA), Boron, Manganese, Zinc, Copper, Molybdenum | Essential catalysts for enzymatic and metabolic reactions. |
| Vitamins & Organics | Myo-inositol, Thiamine HCl, Nicotinic acid, Pyridoxine | Support cellular health and prevent metabolic deficiencies. |
| Gelling Agents | Agar, Gellan gum, or Agarose | Provides a stable, semi-solid matrix for tissue support. |
The Critical pH Factor: 5.8 Before sterilization, the medium must be adjusted to a pH of 5.8. While the acceptable range is technically 5.4–5.8, the 5.8 set-point is vital to ensure the solubility of mineral salts and to maintain the stability and activity of Plant Growth Regulators (PGRs). If the pH drifts, nutrients may precipitate out of the solution, becoming unavailable to the plant.
Table: Composition of 1 L of MS medium
| Major Salts | Milligram (mg) |
| KNO3 | 1900 |
| MgSO4.7H2O | 370 |
| NH4NO3 | 1650 |
| KH2PO4 | 170 |
| CaCl2.2H2O | 440 |
| Minor Salts | |
| MnSO4.4H2O | 22.3 |
| H3BO3 | 6.2 |
| ZnSO4.7H2O | 8.6 |
| KI | 0.83 |
| Na2MoO4.2H2O | 0.25 |
| CuSO4.5H2O | 0.025 |
| CoCl2.6H2O | 0.025 |
| FeSO4.7H2O | 27.8 |
| Na2EDTA.2H2O | 37.3 |
| Vitamins | |
| Myoinositol | 100 |
| Thiamine HCl | 0.1 |
| Nicotinic Acid | 0.5 |
| Pyridoxine HCl | 0.5 |
| Others | |
| Glycine | 2 mg |
| Casein hydrolysate | 1 g |
| Sucrose | 30 g |
| Agar | 10 g |
| pH | 5.7 – 5.8 |
3.2 The Laboratory Sanctuary: Maintaining Aseptic Conditions
In plant biotechnology, the greatest hurdle is microbial competition. The nutrient-rich media designed for plants is equally enticing to bacteria and fungi, which can easily outcompete delicate plant tissues. Maintaining aseptic conditions is the absolute prerequisite for success.
The three pillars of a sterile workspace are:
- Surface-Sterilized Tools and Explants: Scalpels, forceps, and the plant tissue itself must be treated with sterilants (like bleach or ethanol) to eradicate surface pathogens.
- Defined, Autoclaved Media: The growth medium must be pressure-sterilized to ensure it is completely free of spores.
- Controlled Environment: Using Laminar Airflow Hoods and sealed vessels to prevent the introduction of airborne contaminants.
The Consequence of Failure: Contamination leads to “culture death.” Microbes deplete nutrients and release toxins that cause tissue necrosis, resulting in the total loss of valuable genetic material and laboratory resources.
4. Basic Steps of Plant Tissue Culture
4.1 Step 1: Selection and Initiation (The Explant)
The “explant” is the fragment of living tissue used to start a culture. Common choices include shoot tips, leaf segments, or root tips. The goal is to select a “true-to-type” mother plant with superior agronomic or aesthetic traits.
The Sensitivity of Initiation For beginners, the initiation phase is the most delicate. The explant must undergo dedifferentiation—the process where specialized cells “reset” their developmental program. This is a period of high physiological stress as the tissue moves from an endogenous (internal) hormone environment to an exogenous (added) one. Success depends on the cells being “competent”—meaning they are biologically ready to receive and respond to the new chemical signals.
As the explant settles, it begins its transformation through rapid cell division.
4.2 Step 2: Multiplication and the Hormone Balance
In this stage, the biotechnologist directs the plant’s development using the “Hormone Seesaw” model, famously established by Skoog and Miller (1957). By manipulating the ratio of two hormone groups, we control the developmental fate of the tissue.
The Phytohormone Model
| Hormone Type | Examples | Primary Effect |
|---|---|---|
| Auxins | NAA, 2,4-D, IBA, IAA | Promotes root formation; 2,4-D is the potent inducer for Callus and SE. |
| Cytokinins | BA (Benzyladenine), Kinetin, TDZ | Induces adventitious shoot formation and cell division. |
- High Cytokinin : Low Auxin = Rapid Shoot Proliferation.
- High Auxin : Low Cytokinin = Root Development.
- Balanced Ratio = Formation of Callus (a mass of undifferentiated, unorganized cells).
Depending on these signals, the cells choose one of two distinct paths to regeneration.
4.3 Step 3: Choosing a Path—Organogenesis vs. Somatic Embryogenesis
1. Organogenesis
This pathway involves the development of specific organs (roots or shoots) either directly from the explant or from a callus.
- Application: Widely used for Orchids (Pholidota pallida).
- Vascular Connection: Crucially, organogenic shoots are physically and vascularly attached to the parent tissue, meaning they must be “induced” twice—once for shoots and once for roots.
2. Somatic Embryogenesis (SE)
Here, somatic cells are reprogrammed to form bipolar embryos, containing both a shoot and root pole simultaneously.
- Application: Essential for crops like Soybean, Sandalwood, and certain conifers.
- Vascular Connection: Unlike organogenesis, somatic embryos are independent structures with no vascular connection to the parent tissue, allowing them to be easily separated and even encapsulated into “synthetic seeds.”

Biological Choice: 3 Major Differences
- Bipolarity: SE produces a complete root/shoot unit; organogenesis produces one organ at a time.
- Vascularity: Organogenic tissues are attached; SE embryos are independent.
- Reprogramming: SE requires deeper cellular “resetting” (often induced by the auxin 2,4-D).
| Feature | Organogenesis (Direct/Indirect) | Somatic Embryogenesis (SE) |
|---|---|---|
| Mechanism | Regeneration of organs (shoots/roots) from meristems or callus tissue. | Development of differentiated embryos from somatic cells (non-sexual). |
| Commercial Scalability | High; standard for ornamentals and fruit crops. | Superior; optimal for bioreactor systems and massive synchronization. |
| Key Differentiator | Rapid multiplication of existing structures. | The only viable pathway for synthetic seed production. |
| Primary PGR Role | BA/TDZ for shoot proliferation (e.g., Black Pepper); NAA for root induction (e.g., Stevia). | Auxins like 2,4-D and PIC to induce “commitment” to the embryogenic pathway. |
Three Critical Factors Influencing SE Success:
- Genotype: The genetic predisposition of the donor plant significantly affects its regenerative capacity.
- Explant Selection: Using juvenile or undifferentiated tissues, such as the immature embryos of Chir pine (Pinus roxburghii).
- Signaling and Transcription Factors: The use of 2,4-D is often required for induction, though it must be removed to avoid suppression of subsequent embryo development. Molecular triggers such as the transcription factor WOX2 are also critical in maintaining totipotency.
4.4 Step 4: Environmental Factors and Hardening Off
Beyond chemistry, physical factors determine success.
- Temperature: Usually maintained between 22–28°C.
- Light: Intensity should be calibrated to 50–100 μmolm−2s−1 to support growth without causing tissue scorching.
The Hardening Process In vitro plantlets live in a “luxury” environment of 100% humidity and constant nutrients. To survive the real world, they must undergo Hardening. This involves the gradual reduction of humidity and a slow increase in light intensity. This process allows the plant to develop a protective cuticle and functional stomata, transitioning it from lab-dependency to soil-independence.
The “So What?” Layer: SE facilitates the creation of synthetic seeds—encapsulated embryos that can be stored and handled like natural seeds. This provides a mechanism for mass-scaling plants that do not produce viable seeds, such as certain bamboos or high-value hybrids.

5. Specialized Laboratory Methods and Their Real-World Purpose
5.1 Anther Culture
- Technical Summary: The in vitro culture of immature pollen (microspores) to produce haploid plants.
- Key Benefit: These plants are converted into doubled haploids, achieving 100% genetic homozygosity in a single generation. This dramatically accelerates the breeding cycle for crops like rice and wheat.
5.2 Synthetic Seeds
- Technical Summary: The encapsulation of somatic embryos or vegetative propagules in a biodegradable coating (often sodium alginate) enriched with nutrients.
- Key Benefit: Allows for easy handling, long-term storage, and the mass multiplication of species that do not produce reliable seeds, such as various high-value bamboos.
5.3 Protoplast Fusion
- Technical Summary: The removal of cell walls via enzymatic digestion to create “protoplasts,” which are then fused via electrofusion or chemical means.
- Key Benefit: Creation of Somatic Hybrids to transfer specific traits, such as transferring salt-stress resistance from ditch reed into rice, bypassing natural breeding barriers.
The Economic Impact: These specialized methods have a profound global footprint. India’s floriculture exports reached USD 86.62 million in FY 2023-2024, highlighted by the milestone export of Anthurium flowers from Mizoram to Singapore. Such success demonstrates the role of biotechnology in expanding high-value agricultural markets.
6. Commercial Feasibility Assessment: Industrial-Scale Plant Tissue Culture Operations
6.1 Global and Regional Market Dynamics
The global agricultural sector is undergoing a definitive strategic pivot toward biotechnology-driven propagation. This transition is a direct response to the dual pressures of global food security and an escalating demand for high-value botanical products. Traditional methods are no longer sufficient to meet the rigorous quality and volume requirements of modern supply chains. Industrial-scale Plant Tissue Culture (PTC) provides the necessary solution, offering a controlled, aseptic environment for the mass production of superior genotypes. This shift allows for year-round, weather-independent production, stabilizing horticultural and pharmaceutical markets.
The scale of this commercial opportunity is delineated by the following market projections:
- 2023 Market Valuation: USD 116 Billion
- Projected 2034 Valuation: USD 293.35 Billion
- Compound Annual Growth Rate (CAGR): 8.8%
- Strategic Core: Integration of genetic engineering, marker-assisted breeding, and industrial micropropagation.

The competitive landscape is dominated by a select group of eleven nations: the Netherlands, India, China, Japan, South Korea, Taiwan, France, Colombia, Italy, Australia, and Israel. Collectively, these leading exporters control approximately 80% of the global market. The “So What?” for stakeholders: This extreme concentration of market share creates high entry barriers for new laboratories. Success for new entrants is not merely a matter of biological output, but of achieving the established infrastructure and rigorous “disease-free” quality certifications that allow these eleven nations to dictate global pricing and standards.
This industrial capacity is fundamentally enabled by the unique biological mechanisms of plant cells.

6.2. High-Value Applications: Micropropagation and Germplasm Conservation
PTC is the intersection of commercial profit and ecological stewardship.
- Woody Species: India has established clear leadership in Tectona grandis (Teak) and Santalum album (Sandalwood). Industrial leaders such as Motherbiotech Inc. (Teak) and Vatican Shona Agrotech (Sandalwood) demonstrate how PTC captures breeding benefits in long-gestation species, providing elite quality nursery stock that traditional seeds cannot match.
- Floriculture: This sector remains a massive financial engine, with Indian floriculture exports reaching USD 86.62 million in FY 2023-2024. A critical milestone occurred on December 6, 2024, with the first-ever export of Anthurium flowers from Mizoram to Singapore, proving the viability of regional hubs in global trade.
- Medicinal Extracts: Labs are increasingly focused on secondary metabolite production for pharmaceutical interests. While species like Cannabis sativa are often “recalcitrant” under in vitro conditions, mastering these difficult protocols creates a strategic “moat,” as the ability to produce Bioactive compounds in a regulated environment is a significant competitive advantage.
- Secondary Metabolites: The Bio-Factory Plants produce secondary metabolites (antioxidants, anti-cancer agents) for defense. Hairy Root Culture, achieved through the integration of T-DNA from the Ri plasmid of Agrobacterium rhizogenes, creates genetically stable, hormone-independent root systems. These act as stable bio-factories, producing pharmaceutical compounds without depleting wild plant populations.

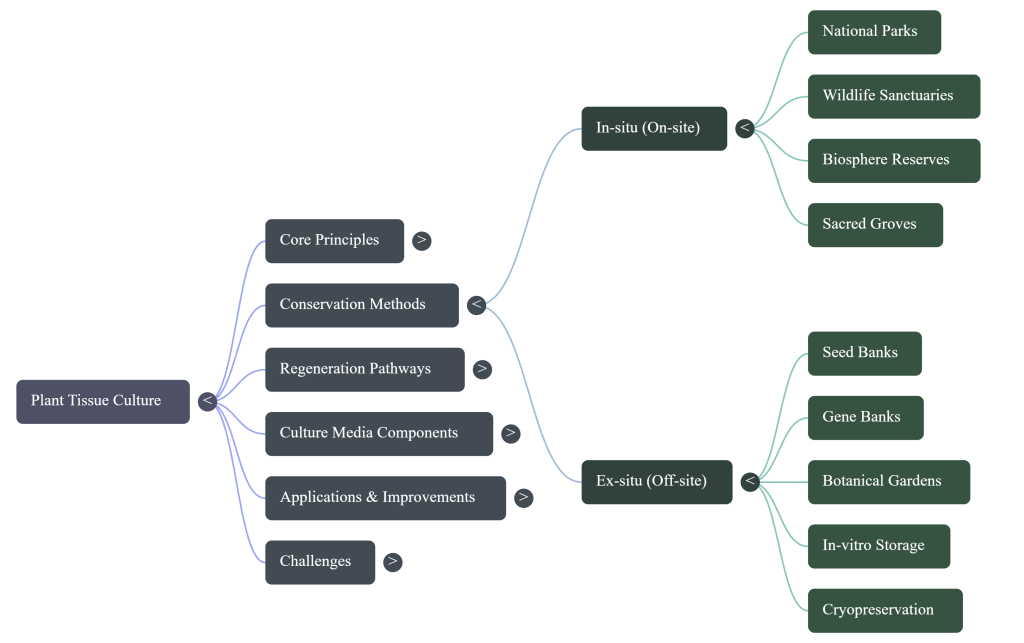
- Ex-Situ Conservation: When in-situ habitats are threatened by urbanization, ex-situ solutions like in-vitro gene banks provide the ultimate insurance policy for biodiversity.
- Cryopreservation: Utilizing liquid nitrogen (-196°C) for long-term genetic stability. The “Cryobionomics” approach ensures that genetic integrity and pharmaceutical IP are maintained over decades of storage.

The “So What?” Layer: For the pharmaceutical industry, PTC ensures a standardized, contaminant-free supply of active ingredients, protecting Intellectual Property by maintaining a consistent genetic profile across production batches.
6.3 Logistical Requirements and Export Infrastructure
Biological success in the lab is irrelevant without specialized logistical infrastructure capable of maintaining “disease-free” status during international transit. Maintaining the viability of in vitro products is a critical barrier to entry and a primary differentiator for established commercial operators.
The Agricultural and Processed Food Products Export Development Authority (APEDA) in India plays a strategic role in facilitating this market access. The International Conclave cum Buyer-Seller Meet (IBSM) held in Aizawl on December 6, 2024, illustrates this impact. The event established trade connections with nine international buyers from Singapore, UAE, Nepal, Jordan, Oman, Azerbaijan, Russia, and Ethiopia. These initiatives align domestic lab outputs with the specific demands of the Middle East, Africa, and Southeast Asia.
Export success relies on two technical-logistical pillars:
- Aseptic Packaging: Crucial for maintaining sterility and viability over long distances.
