1. Introduction: The Strategic Role of Male Sterility in Hybrid Breeding
The strategic utilization of heterosis, or hybrid vigor, remains the primary driver for elevating global crop yields. To effectively capture this biological phenomenon at a commercial scale, male sterility systems provide indispensable biotechnological tools. These systems are highly valued for being biosafety-compliant and offering a high-efficiency alternative to the prohibitive labor costs and logistical constraints of manual emasculation. By facilitating controlled cross-pollination through the arrest of functional pollen development, these systems ensure the genetic purity of hybrid seed lots.

Male sterility is fundamentally classified into two categories based on genetic inheritance: Genic Male Sterility (GMS) and Cytoplasmic Male Sterility (CMS). While GMS is governed by independent nuclear loci, CMS represents a sophisticated manifestation of genomic conflict. It arises from the uncoordinated interplay between the mitochondrial (MT) and nuclear genomes, where specific mitochondrial rearrangements interfere with the developmental program of the stamen, necessitating nuclear restoration to regain fertility.
2. Comparative Architecture: Cytoplasmic (CMS) vs. Genic Male Sterility (GMS)
The divergence between CMS and GMS is rooted in their distinct genomic origins and the evolutionary pathways of their respective causal genes. While both result in the failure to produce viable pollen, their biological derivation and breeding utility differ significantly. Systematic observations of these phenotypes date back to 1763, when Joseph Gottlieb Kolreuter first documented male sterility, a trait now identified in hundreds of diverse plant species.
The following table delineates the genetic and mechanistic distinctions between these two primary systems:
| Criteria | Cytoplasmic Male Sterility (CMS) | Genic Male Sterility (GMS) |
|---|---|---|
| Genomic Origin | Mitochondrial (MT) Genome | Nuclear Genome |
| Inheritance Patterns | Uncoordinated inheritance between the organellar (mitochondrial) and nuclear genomes | Independent nuclear gene action; Mendelian inheritance |
| Primary Causal Mechanism | Mitochondrial-nuclear incompatibility; typically involving chimeric ORFs and transmembrane proteins | Disruption of nuclear-encoded genes essential for microsporogenesis |
While mitochondrial mutations provide the intrinsic trigger for CMS, modern genomic analysis has revealed a multilevel regulatory landscape where non-coding RNAs (ncRNAs) serve as critical mediators of the sterile phenotype.
3. The ncRNA Regulatory Landscape in Pollen Development
Non-coding RNAs (ncRNAs) are paramount in the multilevel regulation of microsporogenesis and stamen primordia differentiation. These molecules do not encode proteins but instead act as master regulators of the transcriptional and post-transcriptional environment. The ncRNA landscape is categorized into three primary classes:
• MicroRNAs (miRNAs):
◦ Biogenesis: Derived from MIR genes, pri-miRNAs are processed into pre-miRNAs in the nucleus by a complex involving RNA Polymerase II, DICER-LIKE 1 (DCL1), and auxiliary proteins HYPONASTIC LEAVES 1 (HYL1) and SERRATE (SE). After the mature miRNA duplex is formed and methylated by HEN1, it is exported to the cytoplasm via HASTY.
◦ Modes of Action: Mature miRNAs are loaded into Argonaute (AGO) proteins to form the RNA-induced silencing complex (RISC), which regulates target mRNAs through mRNA cleavage, translation inhibition, and expression silencing.
• Long Non-Coding RNAs (lncRNAs):
◦ Classification: Transcripts >200 nt categorized by genomic location as intronic, antisense, or long intergenic.
◦ Function: LncRNAs act as endogenous target mimics (eTMs) to sequester miRNAs or serve as precursors to miRNAs, effectively buffering the regulatory network.
• Phased Small Interfering RNAs (phasiRNAs):
◦ Biogenesis: Produced from PHAS loci and triggered by miRNAs like miR2118 or miR2275. In maize, the biogenesis of 24-nt phasiRNAs is strictly dependent on Dcl5.
◦ Reproductive Specificity: These molecules show tapetum-specific expression, and their absence or disruption is precisely timed with the onset of pollen abortion, highlighting their role in maintaining reproductive stability.