- Disease-Free Certification: This serves as a vital technical “passport,” allowing products to bypass restrictive phytosanitary barriers that frequently delay or block traditional plant exports.
6.4 Operational Limitations and Risk Mitigation
High-yield, PTC operations are inherently fragile and sensitive to biological and economic disruptions. A significant operational risk is Somaclonal Variation—unpredictable genetic changes during culture that can result in chimeric plants or low-yielding “off-types.” For a commercial laboratory, early detection of these variations is mandatory to protect the firm’s reputation and the grower’s ROI.
To mitigate these risks, stakeholders must evaluate the cost-benefit of emerging technologies:
| Innovation | Barrier |
|---|---|
| Artificial Intelligence (AI): Utilization of Machine Learning (ML) and Deep Learning (DL) for real-time growth monitoring and optimized harvesting. | Regulatory Frameworks: Complex and varying global biotechnology regulations can delay the adoption of automated systems. |
| CRISPR/Cas9: Targeted genome editing to introduce traits like drought tolerance or disease resistance. | Technical Hurdles: Issues with off-target effects and low variability in editing efficiency (e.g., ZFNs having only 1% to 10% efficiency). |
Technical vigilance, including the adoption of ML/DL for early variant detection, is no longer optional for laboratories seeking to maintain industrial-scale reliability.
6.5 Strategic Roadmap for Stakeholders
Industrial-scale plant tissue culture is the definitive cornerstone of sustainable, high-productivity modern agriculture. The convergence of a projected USD 293.35 billion market, the technical superiority of somatic embryogenesis for synthetic seed production, and the logistical framework provided by APEDA creates a robust environment for commercial investment.
Strategic Mandates for Professional Stakeholders:
- Invest in Recalcitrant Species Research: Focus specifically on protocols for monocotyledons and woody plants that are currently difficult to regenerate to secure untapped market segments.
- Align with APEDA Export Protocols: Ensure all lab outputs meet the disease-free certification standards required to bypass phytosanitary barriers in emerging markets like Nepal, Jordan, and Azerbaijan.
- Implement AI for Risk Mitigation: Adopt Machine Learning and Deep Learning systems to provide real-time monitoring of somatic embryogenesis and early detection of somaclonal variation.
By adhering to these mandates, stakeholders can leverage the power of cellular totipotency to ensure a stable, high-quality, and highly profitable botanical supply chain for the global market.
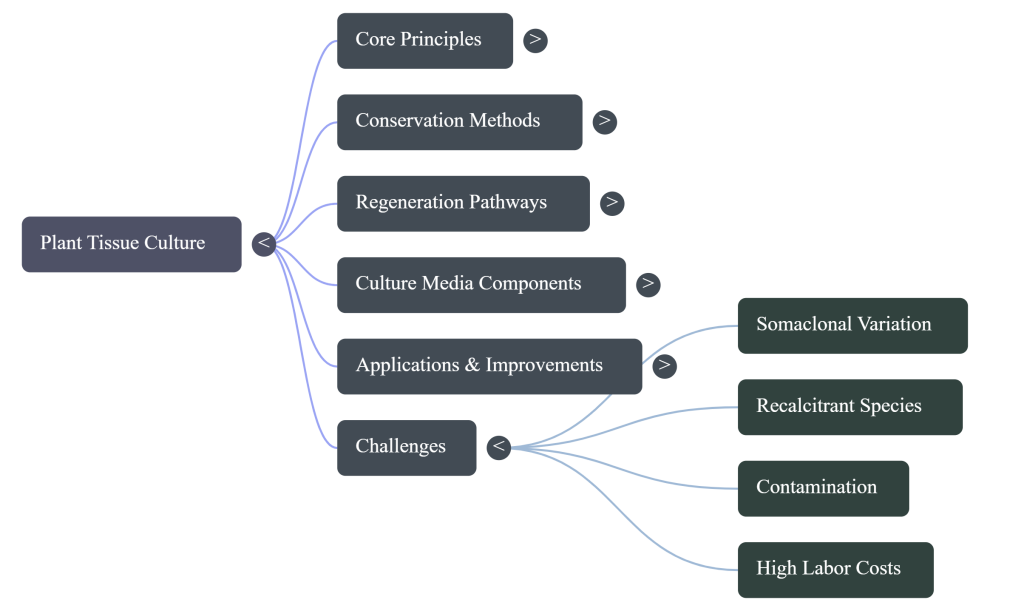
7. Identifying Bottlenecks in Traditional Tissue Culture
Our strategic upgrade begins with a rigorous audit of the limitations inherent in manual systems. Understanding these constraints is essential for converting operational liabilities into data-driven assets. Traditional facilities are currently vulnerable to escalating labor costs and a persistent deficit of highly trained personnel required for delicate aseptic manipulations.
Critically, we must recognize that traditional in-situ conservation is increasingly unreliable due to urbanization, natural disasters, and the volatility of changing government policies. This places the burden of genetic “patrimony” on our facilities. Without automation, the risk of losing genetic diversity to human error, pathogen introduction, and abiotic stress is unacceptably high.

7.1 Critical Constraints: Limitations and Somaclonal Variation
Despite its potential, PTC faces significant biological and economic hurdles.
- Somaclonal Variation: Unpredictable genetic changes during culture. We now identify DNA methylation, histone modification, and RNA interference as the primary epigenetic drivers of this variation.
- Recalcitrance: Many species (monocots and certain woody trees) remain stubbornly resistant to in-vitro regeneration.
- Economic Costs: The high overhead of sterile facilities, specialized labor, and constant electricity.
The “So What?” Layer: Managing somaclonal variation is the “major impediment” to utilizing PTC for pharmaceutical-grade metabolites. Consistency is the currency of the medical industry; any epigenetic drift can jeopardize the regulatory approval of a botanical drug.
7.2 Operational Evolution: Strategic Transformation
| Traditional PTC Constraints | Desired Strategic Outcomes |
|---|---|
| Labor Intensive: High reliance on manual cutting/subculturing. | Scalable Efficiency: High-throughput production via mechanical precision. |
| High Contamination Risk: Human handling leads to aseptic breaches. | Controlled Sterility: Automated, closed-loop systems to prevent pathogen introduction. |
| Physical Space Limits: Land requirements for large forest trees. | In Vitro and Cryo-storage: Maximizing genetic stock per square foot. |
| Variable Protocol Execution: Human error in media and hormone delivery. | Standardized Consistency: AI-driven optimization of nutrient and PGR activity. |
The move from reactive manual labor to proactive data-driven management is facilitated by the integration of Artificial Intelligence.
8. The Modern Frontier: AI Integration and Genome Editing
8.1 Strategic Pillar I: AI-Driven Real-Time Monitoring and Protocol Optimization
Artificial Intelligence transforms the “black box” of plant development into a transparent, optimized pipeline. By deploying neuro-fuzzy logic software and Machine Learning (ML), we can simulate complex growth trajectories and refine in vitro environments with a level of precision impossible for human operators.
A primary focus for our AI systems is the dynamic management of nutrient media, such as MS, B5, or WPM. AI-driven sensors will maintain a precise pH range of 5.4–5.8, a critical parameter that directly dictates the activity of plant growth regulators (PGRs). By optimizing this chemical signaling environment, we ensure that cellular commitment toward the desired developmental pathway—whether rooting or shooting—is achieved with maximum efficiency.

High-Value AI Applications and Phenotyping
- High-Throughput Phenotyping: Utilizing automated multi-spectral imaging systems like the ScanAlyzer to screen thousands of regenerated plants for transgenic or mutant phenotypes.
- Predictive Analytics for Secondary Metabolites: Leveraging Deep Learning to map and enhance biosynthetic pathways for pharmaceutical and cosmetic compounds.
- Contamination Prediction: Using Artificial Neural Networks (ANNs) to detect sub-visual markers of failure, minimizing batch loss through early intervention.
- Metabolic Engineering: Real-time calibration of gaseous and temperature environments (22–28°C) to maximize the production of bioactive phytoconstituents.
These data insights provide the intelligence required to guide our second pillar: the physical execution of micropropagation through robotics.
8.2 Strategic Pillar II: Robotic Automation for Explant Handling and Scalability
Mechanical precision is the key to transitioning from a laboratory scale to a bio-factory model. The strategic deployment of robotics standardizes the most delicate operations of the PTC cycle—the cutting and transplanting of explants.
By removing the human element from these tasks, we significantly mitigate the risk of pathogen introduction and accidental tissue damage. Our automated lines are designed to integrate seamlessly with established biological pathways, including organogenesis and somatic embryogenesis. This is particularly critical for the mass multiplication of ornamentals and woody species where “true-to-type” uniformity is the primary commercial metric.
Automation and Scalability Value Propositions
- Transition to Bioreactors: Moving from manual flasks to chemostat and batch bioreactors for continuous, large-scale production of secondary metabolites.
- 24/7 Operational Capacity: Automated systems operate outside of standard shifts, maximizing the ROI of facility infrastructure.
- Pathogen Mitigation: Closed-loop robotic handling reduces aseptic breaches by 90% compared to manual bench work.
- Uniform PGR Delivery: Precise robotic application of auxins and cytokinins ensures consistent development across millions of clones.
With a healthy, automated pipeline established, we can utilize these clones as the substrate for advanced genetic enhancement.
8.3 Strategic Pillar III: CRISPR/Cas9 for Precise Genetic Modification and Resilience
The final pillar of our roadmap involves moving from mere propagation to active “crop design.” The integration of CRISPR/Cas9 within our tissue culture pipeline allows us to engineer “super crops” with specific traits like drought tolerance and disease resistance.
Our strategy prioritizes the most advanced iterations of gene editing: Prime editing, which allows for accurate insertions and deletions without double-strand breaks, and Base editing, which enables single-base alterations. These methods offer superior specificity compared to traditional techniques. For example, while Zinc Finger Nucleases (ZFNs) typically exhibit an efficiency of only 1% to 10% and carry high risks of non-specific genetic changes, CRISPR-based systems provide a more predictable and scalable solution for commercial trait enhancement.
Strategic “So What?”: Addressing Global Security Gene editing addresses the fundamental challenges of global food security. By bypassing the random DNA integration associated with Agrobacterium-mediated transformation or biolistics, we can rapidly tailor crops to survive regional environmental stressors. This precision accelerates the delivery of high-yielding, resilient varieties to the global market.
8.4 Implementation Roadmap: Phased Integration and Facility Modernization
We will deploy this transformation through a three-phase approach to ensure operational continuity and protect existing germplasm.
Phase 1: Foundation Enhancement
- Media Optimization: Standardize the use of BABI media—a B5 formulation modified with extra calcium that separates nitrate from ammonium to allow for finer nutrient tuning.
- Environmental Calibration: Install high-precision sensors to maintain light intensity at exactly 50–100 μmol m⁻² s⁻¹ and temperature stability within 22–28°C.
- Aseptic Hardening: Transition to chemically defined media to eliminate batch-to-batch variability.
Phase 2: Digital & Mechanical Integration
- AI Deployment: Implement ANNs and neuro-fuzzy logic for real-time monitoring of explant health.
- Throughput Optimization: Use AI to time subculture intervals to coincide specifically with the mid-exponential or early-linear growth phases, maximizing biomass yield and regenerative potential.
- Robotic Integration: Launch automated cutting and transplanting stations to eliminate manual handling risks.
Phase 3: Genetic Advanced Tier
- CRISPR/Cas9 Pipeline: Establish a dedicated pipeline for Prime and Base editing for trait development.
- Long-Term Germplasm Protection: Fully integrate cryopreservation (-196°C) to protect our genetically edited stocks and endangered germplasm from the risks of somaclonal variation and contamination during routine subculturing.
Forward-Looking Statement The integration of AI, robotic automation, and advanced CRISPR technologies marks our transition into a sustainable, precision-engineered bio-factory. By adopting this roadmap, we secure our ability to provide the high-quality, resilient genetic material required for the 21st-century agricultural economy.
9. Conclusion: The Future of Plant Biodiversity and Food Security
Plant Tissue Culture is the indispensable engine of 21st-century biotechnology. By harnessing totipotency, advanced media like BABI, and AI-driven optimization, we can safeguard the planet’s botanical heritage while meeting the needs of a 10-billion-person population. The future requires continued innovation in sustainable practices and ethical vigilance in genetic engineering to ensure that the “green revolution” of the lab translates into food security and ecological stability for the world.
Image Summary




Questions/Answers
1. How does plant cell totipotency enable whole plant regeneration?
Plant cell totipotency is the fundamental principle that enables the regeneration of an entire plant from a single cell or a small piece of tissue (explant). This unique biological property allows differentiated somatic cells to reset their genetic program and undergo new developmental pathways.
The Principle of Totipotency
Totipotency is defined as the genetic potential of a single plant cell to express its full genome, divide, and develop into a fully differentiated, multicellular organism. First proposed by Gottlieb Haberlandt in 1902, this concept suggests that because every cell contains all the necessary genetic information, any cell can theoretically become a complete plant if provided with the correct nutritional and environmental cues.
Mechanisms of Regeneration
Totipotency enables whole plant regeneration through two primary pathways: somatic embryogenesis and organogenesis.
- Somatic Embryogenesis: In this process, somatic (non-sexual) cells are induced to behave like zygotes. These cells develop into bipolar structures—somatic embryos—that possess both a shoot and a root meristem simultaneously. These embryos develop through stages similar to those of natural seeds (globular, heart, and torpedo stages) before germinating into whole plantlets.
- Organogenesis: This pathway involves the formation of specific organs (roots, shoots, or leaves) from cultured tissues. Unlike somatic embryos, these are unipolar structures that develop in a sequence: typically, shoots are induced first, followed by a separate phase for root induction to complete the plantlet.
Cellular Reprogramming
The transition from a differentiated cell to a whole plant requires a process of dedifferentiation and redifferentiation:
- Dedifferentiation: Differentiated cells (like leaf or stem cells) revert to a less committed, more flexible state. This often results in the formation of a callus, an unorganized mass of undifferentiated cells.
- Redifferentiation: Under specific stimuli, these undifferentiated cells gain “competence” and are “determined” for a new developmental fate, eventually forming the organized tissues of a new plant.
Factors Enabling Expression of Totipotency
While most plant cells are theoretically totipotent, they require specific external signals to express this potential:
- Plant Growth Regulators (PGRs): The ratio of auxins to cytokinins is the primary chemical signal for regeneration. High auxin-to-cytokinin ratios generally favor root formation, while high cytokinin-to-auxin ratios favor shoot formation.
- Nutrient Medium: A meticulously controlled environment provides essential macro- and micronutrients, vitamins, and carbon sources (like sucrose) to support the metabolic demands of rapidly dividing and differentiating cells.
- Aseptic Conditions: Regeneration must occur in a sterile environment to prevent contaminating microorganisms from outcompeting the fragile cultured cells for nutrients.
- Organizer Cells: Successful regeneration often begins with the formation of an organizer cell niche—a group of slowly dividing cells that synthesize their own auxin to create the developmental gradients necessary for building new plant organs.
2. How is artificial intelligence revolutionizing modern plant tissue culture?
Artificial intelligence (AI) and machine learning are revolutionizing modern plant tissue culture by transitioning the field from an empirical, “trial-and-error” science to a data-driven, predictive technology. By analyzing complex biological datasets, AI is enhancing the efficiency, precision, and scalability of in vitro propagation.
According to the sources, AI is impacting modern plant tissue culture in the following key ways:
1. Optimization of Culture Media and Growth Conditions
AI algorithms are used to analyze extensive datasets to predict optimal growth outcomes and dynamically adjust protocols.
- Data-Driven Customization: New approaches use AI for the optimization of culture medium composition, helping to identify the most effective balance of macro- and micronutrients for specific genotypes.
- Predictive Modeling: Artificial Neural Networks (ANNs) are employed to simulate plant growth under various in vitro conditions, allowing researchers to anticipate how plants will respond to different environmental factors before physically conducting the experiments.
2. Modeling Complex Regeneration Processes
AI is particularly effective at modeling intricate developmental pathways like somatic embryogenesis and organogenesis.
- Machine Learning Algorithms: Scientists have begun applying machine learning, such as Multilayer Perceptron (MLP) and Support Vector Regression (SVR), to design model regeneration processes. For example, these algorithms have been used to model somatic embryogenesis in species like chrysanthemum.
- Anther Culture and Image Processing: AI is used alongside image processing to model callus induction and regeneration in anther cultures (e.g., in tomatoes), providing a more accurate way to monitor developmental progress.
3. Automation and Robotics
The integration of AI with automation is setting new industry standards for producing high-quality, genetically uniform plantlets.
- Precision Handling: Robotic systems equipped with AI can now perform delicate operations, such as cutting and transplanting tissue cultures. This reduces the need for expensive manual labor, which typically accounts for a large percentage of production costs, and significantly lowers the risk of human-introduced contamination.
- Scaling Production: By automating routine tasks and optimizing resource use (like water and light), AI enables laboratories to scale up production to meet global food security demands more efficiently.
4. Real-Time Monitoring and Industry Applications
AI enhances real-time monitoring throughout the tissue culture lifecycle, from initiation to marketing.
- Case Study (Cannabis): In the Cannabis sativa industry, AI and machine learning are being used to optimize in vitro germination and growth indices, improving productivity and product quality while maintaining soil fertility and reducing the excess use of pesticides and water.
- High-Throughput Phenotyping: AI is increasingly used for high-throughput phenotyping, which involves screening large numbers of in vitro regenerated plants or germinated seedlings for specific transgenic or mutant phenotypes using automated imaging systems.
In conclusion, the burgeoning role of AI and machine learning provides novel approaches to long-standing problems in plant biotechnology, such as overcoming recalcitrance in difficult-to-propagate species and reducing the high costs associated with traditional manual tissue culture methods.
3. What is the specific role of the auxin-to-cytokinin ratio?
The specific role of the auxin-to-cytokinin ratio is to act as a chemical signal that determines the morphogenetic fate of plant tissues in culture. This fundamental concept, first demonstrated by Skoog and Miller in 1957, establishes that the relative concentrations of these two hormones dictate whether a tissue will develop roots, shoots, or remain as undifferentiated callus.
Key Morphological Outcomes
The interaction between auxins and cytokinins typically results in the following developmental pathways:
- Shoot Formation: A high cytokinin-to-auxin ratio stimulates the formation and proliferation of adventitious and axillary shoots. For example, in tobacco callus, increasing the cytokinin concentration relative to auxin initiates shoot bud differentiation.
- Root Formation: A high auxin-to-cytokinin ratio (or a moderate-to-high auxin level in the absence of cytokinin) favors the induction and proliferation of adventitious roots.
- Callus Induction: A balanced ratio where both auxin and cytokinin concentrations are relatively high or intermediate leads to the development of an unorganized mass of undifferentiated cells known as callus.
Factors Influencing the Ratio
While the general principle of the ratio is widely applicable, several factors can modify how a specific tissue responds:
- Species and Tissue Specificity: The precise ratio required for a particular response is unique to each plant species and variety. Some genotypes respond to the same hormone treatments differently based on their internal genetic setup.
- Endogenous Hormone Levels: The success of the exogenous (added) ratio depends heavily on the endogenous (naturally occurring) hormones already present within the explant. For instance, meristematic tissues often have higher natural auxin concentrations, which must be accounted for when balancing the media.
- Sequential Manipulation: Regeneration often requires multiple steps where the ratio is adjusted over time. For instance, in some species, a high auxin ratio may first be used to induce somatic embryos, followed by a move to a hormone-free or cytokinin-dominant medium to allow those embryos to mature and germinate into whole plants.
- Hormonal Cross-talk: Phytohormones do not act in isolation; they interact with other regulators like gibberellins, abscisic acid, and ethylene to coordinate complex cellular processes.
Physiological and Molecular Basis
The ratio influences development by regulating cell division and differentiation. Auxin is primarily responsible for cell elongation and root initiation, while cytokinins are essential for cell division and the development of lateral or axillary buds. On a molecular level, this hormonal balance interacts with gene signaling pathways (such as WUSCHEL and WOX genes) to establish the necessary cellular polarity and “organizer cells” required for building a new plant organism.
4. How do somatic embryogenesis and organogenesis differ in plant regeneration?
Somatic embryogenesis and organogenesis are the two primary pathways for plant regeneration in tissue culture, differing fundamentally in their anatomical structures, hormonal requirements, and developmental sequences.
Structural and Anatomical Differences
- Polarity: Somatic embryogenesis (SE) results in the formation of bipolar structures, meaning the embryo possesses both a shoot and a root meristem simultaneously. In contrast, organogenesis produces unipolar structures, where either a shoot or a root is formed independently, requiring a separate subsequent step to induce the missing organ to complete the plantlet.
- Vascular Connection: Somatic embryos develop with no vascular connection to the original explant or maternal tissue. Organogenic structures, however, establish a direct connection with pre-existing vascular tissue dispersed within the callus or explant.
- Cellular Origin: Somatic embryos are generally considered to have a unicellular origin. Organogenic shoots often develop through the simultaneous division of several cells and are thus multicellular in origin.
Hormonal Regulation
- Auxin vs. Cytokinin Ratio: Organogenesis is primarily governed by the auxin-to-cytokinin ratio; high cytokinin favor shoot formation, while high auxin favor root formation.
- Induction Stimuli: Somatic embryogenesis is typically induced by high concentrations of synthetic auxins, most notably 2,4-D, which triggers the formation of proembryogenic masses (PEMs). Development of the embryos then usually proceeds after the auxin is removed or its concentration is significantly reduced.
Developmental Stages
- Embryogenic Pathway: Somatic embryos follow a developmental sequence identical to zygotic embryos, progressing through globular, heart, torpedo, and cotyledonary stages in dicots. In monocots, they pass through globular, scutellar, and coleoptile stages.
- Organogenic Pathway: Organogenesis proceeds through stages of dedifferentiation (attaining competence), induction (becoming determined for a specific organ), and differentiation (morphological development).
Practical Advantages and Limitations
- Scalability: Somatic embryogenesis is considered more efficient for mass propagation because a single flask or bioreactor can produce millions of embryos that germinate directly into plantlets. Organogenesis is generally more labor-intensive and time-consuming.
- Genetic Fidelity: While both processes can occur directly (from explant) or indirectly (via callus), direct organogenesis from pre-existing meristems is often preferred for maintaining high genetic fidelity, as it minimizes the risk of somaclonal variation associated with an intervening callus phase. Somatic embryos are also useful for producing synthetic seeds by encapsulating them in protective gels for long-term storage and easier transport.
5. What are the common challenges or limitations of plant tissue culture?
While plant tissue culture is a powerful biotechnological tool, its implementation is hindered by several significant biological, genetic, and economic challenges.
Biological and Physiological Challenges
- Microbial Contamination: This is a persistent and primary challenge caused by bacteria, fungi, yeast, and other microorganisms. These contaminants compete with the explant for nutrients, release toxins, and can lead to total culture loss. Endophytic contamination (microbes living within the plant tissue) is particularly difficult to eliminate through surface sterilization.
- Hyperhydricity (Vitrification): This physiological disorder results in plantlets having a glassy, translucent, and fragile appearance. It is often caused by high humidity in culture vessels or imbalances in growth regulators, leading to poor survival during the hardening phase.
- Browning and Phenolic Exudation: Explants, especially from woody or mature trees, often exude phenolic compounds into the medium. Upon oxidation, these form toxic quinones that turn the medium black and cause tissue necrosis and death.
- Acclimatization Difficulties: In vitro plants are developed under high humidity and low light, resulting in malfunctioning stomata and a lack of protective epicuticular wax. These abnormalities make them highly susceptible to desiccation and high mortality when transferred to soil.
Genetic Challenges
- Somaclonal Variation: Unintended and unpredictable genetic or epigenetic changes often occur during the culture process. This instability leads to phenotypic variations (off-types) that make regenerated plants different from the mother plant, which is a major drawback for commercial cloning.
- Loss of Morphogenetic Potential: In many species, the ability of a cell line to regenerate or undergo embryogenesis declines over time, eventually making regeneration impossible in aged cultures.
Economic and Operational Challenges
- High Production Costs: Tissue culture is capital, energy, and labor-intensive. Costs are driven by the need for specialized equipment, sophisticated infrastructure, and expensive consumables like chemicals and gelling agents. Electricity for lighting and climate control can account for up to 60% of production costs in some regions.
- Labor Intensity: The process involves tedious manual tasks, such as the surgical excision and transfer of explants, which typically requires highly skilled personnel.
Species-Specific Recalcitrance
- Monocots and Woody Plants: Many plant species are categorized as recalcitrant, meaning they are difficult or impossible to regenerate or transform under existing in vitro protocols. This is common in gymnosperms, monocotyledons (like cereals), and mature woody trees that have lost their juvenile physiological state.
- Non-Synchronous Development: In somatic embryogenesis, embryos often develop at different rates, making it difficult to harvest a uniform population of plantlets simultaneously.
6. How do Artificial Neural Networks simulate plant growth in vitro?
Artificial Neural Networks (ANNs), a subset of machine learning and artificial intelligence, simulate plant growth in vitro by acting as computational tools that transition tissue culture from an empirical “trial-and-error” approach to a data-driven predictive science.
According to the sources, ANNs simulate and enhance growth through the following mechanisms:
1. Modeling Complex Developmental Pathways
ANNs are used to create predictive models for intricate biological processes that are otherwise difficult to standardize.
- Somatic Embryogenesis: Scientists use algorithms such as the Multilayer Perceptron (MLP)—a type of ANN—to design models for regeneration processes. For example, these models have been used to predict the success of somatic embryogenesis in species like chrysanthemum, comparing accuracy with other methods like Support Vector Regression (SVR).
- Rhizogenesis and Acclimatization: ANNs have been specifically applied to model the rooting phase (rhizogenesis) and the subsequent adaptation of plants to external environments (acclimatization) in species like Vitis vinifera (grape).
- Callus Induction: In crops like tomato, ANNs are combined with image processing to model callus induction and regeneration in anther cultures, providing a more accurate way to monitor and predict developmental progress [Previous Context].
2. Optimization of Growth Conditions and Media
ANNs analyze extensive and complex biological datasets to identify the best environmental and nutritional variables for growth.
- Data-Driven Customization: AI algorithms can predict optimal growth outcomes and dynamically adjust protocols based on the results. This includes optimizing the composition of culture media—fine-tuning the balance of macro- and micronutrients for specific genotypes.
- Predicting Growth Indices: In the cannabis industry, ANNs and machine learning are employed to optimize in vitro germination and growth indices. This modeling helps save resources like water, pesticides, and herbicides while maintaining soil fertility.
3. Simulation Under Variable Conditions
ANNs can simulate how plants will respond to different environmental factors before physical experiments are conducted.
- Scenario Testing: These networks can simulate the growth of plants under various in vitro conditions, allowing researchers to anticipate responses to light intensity, temperature, and hormone concentrations.
- High-Throughput Phenotyping: AI systems are increasingly used to screen large numbers of regenerated plants or germinated seedlings for specific transgenic or mutant phenotypes using automated imaging, allowing for rapid data collection that feeds back into the neural network for further simulation accuracy.
4. Integration with Automation
The simulations provided by ANNs often drive robotic systems that perform delicate operations such as cutting and transplanting tissue cultures. By using AI to ensure consistent handling, laboratories can reduce the risk of human-introduced contamination and lower production costs, which typically account for a large portion of manual labor.
7. How do robotic systems reduce human error in tissue culture?
Robotic systems reduce human error in plant tissue culture by replacing the most delicate and repetitive manual tasks with high-precision automation, thereby minimizing the primary source of laboratory contamination and operational inconsistency.
The sources highlight several specific ways these systems improve outcomes:
1. Minimizing Human-Introduced Contamination
Humans are a major source of contamination in the laboratory environment; it is estimated that a single operator generates one to five million particles per minute.
- Aseptic Handling: Robots perform tasks like cutting and transplanting within sterile environments, significantly lowering the risk of introducing bacteria, fungi, or spores from skin, clothing, or hair.
- Reduced Traffic: By automating routine tasks, laboratories can reduce the “foot traffic” and physical presence of personnel in ultra-clean zones, which helps maintain higher levels of asepsis.
2. Precision in Surgical Tasks
Manual micropropagation is highly labor-intensive, often accounting for 60% to 70% of total production costs.
- Consistent Excision: Robotic systems can perform delicate surgical operations, such as the excision of tiny shoot tips or the cutting of nodal segments, with a level of precision and consistency that is difficult for human operators to maintain over long shifts.
- Standardized Transfers: Robots ensure that explants are handled uniformly during transfer to fresh media, reducing the physical bruising or squashing that can occur when humans use forceps and scalpels.
3. Automation of Repetitive Processes
Robotic automation eliminates the variability inherent in manual “trial-and-error” procedures.
- Media Preparation and Dispensing: Automated systems replace manual filling of culture containers, ensuring that precisely measured volumes of nutrient-rich media are dispensed every time.
- Surface Sterilization: Automated series of events in surface sterilization help avoid the human error of over-exposing or under-exposing tissues to harsh chemicals like bleach or mercuric chloride, which can cause tissue necrosis if handled incorrectly.
4. Integration with Bioreactors and AI
The combination of robotics with other advanced technologies further limits the opportunity for human error.
- Large-Scale Handling: In automated bioreactor systems, cultures can be moved from the multiplication phase to the growth phase with minimal human intervention. For instance, a single operator managing a robotic system can move 10,000 to 12,000 plants in less than a day, a volume that would be impossible and highly prone to error if done manually.
- Data-Driven Adjustments: Robotic systems integrated with AI and Artificial Neural Networks (ANNs) can analyze datasets to predict optimal growth outcomes and dynamically adjust protocols without human bias or oversight.
By standardizing these complex biological protocols, robotic systems transform tissue culture from an “art” dependent on the skill of an “experienced eye” into a data-driven, reproducible industrial technology.
8. What are the key benefits of using somatic embryogenesis?
Somatic embryogenesis (SE) is a highly efficient biotechnological tool for plant regeneration that offers several distinct advantages over other methods like organogenesis.
1. High-Efficiency Mass Propagation
Somatic embryogenesis is regarded as the most efficient in vitro procedure for large-scale plant multiplication.