The three primary types of ncRNA conductors vary by size and “operational style”:
| Type | Size (Nucleotides) | Origin / Key Characteristic | Functional Mode |
|---|---|---|---|
| miRNA (MicroRNA) | 21–24 nt | Derived from MIR genes; forms a stable hairpin shape. | Cleavage or Translation Inhibition |
| lncRNA (Long Non-coding RNA) | > 200 nt | Lacks an open reading frame (ORF); can be “decoys.” | eTM (Target Mimicry) or Scaffolding |
| phasiRNA (Phased siRNA) | 21 or 24 nt | Produced from PHAS loci; often triggered by miRNAs like miR2118. | Direct mRNA Silencing |
This integrated miRNA–lncRNA–mRNA interaction network creates a robust regulatory logic that dictates fertility.
4. Phytohormone Homeostasis and ncRNA-Mediated Sterility
Phytohormones are the primary physiological coordinators of male reproductive development. As established in the literature, “Plant hormones are critical regulators of reproductive development, and inhibition of their synthesis and signal transduction will cause male sterility.” The homeostatic balance of these hormones is frequently modulated by conserved miRNA families:
• The Auxin Signaling Axis: Conserved families miR160 and miR167 target Auxin Response Factors (ARFs). Notably, in cotton, the overexpression of miR160 results in anther indehiscence and sterility specifically under High Temperature (HT) stress, whereas plants remain fertile under normal thermal conditions. This environmental dependency is a critical factor in field-based hybrid production.
• Brassinosteroids (BRs) and Gibberellins (GAs): These phytohormones coordinate organogenesis through the miR159–OsGAMYBL2 module. In rice, this network regulates the BR-responsive gene BU1 and GA biosynthetic genes CPS1 and GA3ox2, ensuring proper formation of the male genital organs.
• Jasmonic Acid (JA) and Abscisic Acid (ABA): Stress-responsive pathways involving miR825 and miR1120 modulate pollen development by targeting Leucine-Rich Repeat (LRR) genes, demonstrating how ncRNAs integrate environmental cues into reproductive timing.
Mechanism Snapshot
| Hormone | ncRNA Regulator | Effect on Fertility | Phenotype |
|---|---|---|---|
| Auxin | miR160 / miR167 | Regulates ARF10, 16, 17 | Controls stamen length; failure leads to short stamens. |
| Jasmonic Acid (JA) | miR160 (High Heat) | Suppresses JA synthesis | Anther Indehiscence: The anther fails to open and release pollen. |
| Brassinosteroids | miR159 | Coordinates signals via OsGAMYBL2 | Pollen abortion; failure to reach maturity. |
5. Mitochondrial Genetic Drivers: Chimeric ORFs and Causal Mechanisms
CMS is fundamentally driven by the dynamic and rearranged nature of the plant mitochondrial (MT) genome. Recombination events frequently produce chimeric open reading frames (ORFs) that typically possess transmembrane domains, allowing them to integrate into the inner MT membrane and disrupt organelle function.
Four critical models define MT gene-mediated CMS:
• Mode I: Cytotoxicity: Chimeric proteins, such as T-urf13 (maize) and orf79 (rice), exert toxic effects on cell viability. T-urf13 specifically encodes a protein with an inner MT membrane domain that compromises membrane integrity.
• Mode II: Energy Deficiency: Chimeric genes like orfH79 interact with the respiratory chain (e.g., Complex III), leading to reduced ATP production and increased reactive oxygen species (ROS) during energy-intensive pollen development.
• Mode III: Premature Programmed Cell Death (PCD): Genes such as WA352 (rice) and atp6c (maize) trigger premature tapetal PCD by inhibiting COX11 or inducing ROS bursts, depriving developing microspores of essential nutrients.
• Mode IV: Retrograde Regulation: This involves signaling from the MT genome to the nucleus. In maize, the MT gene orf355 activates the nuclear regulator ZmDREB1.7, which in turn upregulates orf355, establishing a positive feedback loop that reinforces the sterile phenotype.
6. Case Study: Genomic Editing of orf137 in Solanum lycopersicum (Tomato)
Historically, identifying CMS-associated genes in tomato was hampered by genomic complexity. The CMS line in this study was originally generated through asymmetric cell fusion between the wild relative Solanum acaule (cytoplasmic donor) and S. lycopersicum (nuclear donor), creating a mitochondrial-nuclear mismatch.