- Scale of Production: A single liter of culture medium can potentially hold up to 1.35 million somatic embryos.
- Automation: Because embryos can be produced in liquid media, the process is highly amenable to automation using bioreactors, which significantly reduces manual labor costs, space requirements, and energy inputs.
- Rapid Cycling: It permits the production of hundreds and thousands of plants in a continuous process from a small piece of initial tissue.
2. Developmental and Structural Efficiency
Unlike organogenesis, which produces unipolar structures (only shoots or only roots), somatic embryos are bipolar structures.
- Simultaneous Growth: Somatic embryos possess both a shoot and a root meristem simultaneously, allowing them to germinate directly into whole plantlets without the need for a separate, time-consuming root induction phase.
- No Vascular Connection: These embryos develop with no vascular connection to the original maternal tissue, which facilitates their easy isolation and handling.
3. Ideal Platform for Genetic Engineering
SE is often the preferred method for producing genetically modified (transgenic) plants.
- Single-Cell Origin: Somatic embryos generally originate from a single cell. This is a major advantage for genetic transformation because it ensures that the resulting plant is not a chimera (a mix of transformed and untransformed cells) and maintains high genetic fidelity.
- Selection and Transformation: Embryogenic cell lines and suspensions are excellent targets for DNA delivery methods like Agrobacterium-mediated transformation or particle bombardment.
4. Advanced Storage and Transportation Options
The nature of somatic embryos allows for unique applications in conservation and logistics:
- Synthetic Seeds: Somatic embryos can be encapsulated in protective gels (like calcium alginate) to create synthetic or artificial seeds. These seeds are easy to handle, transport, and can be sown directly into soil like conventional seeds.
- Long-Term Conservation: SE provides a platform for germplasm preservation. Dormancy can be induced in these embryos, enabling them to be stored for extended periods through cryopreservation in liquid nitrogen.
- Year-Round Availability: Clonal propagation can be maintained regardless of weather or seasonal variations, ensuring a steady supply of planting material.
5. Research and Industrial Applications
- Secondary Metabolites: SE can be used to scale up the production of secondary metabolites—valuable chemicals used in pharmaceuticals and agrochemicals—by using bioreactor systems.
- Virus Elimination: It is an effective tool for developing virus-free plants, as the regeneration process, sometimes combined with heat therapy, can eliminate systemic infections.
- Basic Research: SE serves as a model system for studying the morphological, physiological, and molecular events of embryo development and cell differentiation in higher plants.
9. Tell me more about AI and automation in Cannabis sativa.
The integration of artificial intelligence (AI) and automation is transforming the production of Cannabis sativa, shifting it from a traditional manual practice into a highly efficient, data-driven industrial technology. This is particularly significant because C. sativa has historically been considered recalcitrant to in vitro manipulations, a challenge that modern AI is beginning to overcome.
1. Artificial Neural Networks (ANNs) and Modeling
Artificial Neural Networks (ANNs), along with Machine Learning (ML) and Deep Learning (DL), are being used to gather and analyze complex data across the cannabis industry.
- Simulating Growth: ANNs can simulate the growth of cannabis plants under various in vitro conditions, allowing researchers to predict how they will respond to different environmental factors before physical trials are conducted.
- Optimizing Protocols: AI algorithms, such as the Multilayer Perceptron (MLP), help optimize in vitro germination and growth indices specifically for industrial hemp. This data-driven approach allows for the development of new protocols for difficult tasks like somatic embryogenesis, cryopreservation, and anther culture for haploid production.
- Genetic and Metabolic Insights: AI applications are being explored to investigate cannabinoid gene expression and the effects of growth regulators on cannabinoid content, providing deeper insights into metabolic engineering.
2. Operational Efficiency and Resource Management
AI technology provides cannabis businesses with the tools to improve productivity while reducing environmental impact.
- Resource Saving: These technologies enable precise control that saves water and reduces the excessive use of pesticides, herbicides, and fertilizers, which helps maintain soil fertility.
- Real-Time Monitoring: AI is utilized for real-time monitoring of crop health, which enhances harvesting, processing, and marketing strategies.
- Manpower Optimization: By automating routine data collection and analysis, AI leads to the more efficient use of manpower and elevated productivity.
3. Robotic Automation and Precision
Robotic systems are setting new industry standards for the mass production of high-quality, genetically uniform cannabis plantlets.
- Reducing Human Error: Robotic systems perform delicate surgical tasks such as cutting and transplanting tissue cultures, which minimizes human error and the risk of contamination—a frequent “nightmare” for the industry.
- Scalability: Automation handles the repetitive, labor-intensive aspects of micropropagation, significantly reducing production costs and allowing for larger-scale operations that were previously impossible with manual labor.
4. Advanced Genetic Tools
Beyond monitoring and growth, AI and automation support the application of advanced molecular tools in C. sativa.
- Genome Editing: Automation facilitates the delivery of CRISPR/Cas9 systems into cannabis tissues to achieve targeted mutagenesis (e.g., for albinism or modified metabolite profiles).
- Nanotechnology Integration: Automated processes are being used to develop nanoparticle-based genetic transformation methods for cannabis, which can deliver DNA directly through cell walls to improve metabolic traits like cannabinoid and terpene production.
10. How do AI neural networks simulate plant growth conditions?
Artificial Neural Networks (ANNs), a subset of machine learning, simulate plant growth conditions by acting as computational tools that transition tissue culture from an empirical, “trial-and-error” approach to a data-driven predictive science. They process large, complex biological datasets to identify patterns and predict how plant tissues will respond to specific environmental and nutritional variables.
Based on the sources, ANNs simulate growth conditions through the following mechanisms:
1. Modeling Complex Developmental Pathways
ANNs are employed to create predictive models for intricate biological processes that are otherwise difficult to standardize.
- Somatic Embryogenesis: Scientists use algorithms such as the Multilayer Perceptron (MLP)—a type of ANN—to design models for regeneration. For example, these models have predicted the success of somatic embryogenesis in chrysanthemum by analyzing the effects of different plant growth regulators.
- Rhizogenesis and Acclimatization: ANNs have been specifically applied to model the rooting phase (rhizogenesis) and the subsequent adaptation of plants to external environments (acclimatization) in species like Vitis vinifera (grape).
- Callus Induction: In crops like tomato, ANNs are combined with image processing to model callus induction and regeneration in anther cultures, providing a precise way to monitor and predict developmental progress.
2. Optimization of Culture Media
ANNs analyze extensive datasets to identify the ideal balance of nutrients for a given species or genotype.
- Nutrient Customization: AI algorithms can predict optimal growth outcomes and dynamically adjust protocols by fine-tuning the balance of macronutrients and micronutrients in the basal medium.
- Growth Indices: In the industrial hemp (Cannabis sativa) industry, ANNs and machine learning algorithms are used to optimize in vitro germination and growth indices. This modeling helps improve productivity and product quality while reducing the excess use of water, pesticides, and herbicides.
3. Simulation and Scenario Testing
ANNs can simulate the growth of plants under various in vitro conditions, allowing researchers to anticipate responses before conducting physical experiments.
- Predictive Simulation: These networks simulate how plants will react to different levels of light intensity, temperature, and hormone concentrations.
- High-Throughput Phenotyping: AI is increasingly used for high-throughput phenotyping, which involves screening large numbers of in vitro regenerated plants for specific transgenic or mutant phenotypes using automated imaging systems. The data from these screenings is fed back into neural networks to further refine the accuracy of the simulations.
4. Integration with Laboratory Automation
The simulations provided by ANNs often drive robotic systems that handle delicate operations like cutting and transplanting tissue cultures. By using AI to analyze datasets and predict optimal outcomes, these systems ensure consistent handling, minimize human error, and reduce the risk of contamination, which are critical factors for successful plant growth.
11. What is the history behind the concept of totipotency?
The history of the concept of totipotency is deeply intertwined with the development of cell theory and the birth of plant tissue culture as a scientific discipline.
Foundational Roots in Cell Theory
The theoretical roots of totipotency trace back to the mid-19th century with the work of biologists M.J. Schleiden (1838) and T. Schwann (1839). They proposed the cell theory, establishing that the cell is the basic structural and functional unit of all living organisms. Schleiden and Schwann specifically hypothesized that plant cells are autonomous and possess totipotent properties, meaning each cell independently regulates its metabolic activity and retains the inherent ability to regenerate into a whole plant.
Haberlandt’s Formal Proposition (1902)
While the idea existed conceptually, Gottlieb Haberlandt formally proposed the principle of totipotentiality in a visionary address to the German Academy of Science in 1902. Haberlandt viewed the plant cell as an “elementary organism” and predicted that one could successfully cultivate “artificial embryos from vegetative cells” if provided with a suitable environment.
Although Haberlandt is regarded as the “father of plant tissue culture,” his own experiments with isolated photosynthetic leaf cells and epidermal hairs failed to produce cell division. Despite this experimental failure, his work established the theoretical foundation that defined the field for the next century.
Experimental Validation (1950s–1960s)
The experimental evidence required to support Haberlandt’s hypothesis was not achieved for more than 50 years. A turning point occurred in the late 1950s:
- Steward et al. (1958) and Reinert (1958/1959) independently provided the first proof of totipotency by regenerating whole plants from somatic cells of carrot (Daucus carota).
- Steward successfully regenerated entire plants from differentiated segments of secondary phloem, demonstrating that even mature, specialized cells could reset their developmental program.
- In 1965, Vasil and Hildebrandt realized Haberlandt’s “dream” by producing a whole tobacco plant from a single isolated cell in microculture.
Expanding the Scope of Totipotency
Following these initial successes, the concept was demonstrated in other cellular systems:
- Haploid/Gametic Cells: In 1964/1966, Guha and Maheshwari demonstrated the totipotency of haploid cells (pollen grains) by producing embryos and plantlets from cultured anthers of Datura innoxia.
- Protoplasts: In 1971, Takebe et al. achieved a major milestone by demonstrating the totipotency of protoplasts (cells with the cell wall removed), regenerating whole tobacco plants from isolated mesophyll protoplasts.
Today, totipotency remains the fundamental principle of plant biotechnology, defined as the genetic potential of a single plant cell to express its entire genome and develop into a fully differentiated, multicellular organism.
12. How does somatic embryogenesis compare to zygotic embryogenesis?
Somatic embryogenesis (SE) and zygotic embryogenesis (ZE) are two pathways of plant development that result in the formation of an embryo—a bipolar structure containing both shoot and root meristems. While they share many morphological and developmental similarities, they differ fundamentally in their origin, nourishment, and maturation processes.
Key Similarities
- Structural Polarity: Both somatic and zygotic embryos possess a bipolar organization with distinct shoot and root axes.
- Developmental Stages: Both follow a nearly identical sequence of morphological transformations. In dicots, this includes the globular, heart, torpedo, and cotyledonary stages. In monocots, they progress through globular, scutellar, and coleoptile stages.
- Molecular Markers: Certain genes, such as SERK (Somatic Embryogenesis Receptor Kinase), are expressed specifically during the early stages of both somatic and zygotic embryogenesis, indicating shared molecular mechanisms for acquiring embryonic identity.
Fundamental Differences
| Feature | Somatic Embryogenesis (SE) | Zygotic Embryogenesis (ZE) |
|---|---|---|
| Origin | Derived from somatic (vegetative) cells. | Derived from a fertilized egg (zygote). |
| Reproduction | Asexual; bypasses meiosis and fertilization. | Sexual; the result of gametic fusion. |
| Connection | No vascular connection to the maternal tissue; embryos are physically independent. | Historically nourished via a suspensor and endosperm within the seed. |
| Maturation | Often skips the final maturation phase, lacking a natural quiescent (resting) stage. | Undergoes a defined maturation phase characterized by dormancy and desiccation tolerance. |
| Synchrony | Typically asynchronous; all developmental stages may coexist in one culture. | Highly synchronous within the developing seed. |
| Morphology | Somatic embryos are often larger and have wider hypocotyls and cotyledons due to a lack of physical constraints from a seed coat. | Shape is often compressed or flattened by the physical constraints of the surrounding seed. |
Practical Implications
- Genetic Uniformity: Because somatic embryos typically originate from a single cell, they are ideal for cloning and genetic engineering, as they maintain high genetic fidelity and avoid the formation of chimeras. However, the intervening callus phase in indirect SE can sometimes lead to somaclonal variation (mutations).
- Regeneration Ability: Somatic embryos can be germinated directly into whole plantlets without needing a separate rooting step, making them more efficient for large-scale mass propagation than organogenesis.
- Biotechnology: SE serves as an essential tool for creating synthetic seeds—somatic embryos encapsulated in protective gels—which can be handled and sown similarly to natural seeds. Conversely, embryo rescue is a specialized ZE technique used to save hybrid embryos from distant crosses that would naturally abort on the plant due to endosperm failure.
13. What is somaclonal variation and why is it a problem?
Somaclonal variation refers to the phenotypic and genotypic variability that arises in plants regenerated from any form of cell culture. While tissue culture is typically designed to produce identical “true-to-type” clones, the process often results in offspring with different characteristics from the mother plant.
What is Somaclonal Variation?
The term covers a wide spectrum of genetic and epigenetic changes induced during the in vitro process. These variations can be categorized by their biological nature:
- Genetic Changes: These are stable, heritable mutations that are sexually transmitted to the next generation. They include chromosomal rearrangements (deletions, inversions, duplications), changes in chromosome number (polyploidy or aneuploidy), and point mutations.
- Epigenetic Changes: These are typically non-heritable phenotypic variations that often disappear when the plant is reproduced sexually. Common examples involve alterations in DNA methylation and chromatin remodeling.
Causes and Origins
Somaclonal variation originates from a combination of pre-existing variability in the donor tissue and culture-induced variation caused by the stress of the in vitro environment.
- Genomic Stress: Removing cells from their natural environment and placing them in artificial media is a “traumatic experience” or shock that can lead to the breakdown of normal cellular controls.
- Plant Growth Regulators (PGRs): High concentrations of certain hormones, particularly the synthetic auxin 2,4-D, are highly mutagenic and are frequently correlated with increased variation.
- The Callus Phase: Regeneration protocols that involve an intervening callus phase (indirect organogenesis or somatic embryogenesis) are significantly more prone to variation than those utilizing pre-existing meristems.
- Culture Age: The frequency of variation generally increases with the duration of the culture and the number of subculture cycles.
Why is it a Problem?
In the context of commercial micropropagation, somaclonal variation is primarily viewed as a serious limitation due to its unpredictability and the following risks:
- Loss of Clonal Fidelity: The fundamental goal of many tissue culture operations is mass cloning of elite genotypes. Somaclonal variation undermines this by producing “off-types” that do not match the desirable traits of the mother plant.
- Economic Impact: Unintended variations can lead to crop failure, reduced yields, or poor field performance, resulting in massive economic losses for the tissue culture industry.
- Production Limitations: High frequencies of variation have historically prevented the use of tissue culture for mass propagation in some species, most notably oil palm.
- Difficulties in Genetic Engineering: Unwanted variations in transgenic plants can confound the analysis of the introduced gene, as the resulting phenotype may be due to a culture-induced mutation rather than the transgene itself.
Despite these problems, somaclonal variation is sometimes considered a “double-edged sword” because it provides a novel source of genetic variability that breeders can exploit to develop new varieties with improved traits like disease resistance, salt tolerance, or higher yield.
14. How do auxins and cytokinins balance root and shoot growth?
The balance between auxins and cytokinins is the primary chemical signal that determines the developmental pathway of plant cells, specifically regulating whether a tissue will regenerate into roots, shoots, or remain as an unorganized mass. This fundamental discovery was first demonstrated by Skoog and Miller in 1957, establishing the concept of hormonal control of organ formation through varying concentration ratios.
The Morphogenetic Outcomes
The relative concentrations of these two plant growth regulators (PGRs) dictate the following developmental fates:
- Root Formation: A high auxin-to-cytokinin ratio favors the induction and proliferation of adventitious roots. For instance, in Stevia rebaudiana, maximum root induction was achieved when the medium was supplemented with the auxin NAA.
- Shoot Regeneration: A high cytokinin-to-auxin ratio promotes the initiation and proliferation of adventitious and axillary shoots. For example, in tobacco pith cultures, high kinetin concentrations relative to auxin were required to induce shoot parts.
- Callus Induction: A balanced ratio of both hormones leads to the development of an unorganized mass of undifferentiated cells known as callus.
Natural Reciprocal Gradients
In intact plants, this balance is maintained through a system of reciprocal gradients. Auxin is synthesized primarily in the shoot apical meristem and transported in a basipetal (downward) direction to the roots. Conversely, cytokinins are synthesized mostly in the roots and transported in an acropetal (upward) manner via the xylem to promote shoot development. This interaction serves as a signal system to coordinate the growth and development of the entire plant.
Cellular and Molecular Interaction
At the cellular level, auxins and cytokinins interact through complex signaling pathways and gene regulation:
- Cell Division and Expansion: Auxin generally favors root induction at lower concentrations but initiates callusing at higher levels, while cytokinins primarily trigger cell division and axillary bud development.
- Hormonal Cross-talk: The concentration of one hormone added to a culture can indirectly influence morphogenesis by inducing changes in the levels of the other hormone within the cells. For example, auxin-induced organogenesis is often accompanied by endogenous cytokinin production.
- Gene Regulation: These hormones regulate “organizer cells” and stem cell niches by influencing transcription factors such as WUSCHEL (WUS) and WOX. Cytokinins act as inducers and maintainers of auxin biosynthesis, which is necessary for creating the gradients that drive cell specification and differentiation.
While the general model of the auxin-to-cytokinin ratio is widely applicable, the specific response is unique to each species, variety, and tissue type used as an explant. Factors such as the endogenous hormone levels already present in the tissue also significantly influence the outcome of exogenous hormone treatments.
15. Explain the difference between somatic embryogenesis and organogenesis.
Somatic embryogenesis and organogenesis are the two primary pathways for plant regeneration in tissue culture, and they differ fundamentally in their anatomical structure, cellular origin, and hormonal requirements.
1. Structural Polarity and Vascular Connection
- Somatic Embryogenesis (SE): This process results in the formation of bipolar structures—somatic embryos—that possess both a shoot and a root meristem simultaneously. A defining feature of somatic embryos is that they have no vascular connection to the original explant or maternal tissue.
- Organogenesis: This pathway produces unipolar structures, meaning it forms either a shoot or a root independently. Unlike somatic embryos, these structures develop procambial strands that establish a direct connection with pre-existing vascular tissue within the callus or explant.
2. Cellular Origin
- Somatic Embryogenesis: Somatic embryos are generally considered to be unicellular in origin, meaning they develop from a single competent somatic cell.
- Organogenesis: Regenerated organs, such as shoots, typically develop through the simultaneous division of several cells and are therefore multicellular in origin.
3. Hormonal Regulation
- Somatic Embryogenesis: The primary trigger for SE is typically a high concentration of auxins, most notably the synthetic auxin 2,4-D. The embryos often complete their development and maturation after being transferred to an auxin-free or low-auxin medium.
- Organogenesis: This process is primarily governed by the auxin-to-cytokinin ratio. High cytokinin-to-auxin ratios favor shoot formation, whereas high auxin-to-cytokinin ratios favor root formation.
4. Developmental Modes (Direct vs. Indirect)
Both processes can occur through two different routes:
- Direct: The embryos or organs originate directly from the explant tissue without an intervening callus phase.
- Indirect: The explant first undergoes dedifferentiation to form an unorganized mass of cells called callus, from which embryos or organs later arise.
5. Practical Implications
- Efficiency and Scaling: Somatic embryogenesis is considered more efficient for mass propagation because a single flask or bioreactor can produce millions of embryos that germinate directly into whole plantlets.
- Labor and Time: Organogenesis is generally more time-consuming and labor-intensive because it requires separate induction steps for shoots and then for roots.
- Genetic Fidelity: Direct organogenesis is often preferred for commercial cloning because it is less prone to somaclonal variation (genetic mutations) compared to indirect pathways involving a callus phase.
16. How do ANNs help in overcoming plant recalcitrance?
Artificial Neural Networks (ANNs) help overcome plant recalcitrance by transitioning tissue culture from an empirical, “trial-and-error” approach to a data-driven predictive science. Recalcitrance occurs when a plant cell or tissue fails to respond to in vitro manipulations, such as failing to divide or regenerate into a whole plant, a common issue in many woody species, monocots, and legumes.
According to the sources, ANNs specifically address recalcitrance through the following mechanisms:
1. Modeling Complex Regeneration Processes
ANNs are used to create highly accurate models of intricate biological processes that are difficult to standardize across different genotypes.
- Predicting Somatic Embryogenesis: Scientists apply ANN algorithms, such as the Multilayer Perceptron (MLP), to model and predict the success of somatic embryogenesis in difficult species like chrysanthemum.
- Callus and Rhizogenesis Modeling: ANNs are used to monitor and predict callus induction (e.g., in tomato anther cultures) and rhizogenesis (root formation), allowing researchers to identify the exact conditions needed for these critical stages.
2. Optimization of Culture Media and Growth Conditions
Recalcitrant species often have very specific and narrow nutritional requirements that traditional media optimization cannot easily identify.
- Customized Nutrient Balances: AI algorithms analyze extensive datasets to predict optimal growth outcomes and dynamically adjust protocols, fine-tuning the balance of macro- and micronutrients for specific, unresponsive genotypes.
- Hormonal Control: ANNs help determine the precise auxin-to-cytokinin ratios required for specific species, as endogenous hormone levels (naturally occurring in the plant) can significantly vary and interfere with standard treatments.
3. Simulation and Scenario Testing
ANNs allow researchers to virtually test thousands of variables before conducting time-consuming and expensive physical experiments.
- Virtual Growth Simulation: ANNs can simulate plant growth under various in vitro conditions, such as different light intensities, temperatures, and hormone concentrations.
- Predicting Genotypic Response: Because recalcitrance is often genotype-specific, AI can help predict how a new, untested variety will respond based on data from related species or cultivars.
4. Integration with Automation and Robotics
The simulations driven by ANNs are often integrated with robotic systems to ensure the high level of precision required by recalcitrant tissues.
- Precision Handling: Robotic systems perform delicate surgical tasks, such as cutting and transplanting, which minimizes the human error and contamination risks that frequently lead to the failure of cultures for sensitive species.
- High-Throughput Phenotyping: AI is used to screen large numbers of regenerated plants for specific traits, providing rapid feedback that can be used to refine protocols for difficult species.
Case Study: Cannabis sativa
Cannabis sativa has long been considered recalcitrant to in vitro manipulation. Modern AI and machine learning are being used to optimize its germination and growth indices, which has saved resources like water and pesticides while maintaining soil fertility and improving productivity in a crop that was previously difficult to propagate reliably in a lab setting.
17. How do ANNs help in overcoming plant recalcitrance?
Artificial Neural Networks (ANNs), a subset of machine learning, help overcome plant recalcitrance—the failure of an explant to respond to in vitro manipulations—by transitioning tissue culture from an empirical “trial-and-error” approach to a data-driven predictive science.
According to the sources, ANNs provide several key advantages for handling difficult-to-propagate species:
1. Modeling Complex Regeneration Processes
ANNs are employed as efficient computational tools to interpret and predict outcomes for biological processes that are otherwise highly unpredictable.
- Predicting Somatic Embryogenesis: Researchers use algorithms like the Multilayer Perceptron (MLP) to model regeneration. For example, ANNs have been used to predict the success of somatic embryogenesis in chrysanthemum by analyzing the effects of different plant growth regulators (PGRs).
- Anther Culture and Callus Induction: In crops like tomato, ANNs (often combined with image processing) are used to model callus induction and regeneration in anther cultures, helping to identify the specific window where recalcitrant tissues become responsive.
2. Optimization of Media and Growth Conditions
Recalcitrant species often have very narrow and specific nutritional or hormonal requirements that are difficult to identify through traditional methods.
- Data-Driven Customization: New approaches use AI and ANNs to optimize culture medium composition, helping to find the most effective balance of macronutrients, micronutrients, and vitamins for specific genotypes.
- Interpreting Endogenous Hormone Levels: Recalcitrance is often linked to the internal (endogenous) hormone levels within an explant. ANNs can process complex phytochemical data from LC-MS/MS analysis to help researchers design suitable regeneration protocols that account for these internal variations.
3. Simulation and Prediction
ANNs allow scientists to simulate the growth of plants under various in vitro conditions, predicting responses before conducting costly and time-consuming physical experiments.
- Environmental Variables: These networks can simulate how a plant will react to different levels of light, temperature, and gaseous environments, which is critical for recalcitrant species that are sensitive to stress-induced abnormalities like hyperhydricity.
- Case Study (Cannabis sativa): Historically considered recalcitrant to in vitro manipulation, Cannabis production is being revolutionized by AI technology that improves real-time monitoring and optimizes germination and growth indices.
4. Integration with Automation and Robotics
The predictive power of ANNs is often used to drive robotic systems that handle delicate tasks like cutting and transplanting.
- Precision Handling: For recalcitrant tissues that are easily damaged by manual handling, AI-driven robots ensure consistent surgical precision and minimize the risk of human-introduced contamination, which is a major cause of failure in sensitive cultures.
- High-Throughput Phenotyping: AI is increasingly used for screening large numbers of regenerated plants for specific phenotypes, providing rapid feedback that can be used to further refine and optimize protocols for difficult species.
18. What are the main drawbacks of somaclonal variation?
Somaclonal variation, while occasionally useful for crop improvement, is primarily viewed as a significant limitation in plant tissue culture because it undermines the fundamental goal of producing genetically uniform, “true-to-type” clones.
The main drawbacks of somaclonal variation identified in the sources include:
1. Loss of Clonal Fidelity and Uniformity
The most critical drawback for the commercial micropropagation industry is the loss of genetic stability, which results in “off-types”—regenerated plants that do not match the elite characteristics of the mother plant. This instability can involve chromosomal aberrations, such as changes in ploidy level (aneuploidy or polyploidy), deletions, translocations, and inversions. These unintended changes make the production process unreliable for mass cloning.
2. Morphological and Physiological Abnormalities
Somaclonal variation often leads to a variety of undesirable structural defects:
- Floral and Fruit Defects: A prominent example is the “mantled” phenotype in oil palm, which causes feminization of male flower parts and can lead to fruit abortion and total loss of harvest.
- Structural Malformations: Plants may exhibit abnormalities such as pluricotyledony (multiple cotyledons), fused organs, stunted growth, bushiness, and tumorous outgrowths.
- Loss of Morphogenetic Potential: The frequency of variation generally increases with the number of subculture cycles and the age of the culture, eventually leading to a decline or total loss of the tissue’s ability to regenerate into whole plants.
3. Economic and Industrial Impact
The occurrence of these variations can have massive economic consequences.
- Yield Penalty: Deleterious variants often suffer from reduced yields or poor field performance.
- Scaling Restrictions: High frequencies of somaclonal variation have historically prevented the use of tissue culture for mass propagation in certain economically vital species, such as oil palm.
- Quality Assurance Costs: To ensure quality, laboratories must implement expensive and time-consuming DNA-based monitoring (such as RAPD or ISSR markers) to detect variants before plants are sold.
4. Challenges in Research and Breeding
- Instability of Traits: Epigenetic variations, which are non-heritable changes like altered DNA methylation, may appear as desirable traits in the lab but often disappear during sexual reproduction, making them useless for traditional breeding.
- Complications in Genetic Engineering: Somaclonal variation is highly frequent in transgenic material. These unintended mutations can confound the analysis of introduced genes, as it becomes difficult to determine whether a new phenotype is caused by the transgene or a culture-induced mutation.
- Chimerism: Mutations occurring in multicellular tissues can result in chimeric plants (a mix of mutated and normal cells), which are difficult to stabilize and often produce inconsistent progeny.
To mitigate these drawbacks, researchers often favor direct organogenesis or axillary bud proliferation, which bypass the unorganized callus phase that is most prone to generating these mutations.
19. How is CRISPR/Cas9 improving plant genetic transformation?
CRISPR/Cas9 is a revolutionary genome editing tool that provides unparalleled precision for modifying DNA sequences compared to earlier transformation techniques. While traditional Agrobacterium-mediated transformation often results in random transgene insertion at unpredictable sites, CRISPR allows for specific cutting, adding, or replacing of DNA at targeted genomic locations. This technology utilizes a short guide RNA (gRNA), typically 20 nucleotides long, to direct the Cas9 nuclease to a specific target sequence flanked by a protospacer adjacent motif (PAM). Once the Cas9 protein creates a double-stranded break (DSB) at the site, the cell’s natural repair mechanisms—nonhomologous end joining (NHEJ) or homology-directed repair (HDR)—introduce desired mutations, such as gene knockouts or precise insertions.
The CRISPR/Cas9 system offers several distinct advantages over previous methods:
- Simplicity and Cost: It is easier to design and more cost-effective than Zinc Finger Nucleases (ZFNs) or Transcription Activator-Like Effector Nucleases (TALENs) because target site selection is determined solely by base complementarity to the guide RNA rather than complex protein engineering.
- Multiplexing: It enables multiplex genome editing, where multiple alleles, homeologs in polyploids, or different members of a gene family can be targeted simultaneously.
- Transgene-Free Editing: It provides a pathway for transgene-free engineering; for instance, delivering Cas9/gRNA as ribonucleoprotein (RNP) complexes can produce edited mutants without any integration of foreign DNA into the host genome.
- Regulatory Benefits: Because it can create small genetic changes that do not involve foreign DNA, CRISPR-edited crops can sometimes bypass the stringent and costly regulatory procedures required for traditional genetically modified organisms (GMOs).
CRISPR/Cas9 has already been used to significantly improve various agronomic traits, including disease resistance (e.g., powdery mildew in wheat and bacterial blight in rice), abiotic stress tolerance (e.g., salt and drought tolerance in rice), and nutritional enhancement (e.g., increased β-carotene in bananas). Despite these advancements, challenges such as off-target cleavage and inconsistent editing efficiency across different species remain hurdles that researchers continue to address through new Cas9 variants and orthologs.
20. How can Artificial Neural Networks reduce trial-and-error in tissue culture?
Artificial Neural Networks (ANNs) reduce trial-and-error in plant tissue culture by transforming the field from an empirical, observation-based practice into a data-driven predictive science. Because the fundamental biological processes governing growth and differentiation are not yet fully understood, researchers traditionally rely on “empirical assessments based on trial and error” to find the right protocols. ANNs mitigate this by analyzing complex biological datasets to predict outcomes before physical experiments are conducted.