Experimental Methodology
Researchers utilized mitoTALENs (mitochondrial transcription activator-like effector nucleases) to target and disrupt the candidate gene orf137. These nucleases utilize mitochondrial localization signals to induce site-specific double-stranded breaks (DSBs) within the organelle genome.

Experimental Architecture
The construction of the mTAL137 vector utilized a CaMV 35S promoter to drive the expression of TALENs tagged with a Mitochondrial Localization Signal (MLS). This architecture ensures the nucleases are successfully imported into the organelle to target the orf137 coding sequence. Transformation was achieved via Agrobacterium tumefaciens.
Genomic Deletion Analysis
Post-transformation analysis of T0 lines demonstrated that mitoTALEN-induced double-stranded breaks (DSBs) resulted in substantial genomic shifts. Crucially, in all edited lines, the deleted regions contained only the orf137 gene, providing the functional proof that this single locus is responsible for the CMS trait.
| Line ID | Deletion Size (bp) | Affected Region | Fertility Status |
|---|---|---|---|
| mTAL137 #1 | 74 bp | Targeted orf137 disruption | Fully Restored |
| mTAL137 #2 | 11,491 bp | Large-scale deletion (orf137 only) | Fully Restored |
| mTAL137 #3 | 6,267 bp | Targeted deletion (orf137 only) | Fully Restored |
Summary of Experimental Results
• Mechanism of Repair: Analysis of long-read sequences confirmed that the DSBs were repaired via homologous recombination using other regions of the mitochondrial genome as templates.
• Phenotypic Restoration: The original Dwarf “CMS[P]” line exhibited a burst-aperture phenotype, where pollen grains underwent rapid expansion and rupture upon contact with germination media. In edited T1 lines, the removal of orf137 resulted in full fertility restoration, with pollen germination rates of 82% to 93%, validating orf137 as the causal agent of sterility.
Mechanisms of Double-Stranded Break (DSB) Repair and Recombination
Understanding the repair of mitochondrial DNA is critical for ensuring the genomic stability of edited lines—a prerequisite for commercial release. Mitochondrial DSBs are primarily “healed” through the Homologous Recombination (HR) pathway, using homologous sequences as templates.
Recombination Mapping
Analysis of the mTAL137 T0 lines revealed that the free DNA ends created by the TALENs joined with other homologous regions. Technical mapping confirmed the following specific recombination event:
- Left-side free ends (positions 214,004–214,749) recombined with a homologous genomic region (positions 41,249–41,974) of the reference genome (CMS-PMt002).
Ensuring Stability
This recombination effectively reverts the mitochondrial genome to a functional fertile state while ensuring stability by “healing” the break with homologous templates. This prevents the “reversion” to sterility often seen in unstable mutants. We utilize PacBio long-read sequencing to map these new recombination boundaries across the entire mitogenome, followed by Sanger sequencing for junction verification.
Impact on Pollen Viability and Phenotypic Restoration
The success of the tomato intervention is measured by the transition from total sterility to a state that is commercially viable and “full of seeds.”
The CMS Phenotype
The diagnostic phenotype for tomato CMS is an expanded aperture. In the parental Dwarf “CMS[P]” line, pollen appears normal until incubation, at which point the apertures expand, and the pollen grains burst, resulting in 0% germination. This structural weakness is also observable on the stigma post-pollination.
Quantitative Fertility Restoration
The edited T1 lines showed a total resolution of the bursting phenotype, with pollen tube growth and germination rates that match or exceed wild-type standards.
- Restored Germination Rates (T1 Lines): 82%, 93%, and 89%.
- Control (Micro-Tom): 89%.
- Parental CMS Line: 0%.
The resulting plants were phenotypically indistinguishable from the wild-type and produced mature fruits that were full of seeds, proving the technology’s industrial readiness.
7. Strategic Conclusions and Future Perspectives
The formation of male sterility is an emergent property of the intersection between mitochondrial genetic drivers, ncRNA regulatory networks, and phytohormone homeostasis. Mitochondrial rearrangements produce chimeric proteins that disrupt energy production or trigger premature cell death, while ncRNAs provide the multilevel control necessary to modulate the hormonal signaling required for microsporogenesis.
The successful application of mitoTALENs and the potential of CRISPR/Cas9 for ncRNA manipulation represent a paradigm shift in molecular breeding. These tools allow for the precise validation of candidate genes and the rapid creation of next-generation sterile lines. Such advancements are essential for accelerating the development of high-yielding, biosafety-compliant hybrid crops to meet global food security challenges.