According to the sources, ANNs reduce trial-and-error through the following methods:
1. Optimization of Culture Media and Environmental Factors
The success of tissue culture depends on a meticulous balance of macro- and micronutrients, vitamins, and plant growth regulators (PGRs), which varies significantly between species and genotypes.
- Data-Driven Customization: AI algorithms, including ANNs, analyze extensive datasets to predict optimal growth outcomes for specific species. This allows researchers to dynamically adjust medium formulations—such as fine-tuning the nitrogen-to-phosphate ratio—without having to physically test every possible combination.
- Simulation of Conditions: ANNs can simulate plant growth under various in vitro conditions, such as different light intensities, temperatures, and pH levels. By anticipating how a tissue will respond to these variables, scientists can narrow down the most promising environments, saving time and resources.
2. Modeling Complex Developmental Pathways
Biological processes like somatic embryogenesis and organogenesis are highly unpredictable and often asynchronous.
- Predictive Modeling: Researchers use ANN algorithms like the Multilayer Perceptron (MLP) to design models for these regeneration processes. For example, these models have been used to predict the success of somatic embryogenesis in species like chrysanthemum.
- Anther Culture and Image Processing: In crops like tomato, ANNs are combined with image processing to model callus induction and regeneration in anther cultures. This provides a precise method for monitoring progress and predicting the exact conditions needed for successful plantlet formation.
3. Overcoming Recalcitrance
Many economically important plants are “recalcitrant,” meaning they fail to respond to standard in vitro manipulations.
- Interpreting Endogenous Signals: Success often depends on the plant’s internal (endogenous) hormone levels, which are difficult to measure and account for manually. ANNs act as efficient computational tools to interpret these unpredictable datasets, helping researchers design protocols that specifically address the unique physiological state of recalcitrant explants.
- Case Study (Cannabis sativa): Historically considered difficult to manipulate, Cannabis production is being revolutionized by ANNs that optimize germination and growth indices, reducing the excessive use of water and pesticides while maintaining soil fertility.
4. Integration with High-Throughput Phenotyping
ANNs are increasingly used alongside automated multi-spectral imaging systems for high-throughput phenotyping. This technology allows for the rapid screening of thousands of regenerated plants or seedlings for specific traits. The massive amounts of data generated from these screenings are fed back into the neural networks to further refine their predictive accuracy, creating a cycle that continually reduces the need for manual trial-and-error.
21. How do ANNs specifically optimize nutrient medium composition for plants?
Artificial Neural Networks (ANNs) optimize nutrient medium composition by shifting plant tissue culture from a traditional “trial-and-error” methodology toward a data-driven, predictive science. By analyzing extensive biological datasets, these algorithms can predict optimal growth outcomes and dynamically adjust culture protocols before physical experiments are even conducted.
Specific ways ANNs achieve this optimization include:
- Modeling Complex Interactions: Researchers utilize specific ANN structures, such as the Multilayer Perceptron (MLP), to interpret the intricate relationships between various medium components—like macronutrients, micronutrients, and plant growth regulators (PGRs)—and their cumulative effect on plant development.
- Customizing for Recalcitrant Species: In species that are traditionally difficult to manipulate, such as Cannabis sativa, ANNs have been used to optimize germination and growth indices, significantly improving productivity while reducing the waste of resources like water and pesticides.
- Predicting Specific Nutrient Requirements: Advanced algorithms can identify the precise nutritional needs of a genotype, such as using regression trees to predict minor nutrient requirements for hazelnut shoot cultures.
- Interpreting Endogenous Signals: ANNs serve as computational tools to interpret unpredictable datasets, such as endogenous (naturally occurring) hormone levels quantified by LC–MS/MS, allowing scientists to design media that account for the plant’s internal physiological state.
- Integration with Real-Time Monitoring: Some systems combine ANNs with image processing and automated multi-spectral imaging to monitor real-time progress (e.g., callus induction in tomato), providing constant data feedback to further refine and optimize the nutrient balance.
- Advanced Software Customization: Techniques like neuro-fuzzy logic and response surface methodology are employed to customize basal media formulations in cases where standard media, such as Murashige and Skoog (MS), are found to be inadequate for a particular species.
22. What role does deep learning play in automated image processing?
Deep learning, alongside machine learning, is a key area of artificial intelligence (AI) used to gather and analyze complex biological data in modern plant tissue culture. In the context of automated image processing, it plays a critical role in high-throughput phenotyping and real-time monitoring of developmental processes.
The sources identify the following specific roles of deep learning and AI-driven image processing:
1. High-Throughput Phenotyping
Deep learning algorithms are increasingly utilized for high-throughput phenotyping, which involves the rapid screening of large numbers of in vitro regenerated plants or germinated seedlings.
- Trait Identification: Using automated multi-spectral imaging systems (such as a ScanAlyzer), these technologies can identify specific transgenic or mutant phenotypes more efficiently than manual inspection.
- Data Refinement: The massive datasets generated from these imaging systems are fed back into neural networks to continually refine their predictive accuracy [Previous Context].
2. Modeling Developmental Stages
Automated image processing is combined with Artificial Neural Networks (ANNs) to model and predict intricate biological transitions that are difficult to standardize.
- Monitoring Callus and Regeneration: In crops like tomato, image processing is used to model callus induction and regeneration in anther cultures. This allows researchers to pinpoint the exact windows of time when tissues are most responsive to treatments [Previous Context].
- Somatic Embryogenesis: AI models analyze visual data to track the progress of somatic embryos through various stages (globular, heart, torpedo) to ensure successful plantlet formation [Previous Context].
3. Real-Time Industry Monitoring
In industries such as Cannabis sativa production, deep learning technology is essential for enhancing operational oversight.
- Real-Time Assessment: AI improves real-time monitoring of crop health, which informs better strategies for harvesting, processing, and marketing.
- Resource Optimization: By accurately analyzing images of plant growth, these systems help optimize the use of water, pesticides, and fertilizers, thereby maintaining soil fertility and reducing waste.
4. Integration with Robotics
The visual data processed by deep learning models often drives robotic systems that perform delicate surgical tasks.
- Precision Handling: AI-driven image processing allows robots to identify precise locations for cutting and transplanting tissue cultures.
- Error Reduction: By providing a consistent “eye” for automated systems, deep learning reduces the risk of human-introduced contamination and ensures uniform handling of explants across large-scale operations.
23. What is the role of chemostat bioreactors in simulation?
Chemostat bioreactors are devices specifically built for large-scale continuous culturing in plant tissue culture. While the sources do not explicitly use the term “simulation” in the context of chemostats, they describe several related roles for bioreactors in modeling, automation, and synthesizing outcomes.
Primary Roles of Chemostat and Continuous Bioreactors
- Large-Scale Continuous Culturing: Chemostat bioreactors are designed to maintain a culture in a continuous state by constantly adding fresh nutrient medium and discarding surplus cell suspensions. This allows for a steady state of growth and production.
- Secondary Metabolite Synthesis: These systems are a viable source for the synthesis of important secondary metabolites. This is increasingly effective due to breakthroughs in molecular biology, enzymology, and fermentation technology.
- Modeling Culture Dynamics: Bioreactor studies involve the analysis and modeling of culture dynamics, which includes integrating biosynthesis with product release and separation. They also facilitate the study of flow, mixing, and mass transfer between different phases to optimize bioreactor design.
Role within Automated Systems
Bioreactors function as automated culture systems whose main purpose is to provide a meticulously controlled environment for optimal cell growth.
- Controlled Environment: They provide more precise control over factors such as gaseous exchange, illumination, temperature, and pH compared to conventional semi-solid culture vessels.
- Automation and Predictability: As discussed in our earlier conversation regarding Artificial Neural Networks (ANNs), AI-driven simulations can predict optimal outcomes which then drive these automated systems. This integration allows for the installation of programs that automatically control growth and development by dynamically adjusting parameters like CO2 concentration and medium pH.
- Economic Simulation: Computerization has enabled scientists to use these systems to economically simulate probable outcomes and automate processes like microinjection.
In summary, chemostat bioreactors act as the physical platform for continuous large-scale production, while their automated nature allows for the real-time application of models and simulations to ensure reproducible and efficient plant growth.
24. What is the importance of endogenous vs exogenous hormone levels?
The relationship between endogenous (naturally occurring within the plant) and exogenous (added to the culture medium) hormone levels is a critical factor in plant tissue culture, as the interaction between these two sources ultimately determines the morphogenetic fate of the cultured tissue.
The Primacy of Endogenous Levels
While researchers manipulate growth by adding exogenous regulators, it is the final endogenous concentration within the tissue—not the amount in the medium—that is the decisive factor in determining organogenic differentiation.
- Regulators vs. Modulators: Some researchers postulate that plant morphogenesis is regulated exclusively by endogenous hormones, meaning that exogenous additions serve primarily as modulators of these internal signals.
- Self-Sufficiency: Some plant species or specific tissues already possess sufficiently high levels of endogenous hormones. In these cases, the supply of high levels of exogenous growth regulators is not required for regeneration and may even be unnecessary.
- Androgenesis: The physiological state of a donor plant determines the internal hormone levels of tissues like anthers, which directly affects their regeneration ability and response to treatments.
Interaction and Balance
The success of a tissue culture protocol often depends on achieving a precise balance between internal and external signals.
- Dynamic Response: Soon after an explant is isolated (wounding), an endogenous hormonal system is induced. However, this system is often insufficient to promote significant growth on its own and must be supplemented by exogenous regulators from the medium until the tissue can produce enough meristematic cells to sustain itself.
- Feedback Loops: The exogenous addition of hormones can actively change the levels of internal ones. For instance, adding exogenous auxin can regulate somatic embryo development by altering the levels of endogenous auxins like IAA. Similarly, auxin-induced organogenesis is often accompanied by the production of endogenous cytokinins.
- Specific Ratios: Successful induction, maturation, and germination (particularly in somatic embryogenesis) require a specific ratio between both internal and external auxin, cytokinin, ABA, and gibberellic acid.
Explaining Genotypic Variation and Recalcitrance
The importance of this relationship is most evident when different genotypes respond differently to the exact same culture medium.
- Genotypic Differences: Variations in how different varieties of a species (such as maize or rice) respond to a protocol are often attributed to their varying endogenous levels of plant growth regulators.
- Overcoming Recalcitrance: Species that are difficult to culture (“recalcitrant”) often have unique internal hormonal architectures. Characterizing these endogenous levels using advanced technologies like LC-MS/MS allows researchers to design customized exogenous protocols that account for the plant’s unique internal state.
Risks of Imbalance
An improper balance between these two levels can lead to physiological failures:
- Hyperhydricity: High concentrations of exogenous regulators in the medium can stimulate developmental anomalies, such as hyperhydricity, by creating an internal hormonal imbalance, specifically through the over-accumulation of internal cytokinins or the enhancement of ethylene biosynthesis.
- Inhibited Development: While exogenous auxin is often needed for induction, its continued presence can eventually inhibit the further development of somatic embryos, necessitating a move to an auxin-free medium to allow the internal program to proceed.
25. What role does bacterial cellulose play in plant tissue regeneration?
A recent and significant development in plant biotechnology has identified bacterial cellulose (BC) as a highly effective mediator for plant tissue regeneration and wound healing.
According to the sources, the role of bacterial cellulose in this process is defined by the following characteristics and mechanisms:
- Inducing Rapid Regeneration: In studies using model plants like Nicotiana benthamiana and Arabidopsis thaliana, researchers found that applying BC patches to wounded leaves induced the formation of new cells on both sides of a cut within just two days. Complete wound closure was achieved after only seven days.
- Specific Healing Properties: This regenerative effect is unique to bacterial cellulose; structurally similar matrixes, such as plant cellulose, do not promote the same healing process. This indicates that BC provides specific biological or physical cues that go beyond simply preventing tissue dehydration or facilitating physical contact between cells.
- Regulation of Molecular Networks: The molecular mechanism behind this phenomenon involves the regulation of cytokinin and defense networks. Cytokinins are critical plant hormones known to stimulate cell division and shoot formation, and the BC patches appear to interact with these signaling pathways to trigger the regenerative response.
- Biocompatibility: Bacterial cellulose is synthesized by certain bacteria as a biofilm consisting of highly pure cellulose fibers. While it has been long used in human biomedical applications due to its high biocompatibility, its application as a “healing patch” for plants represents a new frontier in plant tissue culture and protection.
26. What is the difference between direct and indirect organogenesis?
Organogenesis is the process of inducing the formation of plant organs, such as roots, shoots, and leaves, from cultured tissues by manipulating hormonal balance and environmental conditions. The fundamental difference between direct and indirect organogenesis is the presence or absence of an intervening, unorganized cell mass known as callus.
Direct Organogenesis
- Origin: Plant organs originate directly from the explant tissue (the starting sample) with no intervening callus stage.
- Mechanism: This pathway typically involves the proliferation of preexisting meristems, such as shoot tips and axillary buds, or the de novo induction of adventitious meristems on organized tissues like leaves or stems.
- Genetic Stability: A major benefit of the direct route is its high genetic fidelity, as it is significantly less prone to somaclonal variation (mutations) than callus-mediated methods.
- Commercial Use: It is the preferred method for commercial mass propagation where the goal is to produce “true-to-type” clones of elite plant varieties.
- Developmental Order: The progression follows the sequence: Primary Explant → Meristemoid → Organ Primordium.
Indirect Organogenesis
- Origin: The explant cells first undergo dedifferentiation to form an unorganized mass of cells called callus.
- Mechanism: From this callus, new organs are formed through a subsequent process of redifferentiation triggered by specific concentrations of plant growth regulators in the medium.
- Genetic Stability: The intervening callus phase carries a high risk of somaclonal variation, which can result in undesirable genetic diversity or morphological abnormalities in the regenerated plants.
- Specialized Use: Despite the risk of variation, indirect organogenesis is critical for genetic transformation and transgenic plant production, as it allows for the transformation of cells within the callus followed by the regeneration of modified plants.
- Developmental Order: The progression follows the sequence: Primary Explant → Callus → Meristemoid → Organ Primordium.
Key Commonalities
Both forms of organogenesis result in unipolar structures—meaning they form either a shoot or a root independently—that develop procambial strands to establish a vascular connection with the pre-existing tissue. Additionally, both pathways are primarily regulated by the auxin-to-cytokinin ratio in the nutrient medium: a high ratio of cytokinin to auxin promotes shoot formation, while a high ratio of auxin to cytokinin promotes root regeneration.
27. What are the specific challenges in scaling CRISPR for agriculture?
While CRISPR/Cas9 is a revolutionary tool for genome editing, scaling its application for broad agricultural use involves significant technical, environmental, and regulatory challenges.
According to the sources, the specific challenges in scaling CRISPR for agriculture include:
1. Plant Recalcitrance and Regeneration Hurdles
The most fundamental bottleneck is that many economically important crop species and specific genotypes are recalcitrant, meaning they are resistant to in vitro regeneration and transformation.
- Genotype-Specific Responses: Protocols that work for one cultivar often fail for another, making it difficult to create universal scaling methods.
- Regeneration Challenges: Success depends on the ability to regenerate a whole fertile plant from a single edited cell, a process that remains difficult for many monocots, woody plants, and legumes.
2. Delivery and Editing Efficiency
Efficiently delivering editing components into plant cells without causing permanent damage is a major technical hurdle.
- Delivery Limitations: Common delivery methods like Agrobacterium-mediated transformation and biolistics (gene gun) often result in random DNA integration or physical tissue damage, which can impede healthy plant development.
- Low Efficiency in Polyploids: Many major crops are polyploid (having multiple sets of chromosomes). Scaling CRISPR for these crops is challenging due to the low delivery capacity and editing efficiency required to target all homeologs simultaneously.
3. Technical Precision and Off-Target Effects
Scaling requires high predictability, yet CRISPR can sometimes produce unintended consequences.
- Off-Target Mutations: The risk of off-target cleavage—where the Cas9 enzyme cuts DNA at sites other than the intended target—can lead to undesirable genetic alterations.
- PAM Requirements: CRISPR requires a specific Protospacer Adjacent Motif (PAM) sequence to function, which limits the available target sites within a genome.
4. Scalability and Reproducibility Across Environments
Agricultural products must perform reliably across diverse fields, yet gene-editing outcomes are often inconsistent.
- Environmental Variability: Gene-editing outcomes are influenced by environmental factors, leading to inconsistent phenotypic expressions when crops are moved from the lab to diverse agricultural settings.
- Lack of Uniformity: Achieving uniform genetic modifications at a massive scale is difficult due to the genetic diversity among different crop species.
5. Regulatory, Legal, and Social Barriers
Non-technical factors significantly impact the speed and cost of scaling CRISPR technology.
- Fragmented Global Regulation: Regulatory frameworks vary drastically by region. For example, while the USDA may allow some CRISPR-edited crops to bypass strict GMO regulations, the European Union generally regulates them as traditional GMOs. This complicates international trade and adoption.
- Intellectual Property Disputes: Ongoing legal battles over CRISPR patents and IP rights can limit the development and accessibility of the technology for smaller agricultural firms.
- Public Skepticism: Widespread public skepticism and ethical concerns regarding genetically modified or edited foods remain a major barrier to commercial adoption and market trust.
28. What are the common limitations of plant tissue culture?
While plant tissue culture is a powerful biotechnological tool, its widespread adoption and commercial application are hindered by several significant limitations. These challenges range from high economic costs to complex biological and technical hurdles.
1. High Costs and Resource Requirements
The establishment and maintenance of plant tissue culture facilities require substantial financial investment in infrastructure, specialized equipment (such as autoclaves and laminar airflow hoods), and consumables.
- Energy and Labor: The technology is highly energy-intensive, requiring continuous electricity for lighting, air conditioning, and sterilization, which can account for up to 60% of production costs in some regions. Additionally, the process is extremely labor-intensive, as it relies on skilled personnel to perform delicate manual tasks like explant excision and subculturing.
- Market Viability: The unit cost per plant can often be non-competitive with traditionally propagated materials, making it economically unfeasible for some crops unless propagation costs are significantly reduced.
2. Genetic and Epigenetic Instability
A primary goal of tissue culture is to produce uniform, “true-to-type” clones; however, the process frequently induces somaclonal variation.
- Genetic Changes: These are stable mutations such as chromosomal rearrangements, deletions, and changes in ploidy levels (aneuploidy or polyploidy).
- Epigenetic Changes: These involve non-heritable alterations, such as changes in DNA methylation, which can lead to phenotypic abnormalities that may disappear during sexual reproduction. Such instability can result in “off-type” plants with reduced yields or structural defects, such as the “mantled” flower abnormality in oil palm.
3. Biological Recalcitrance and Genotype Specificity
Many economically vital plant species, particularly woody perennials, monocots, and legumes, are considered recalcitrant, meaning they are highly resistant to in vitro manipulation.
- Inconsistent Protocols: There is no universal protocol for tissue culture; requirements for media composition, hormonal balance, and environmental conditions vary significantly between different species and even among individual cultivars of the same species.
- Age and Seasonality: Tissues from mature trees often fail to respond in culture, necessitating a long “rejuvenation” period or the use of limited juvenile seedling sources.
4. Microbial Contamination and Browning
Maintaining an aseptic environment is a constant challenge.
- Latent Pathogens: Explants may harbor internal or endophytic microorganisms that surface sterilization cannot eliminate, leading to the sudden loss of established cultures.
- Phenolic Exudation: Many species, especially trees, release phenolic compounds into the medium upon wounding. These compounds oxidize into toxic quinones, causing browning or blackening of the tissue and medium, which often leads to the death of the explant.
5. Physiological Disorders
The artificial environment of a culture vessel can lead to developmental anomalies, most notably hyperhydricity (also known as vitrification or glassiness).
- Symptoms: Hyperhydric shoots appear translucent, watery, and fragile due to excess fluid in intercellular spaces and a lack of proper cell wall lignification.
- Causes: This disorder is often triggered by high humidity, gas accumulation (like ethylene and CO2) within unventilated vessels, or high concentrations of cytokinins and ammonium ions in the medium.
6. Acclimatization and Field Survival
The transition from a controlled lab environment to the field is a major bottleneck.
- Structural Weakness: In vitro-raised plantlets often have malfunctioning stomata and a poorly developed epicuticular wax layer, making them highly susceptible to rapid water loss and desiccation when moved to lower humidity.
- Weak Systems: These plants may also have inefficient photosynthetic apparatus and weak, non-functional root systems that fail to adapt quickly to soil conditions, resulting in high mortality rates during hardening.
29. How does somatic embryogenesis allow clones to be true to type?
Somatic embryogenesis (SE) allows clones to be true-to-type primarily because it is an asexual reproduction process that originates from a single somatic cell, thereby maintaining the exact genetic makeup of the mother plant.
The mechanisms that ensure this genetic fidelity include:
1. Unicellular Origin
The most critical factor in maintaining genetic uniformity is that somatic embryos generally develop from a single competent somatic cell. Because the entire plant regenerates from one cell, it ensures that the resulting clone is not a chimera (a mixture of genetically different tissues). This single-cell origin is the foundation for producing large quantities of genetically identical individuals.
2. Bypassing Sexual Recombination
Unlike zygotic embryogenesis, which involves meiosis and fertilization, somatic embryogenesis bypasses these processes entirely. Sexual reproduction shuffles genetic material, leading to variation in the offspring. SE avoids this “shuffling,” allowing the genetic background of an elite genotype to remain intact and conserved. This makes SE an ideal tool for the mass propagation of plants that are highly heterozygous and would not produce true-to-type offspring from seeds.
3. High Cytogenetic Stability in Direct SE
The degree of “true-to-type” fidelity often depends on the mode of SE used:
- Direct Somatic Embryogenesis: In this pathway, embryos form directly from the explant without an intervening callus phase. This method is preferred for cloning because it exhibits higher cytogenetic stability and is less prone to mutations.
- Indirect Somatic Embryogenesis: This involves a callus phase (an unorganized mass of cells), which is a known source of somaclonal variation. Prolonged culture in a callus state can lead to chromosomal aberrations, DNA methylation changes, and point mutations, resulting in “off-types” that are no longer true to the mother plant.
4. Efficient Developmental Structure
Somatic embryos are bipolar structures, possessing both shoot and root meristems simultaneously. Because they can germinate directly into whole plantlets without requiring separate induction steps for roots and shoots (as required in organogenesis), they undergo fewer hormonal manipulations, which can reduce the stress-induced genetic instability associated with multiple culture stages.
5. Verification of Clonal Fidelity
To guarantee that regenerated plants are authentic replicas, laboratories utilize clonal fidelity testing. Molecular markers such as RAPD (Random Amplified Polymorphic DNA) and ISSR (Inter Simple Sequence Repeats) are used to compare the DNA profiles of the clones against the mother plant to confirm they are 100% true-to-type.
30. What causes the undesirable somaclonal variation in tissue culture?
Somaclonal variation refers to the phenotypic and genotypic variability that arises in plants regenerated from cell and tissue culture. While it can occasionally be a useful source of genetic diversity for crop improvement, it is generally considered an undesirable limitation in commercial micropropagation because it undermines the goal of producing genetically uniform, “true-to-type” clones.
The causes of undesirable somaclonal variation are complex and stem from a combination of biological, chemical, and environmental factors:
1. The “Genomic Shock” of Explant Isolation
The very act of initiating a culture is a stress-response mechanism. Removing cells from their natural environment and placing them in artificial in vitro surroundings is described as a “traumatic experience” or “genomic shock”. This trauma can disrupt normal cellular controls that regulate the genome, leading to abnormal reprogramming or genomic restructuring.
2. The Intervening Callus Phase
Regeneration protocols that involve an unorganized mass of cells known as callus are the most significant source of variation.
- Mutagenic Environment: The tissue culture process involving an undifferentiated callus phase is inherently mutagenic.
- Cellular Origin: Plants regenerated from callus often have a multicellular origin, which can result in the formation of chimeric plants (a mix of genetically different cells).
- Loss of Control: In callus, the absence of organized tissue structure leads to a breakdown of the finely tuned systems of checks and balances that normally maintain genomic stability.
3. Plant Growth Regulators (PGRs)
The type and concentration of hormones used to trigger growth are primary drivers of instability:
- Synthetic Auxins (2,4-D): High concentrations and prolonged exposure to synthetic auxins, particularly 2,4-D, are highly correlated with DNA mutations and abnormal development. 2,4-D is known to dramatically elevate DNA methylation and can selectively favor the division of polyploid cells.
- Cytokinins: Excess or continuous use of cytokinins, such as BAP, increases the risk of variation and can lead to physiological disorders like hyperhydricity.
- Hormonal Imbalance: High concentrations of growth regulators in the medium can trigger cell-cycle disturbances and lead to chromosomal aberrations.
4. Culture Duration and Subculturing
The frequency and length of time a tissue remains in culture are critical factors:
- Mutation Accumulation: The frequency of variation increases with the length of culture duration and the number of subculture cycles.
- Cell Cycle Transitions: Mutations may accumulate with each mitotic cycle; the more divisions a cell line undergoes in vitro, the more likely it is to exhibit mutations.
5. Pre-existing Variability in the Donor Plant
Variation is not always induced in vitro; it can sometimes be traced back to the source material:
- Polysomaty: In many plants, normal development involves somatic cells that naturally have higher ploidy levels (endoreduplication) than the meristematic cells. If these specialized cells are forced to divide during culture, they produce polyploid offspring.
- Genetic Chimeras: Many donor plants are already chimeras; tissue culture can dissociate these mixed cell layers, resulting in regenerated plants that do not match the parent phenotype.
6. Specific Genetic and Epigenetic Mechanisms
The undesirable variations are expressed through several molecular changes:
- Chromosomal Aberrations: These include changes in chromosome number (polyploidy and aneuploidy) and structure (deletions, inversions, translocations, and duplications).
- Epigenetic Alterations: These involve non-heritable changes, such as DNA methylation and chromatin modification, which cause phenotypic “off-types” that may disappear in future generations.
- DNA Sequence Changes: Activation of transposable elements (jumping genes), gene amplification, and point mutations can permanently alter the genetic makeup of the plant.
31. What are the specific risks of gene flow in GMOs?
The gene flow of Genetically Modified Organisms (GMOs) presents significant biological and ecological risks, primarily centered on the unintended spread of transgenic traits to wild or weedy relatives. These risks can be categorized by their impact on ecosystems, agriculture, and potential horizontal transfer to other organisms.
Ecological and Biodiversity Risks
- Increased Invasiveness: The transfer of transgenic traits to wild populations can increase the invasiveness of these relatives, potentially disrupting local ecosystems.
- Displacement of Native Species: Gene flow can alter natural competition dynamics, leading to the displacement of native species and a subsequent reduction in biodiversity.
- Genetic Homogenization: The persistence of GM traits in the environment can cause genetic homogenization, which diminishes the diversity and resilience of natural ecosystems. This makes these environments less adaptable to environmental changes and new pest pressures.
Agricultural and Weed Management Risks
- Evolution of Resistant Weeds: A major concern is that gene flow from herbicide-resistant crops will lead to the evolution of resistant weeds.
- Increased Chemical Application: The emergence of these “superweeds” often necessitates the use of higher herbicide volumes or more toxic alternatives, which further impacts non-target species and reduces agricultural biodiversity.
- Yield Penalty and Metabolic Burden: Unintended genetic integration or rearrangements in the field can sometimes lead to a yield penalty or metabolic burden on the crops themselves.
Horizontal Gene Transfer (HGT)
- Residual Bacterial Transfer: In plants transformed via Agrobacterium, there is a risk that residual bacteria may survive and remain within the regenerated plants. This could potentially allow the transfer of genes to other bacteria or fungi in the environment, especially in plants that are propagated vegetatively.
- Antibiotic Resistance: There are concerns that antibiotic resistance marker genes used during the transformation process could be transferred horizontally to the gut flora of humans or animals. While many experts consider this occurrence highly unlikely, the potential for such transfer remains a significant point of regulatory and public concern.
Pollen-Mediated Risk
- Transgenic Outcrossing: A primary mechanism for gene escape is through pollen-mediated dispersal to sexually compatible wild relatives.
- GM Trees: This risk is particularly acute for forest trees due to their long lifespans and potential for wide-range outcrossing. Researchers are developing containment strategies, such as plastid transformation (which is maternally inherited) or engineering flower sterility, to eliminate these pollen-mediated escape routes.
32. How do researchers mitigate off-target effects in CRISPR?
Researchers mitigate off-target effects—unintended DNA cleavage at sites other than the desired target—through a variety of bioinformatic, enzymatic, and procedural strategies. Because the guide RNA (gRNA) can sometimes tolerate mismatches to the DNA target, several safeguards are employed to ensure genomic stability.
1. Bioinformatic and Computational Selection
The first step in mitigation is the careful design of the single guide RNA (sgRNA) using specialized software.
- Target Selection Tools: Researchers use web tools like CRISPR-P 2.0 to analyze the plant genome and select guide sequences with high scores, which suggest a low risk of off-target activity.
- Sequence Filtering: To reduce unintended knockdowns, researchers exclude any gRNAs that share more than 16 nucleotides of similarity with other areas of the plant genome.
2. Enzyme Engineering and Variants
Modifying the nuclease itself can significantly improve its specificity.
- Nickases (nSpCas9): Researchers utilize modified Cas9 variants called nickases, which only cut one strand of the DNA, thereby reducing the likelihood of permanent off-target mutations compared to traditional double-strand breaks.
- Cas9 Orthologs: Using different versions of the Cas9 protein from various bacterial species can enhance precision. For example, NmCas9 from Neisseria meningitidis has a more stringent PAM sequence (5′-NNNNGATT-3′), which limits its potential target sites and reduces the chance of accidental binding. Other orthologs like SaCas9 and CjCas9 recognize different and often longer sequences to improve specificity.
3. Advanced Genome Editing Technologies
Newer generations of genome editing tools are designed to be inherently more precise than the original CRISPR/Cas9 system.
- Prime Editing: This technology allows for accurate insertions, deletions, and base conversions without inducing double-strand breaks, which significantly minimizes off-target mutations in plants like rice and Arabidopsis.
- Base Editing: By using a “dead” Cas9 (dCas9) fused to a deaminase enzyme, researchers can achieve single-base alterations without cutting the DNA backbone, thereby expanding the potential for safe, multiplex genetic changes.
4. Delivery Methods and Post-Transformation Handling
The method of introducing CRISPR components into the cell also impacts off-target frequency.
- Ribonucleoprotein (RNP) Complexes: Instead of delivering DNA that continuously produces the nuclease, researchers can deliver pre-assembled Cas9/gRNA RNP complexes. This allows for transient editing where the enzyme is active only briefly before being degraded, which drastically reduces the window of time for off-target events to occur.
- Outcrossing: Once edited plants are produced, researchers can perform outcrossing with wild-type plants. This process allows them to segregate and remove any unintended off-target mutations from the genetic line while retaining the desired edit.
33. How do ML and DL algorithms predict regeneration success?
Machine Learning (ML) and Deep Learning (DL) algorithms predict regeneration success by transitioning tissue culture from an empirical, observation-based practice into a data-driven predictive science. These algorithms analyze complex biological datasets to model intricate processes like somatic embryogenesis and organogenesis, allowing researchers to anticipate outcomes before physical experiments are conducted.
1. Modeling Complex Biological Transitions
Algorithms are used to design models for developmental stages that are otherwise difficult to standardize or predict.
- Artificial Neural Networks (ANNs): Specifically, the Multilayer Perceptron (MLP) algorithm is used to model and predict the success of somatic embryogenesis. For example, research on chrysanthemum compared MLP and Support Vector Regression (SVR) to accurately predict how different plant growth regulators (PGRs) would influence embryo formation.
- Anther Culture and Callus Induction: In crops like tomato, ANNs are combined with image processing to model callus induction and regeneration in anther cultures. This allows for the identification of specific time windows when tissues are most responsive to treatment.
- Regression Trees: These algorithms have been applied to predict specific requirements, such as identifying the minor nutrient needs of hazelnut shoot cultures.
2. Optimization of Media and Environment
Predictive algorithms reduce trial-and-error by simulating how different chemical and physical factors interact to affect growth.
- Nutrient Formulation: Techniques like neuro-fuzzy logic are employed to customize basal media formulations for specific genotypes, particularly when standard media like MS are inadequate.
- Growth Simulations: ML and DL can simulate plant growth under various in vitro conditions, such as different light intensities, temperatures, and gaseous environments.
- Interpreting Endogenous Signals: Algorithms act as computational tools to interpret unpredictable datasets, such as the acquisition of extremely low endogenous PGR concentrations quantified by LC–MS/MS. This helps design customized regeneration protocols that account for the plant’s internal physiological state.
3. Automated Image Processing and High-Throughput Phenotyping
Deep Learning plays a critical role in gathering and analyzing visual data to monitor regeneration progress in real-time.
- Trait Identification: Automated multi-spectral imaging systems use DL algorithms to screen large numbers of regenerated plants or germinated seedlings for specific transgenic or mutant phenotypes.
- Robotic Integration: These predictive models drive robotic systems that perform delicate surgical tasks, such as cutting and transplanting, ensuring consistent handling and minimizing human error or contamination—factors that often determine the ultimate success of a culture.
4. Overcoming Recalcitrance
Algorithms are particularly effective at identifying success patterns in species traditionally considered recalcitrant (resistant to in vitro manipulation).
- Case Study (Cannabis sativa): ML algorithms and ANNs have been used to optimize germination and growth indices for industrial hemp, identifying the precise conditions needed for successful propagation in a crop that has historically been difficult to manipulate in a laboratory setting.
- Data Aggregation: AI helps businesses and researchers gather and analyze vast datasets on crop health, saving resources like water and pesticides while maintaining soil fertility and improving productivity.
34. What makes certain plant species recalcitrant to tissue culture?
Recalcitrance in plant tissue culture is defined as the inability of a plant cell or tissue to respond to in vitro manipulations, often resulting in a failure to divide, dedifferentiate, or regenerate into a whole plant. This phenomenon is driven by a complex interplay of genetic, physiological, and molecular factors.
1. Genetic and Genotypic Factors
The most significant factor contributing to recalcitrance is the nature and type of the donor plant itself.
- Species and Genotype Specificity: Large differences in responsiveness exist among plant genera, species, and even individual cultivars. For example, while some cultivars of a species may regenerate easily, others remain completely non-responsive to the same media and conditions.
- Taxonomic Groups: Generally, monocotyledons (such as cereals and grasses) are more recalcitrant than dicotyledons. Similarly, woody perennials, legumes, and gymnosperms often present significant challenges compared to herbaceous species.
2. Physiological Age and Juvenility
The developmental status of the donor plant significantly influences its performance in vitro.
- Loss of Juvenility: As plants age, their tissues often pass through the juvenile phase and enter a mature state where they lose the “competence” to respond to hormonal signals. Mature woody plants are notoriously difficult to establish in culture, often requiring a long period of “rejuvenation” through sequential subculturing to regain morphogenic potential.
- Explant Choice: The most successful cultures are typically initiated from juvenile or undifferentiated tissues, such as immature embryos, shoot tips, or meristems, which contain a higher proportion of actively dividing cells.
3. Endogenous Hormone Imbalance
Success in tissue culture depends on achieving a balance between endogenous (naturally occurring) and exogenous (supplied in the medium) plant growth regulators (PGRs).
- Internal Signaling Interference: Recalcitrant genotypes often have unique internal hormonal architectures that interfere with standard exogenous treatments. For instance, variations in internal auxin or cytokinin levels can determine whether a tissue will regenerate or remain unresponsive.
- Seasonality: The physiological state of the donor plant changes with the seasons, affecting its internal PGR levels and metabolic status. Explants collected during specific seasons, such as spring flushes, are often more responsive than those collected during dormant periods.
4. Phenolic Exudation and Browning
Many recalcitrant species, particularly woody plants, release phenolic compounds into the medium upon being wounded during explant isolation.
- Oxidative Stress: These phenolics oxidize into toxic quinones, which cause browning or blackening of the tissue and the culture medium. This leads to the death (necrosis) of the explant before it can begin the regeneration process.
5. Molecular and Epigenetic Barriers
Recalcitrance is increasingly understood through a molecular lens, involving gene regulation and epigenetic blocks.
- Signal Transduction Failure: Research suggests that recalcitrance may involve a breakdown in signaling cascades, such as the cytokinin signal transduction cascade, which is necessary to induce shoot regeneration.
- Key Regulatory Genes: The failure to activate essential “master regulator” genes like WUSCHEL (WUS) or BABY BOOM (BBM) can prevent cells from acquiring the necessary totipotency.
- Epigenetic Regulation: The “genomic shock” of removing a cell from its natural environment can lead to abnormal reprogramming. DNA methylation and histone modification patterns in recalcitrant tissues may block the activation of developmental pathways, essentially “locking” the cells in their current state.
6. Physiological Disorders
The artificial in vitro environment itself can induce stress that leads to recalcitrance-related defects.
- Hyperhydricity (Vitrification): High humidity and gas accumulation (like ethylene) in closed vessels can cause shoots to become translucent, watery, and fragile, significantly reducing their ability to grow and adapt.
- Free Radicals: Recalcitrance has also been linked to the generation of free radicals and a poor antioxidant status in the culture, which inhibits healthy cell division.
35. How is CRISPR/Cas9 superior to earlier genetic engineering methods?
CRISPR/Cas9 is considered superior to earlier genetic engineering and genome editing methods due to its unparalleled precision, simplicity in design, and capacity for multiplexing. Unlike traditional methods that often result in unpredictable genetic outcomes, CRISPR provides a programmable and highly efficient toolkit for crop improvement.
Its superiority is defined by the following key advancements:
1. Precision vs. Random Insertion
Earlier transformation methods, such as Agrobacterium-mediated transformation and biolistics (gene gun), primarily rely on the random integration of transgenes into the host genome. This can lead to undesirable effects, such as the disruption of endogenous genes or variable gene expression due to the “position effect”. In contrast, CRISPR/Cas9 uses a guide RNA (gRNA) to direct the Cas9 nuclease to a specific genomic location, allowing for precise deletions, insertions, or modifications.
2. Simplicity and Cost-Effectiveness
Compared to previous site-specific nucleases like Zinc Finger Nucleases (ZFNs) and Transcription Activator-Like Effector Nucleases (TALENs), CRISPR is significantly easier and cheaper to design.
- ZFNs and TALENs: These require complex and laborious protein engineering to create a unique DNA-binding domain for every new target site.
- CRISPR/Cas9: Target specificity is determined solely by the base complementarity of a short 20-nucleotide guide RNA. This eliminates the need for expensive protein re-engineering for different genes.
3. Multiplexing Capabilities
CRISPR/Cas9 enables multiplex genome editing, where researchers can target multiple alleles, homeologs in polyploid crops (like wheat), or different members of a gene family simultaneously. This is far more efficient than earlier methods that typically required sequential rounds of transformation or crossing to combine multiple traits.
4. Gene Knockout vs. Knockdown
Before CRISPR, RNA interference (RNAi) was the standard for suppressing gene expression. However, RNAi has several limitations:
- Incomplete Silencing: RNAi often results in variable “knockdown” rather than a total “knockout,” leaving behind some transcripts that may still influence the plant’s phenotype.
- Instability: Silencing efficiency can be inconsistent and may decline in successive generations.
- Permanent Modification: CRISPR creates permanent and irreversible mutations by inducing double-stranded breaks that the cell repairs through error-prone mechanisms, leading to stable knockouts.
5. Transgene-Free “Cisgenic” Editing
A major advantage of CRISPR is the ability to produce transgene-free edited plants. Traditional genetic engineering results in Genetically Modified Organisms (GMOs) that contain foreign DNA, which face stringent and costly regulatory hurdles. CRISPR can create small, targeted changes—such as single-base edits—without the integration of foreign DNA into the host genome. Because these changes are virtually indistinguishable from natural mutations, they can sometimes bypass the strict regulations applied to traditional GMOs in certain jurisdictions, such as the United States.
References
Bhoite, H. A., & Palshikar, G. S. (2014). Plant Tissue Culture: A Review. World Journal of Pharmaceutical Sciences, 2(6), 565–572. http://www.wjpsonline.org/
Espinosa-Leal, C. A., Puente-Garza, C. A., & García-Lara, S. (2018). In vitro plant tissue culture: means for production of biological active compounds. In Planta (Vol. 248, Number 1). Springer Verlag. https://doi.org/10.1007/s00425-018-2910-1
Gaikwad, A. V, Singh, S. K., & Gilhotra, R. (2017). PLANT TISSUE CULTURE-A REVIEW. In Review Article Journal of Pharmaceutical Research & Education Journal homepage (Vol. 2, Number 1). http://www.gyanvihar.org/researchjournals/
Hussain, A., Ahmed, I., Nazir, H., & Ullah, I. (2012). Plant Tissue Culture: Current Status and Opportunities. In Recent Advances in Plant in vitro Culture. InTech. https://doi.org/10.5772/50568
Loyola-Vargas, V. M., & Ochoa-Alejo, N. (2018). An Introduction to Plant Tissue Culture: Advances and Perspectives. In Methods in Molecular Biology (Vol. 1815, pp. 3–13). Humana Press Inc. https://doi.org/10.1007/978-1-4939-8594-4_1
Negi, S., Singh, P., Trivedi, V. L., Rawat, J. M., & Semwal, P. (2024). The current trends and research progress globally in the plant tissue culture: 90 years of investigation. Plant Cell, Tissue and Organ Culture, 157(3). https://doi.org/10.1007/s11240-024-02788-4
Oseni, O. M., Pande, V., & Nailwal, T. K. (2018). A Review on Plant Tissue Culture, A Technique for Propagation and Conservation of Endangered Plant Species. International Journal of Current Microbiology and Applied Sciences, 7(07), 3778–3786. https://doi.org/10.20546/ijcmas.2018.707.438
Park, S. (2021). Plant Tissue Culture Techniques and Experiments.
Pasternak, T. P., & Steinmacher, D. (2024). Plant Growth Regulation in Cell and Tissue Culture In Vitro. In Plants (Vol. 13, Number 2). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/plants13020327
Phillips, G. C., & Garda, M. (2019). Plant tissue culture media and practices: an overview. In In Vitro Cellular and Developmental Biology – Plant (Vol. 55, Number 3, pp. 242–257). Springer New York LLC. https://doi.org/10.1007/s11627-019-09983-5
Phillips, R. L., Kaepplert, S. M., & Olhoft, P. (1994). Review Genetic instability of plant tissue cultures: Breakdown of normal controls (DNA methylation/genome rearrangements/rearrangement-induced premeiotically/repeat-induced point mutation/heterochromatin). In Proc. Nati. Acad. Sci. USA (Vol. 91). https://www.pnas.org
Pithiya, M. B., Sharma, S. K., Sharma, M., Sharma, M., & kotwal, N. (2022). Advancements and Challenges in Plant Tissue Culture: a Comprehensive Overview. Journal of Plant Biota, 1(1), 12–16. https://doi.org/10.51470/JPB.2022.1.1.12
Polivanova, O. B., & Bedarev, V. A. (2022). Hyperhydricity in Plant Tissue Culture. In Plants (Vol. 11, Number 23). MDPI. https://doi.org/10.3390/plants11233313
Rai, M. K., Shekhawat, N. S., Harish, Gupta, A. K., Phulwaria, M., Ram, K., & Jaiswal, U. (2011). The role of abscisic acid in plant tissue culture: A review of recent progress. In Plant Cell, Tissue and Organ Culture (Vol. 106, Number 2, pp. 179–190). https://doi.org/10.1007/s11240-011-9923-9
Ravindra B. Malabadi, Raju K. Chalannavar, & Kiran P. Kolkar. (2025). Plant cell totipotency: Plant tissue culture applications-an updated review. World Journal of Advanced Engineering Technology and Sciences, 16(2), 112–135. https://doi.org/10.30574/wjaets.2025.16.2.1262
Smith, R. H. (2006). Plant Tissue Culture Techniques and Experiments. www.elsevierdirect.com/rights
Tazeb, A. (2017). Plant Tissue Culture Technique as a Novel Tool in Plant Breeding: A Review Article. J. Agric. & Environ. Sci, 17(2), 111–118. https://doi.org/10.5829/idosi.aejaes.2017.111.118
Thakur, S., Shruti, Hashmi, S., Mishra, S., Ekka, S. K., Kushwaha, A., & Kujur, R. (2024). A Review on Plant Tissue Culture. Asian Journal of Biology, 20(2), 14–18. https://doi.org/10.9734/ajob/2024/v20i2387
Thorpe, T. A. (2007). History of plant tissue culture. In Molecular Biotechnology (Vol. 37, Number 2, pp. 169–180). https://doi.org/10.1007/s12033-007-0031-3 Wijerathna-Yapa, A., & Hiti-Bandaralage, J. (2023). Tissue Culture—A Sustainable Approach to Explore Plant Stresses. In Life (Vol. 13, Number 3). MDPI. https://doi.org/10.3390/life13030780






Pingback: Meristem Culture: The Definitive Guide to In Vitro Virus Elimination and Clonal Propagation - Aneknowledge.com