Comparative Advantage
mitoTALENs currently provide a superior strategic advantage over CRISPR/Cas9 in mitochondrial applications due to the established efficiency of MLS-mediated protein import, avoiding the current technical hurdles of guide RNA transport into the organelle. This allows for the “curing” of elite lines without the genomic shuffling associated with traditional cell fusion.
Strategic Recommendations
- Mitogenome Assembly: Mandatory first step: Utilize PacBio long-read sequencing to assemble the mitochondrial genome of the target CMS line.
- Comparative Analysis: Identify chimeric ORFs by comparing sterile and fertile mitogenomes, prioritizing those with similarity to known CMS genes (e.g., orf137 / orf507).
- Vector Design: Construct MLS-tagged TALENs targeting confirmed CMS genes under robust promoters (e.g., 35S).
- Stability Validation: Use long-read sequencing to map recombination-mediated repair, ensuring the new mitochondrial structure is stable for multi-generational seed production.
Image Summary




Questions/Answers
1. What causes the molecular imbalance between mitochondrial and nuclear genomes?
The molecular imbalance between mitochondrial and nuclear genomes is primarily caused by genomic conflict or incompatibility that arises when the coordinated regulation between these two systems is disrupted. This imbalance most often manifests as cytoplasmic male sterility (CMS), a condition where a plant cannot produce viable pollen.

The primary causes of this molecular imbalance include:
1. Interspecific and Distant Hybridization (Alloplasmy)
The most common cause of molecular imbalance is nuclear substitution, often occurring through successive backcrossing or wide hybridization between distantly related species or subspecies.
- Mechanism: When the nucleus of one species is combined with the mitochondria (cytoplasm) of another (a state called alloplasmy), the interaction between them becomes “imbalanced”.
- Result: The new nuclear genome often lacks the specific fertility restorer (Rf) genes that were present in the original species to suppress harmful mitochondrial mutations. Without these nuclear equalizers, mitochondrial genes that were previously harmless are allowed to express, triggering cellular dysfunction.
2. Mitochondrial DNA Rearrangements and Chimeric ORFs
Plant mitochondrial genomes are unusually large and prone to frequent genomic rearrangements, duplications, and non-homologous recombination.
- Creation of Novel Genes: These recombination events produce chimeric open reading frames (ORFs)—novel genes composed of pieces of known mitochondrial genes fused with sequences of unknown origin.
- Dysfunctional Proteins: These new ORFs often encode transmembrane proteins that interfere with essential mitochondrial functions, such as the electron transport chain or ATP synthesis. The expression of these “selfish” mitochondrial elements creates an imbalance because the nuclear genome must then evolve or acquire new suppressors to regain genomic equilibrium.
3. Failure of Recombination Surveillance
The stability of the mitochondrial genome is largely under nuclear control. When specific nuclear genes responsible for monitoring and suppressing mitochondrial recombination fail, imbalance occurs.
- The Role of MSH1: The nuclear gene MutS HOMOLOG 1 (MSH1) acts as a suppressor of ectopic (abnormal) recombination in mitochondria.
- Inducing Instability: Mutation or suppression of MSH1 allows for “illegitimate” recombination and substoichiometric shifting, where rare, low-copy mitochondrial variants are rapidly amplified. This rapid shift in the mitochondrial genome population creates a sudden molecular imbalance that can induce sterility.
4. Disrupted Retrograde and Anterograde Signaling
Mitochondria and nuclei must constantly communicate to maintain cellular homeostasis. The nucleus provides approximately 95% of the proteins required for mitochondrial biogenesis.
- Retrograde Signaling: This is the process where mitochondria send signals to the nucleus to regulate nuclear gene expression in response to the organelle’s status.
- The Imbalance: In CMS systems, abnormal mitochondrial ORFs can emit detrimental retrograde signals that activate or upregulate specific nuclear factors—such as the RMS gene in rice—which then direct downstream pathways that lead to pollen abortion. This “lost harmony” between the signaling of the two genomes is a core component of the molecular imbalance.
5. Physiological Triggers of Imbalance
The imbalance is often tissue-specific, triggered by the massive increase in mitochondrial activity required during pollen development.
- Energy Demand: During microsporogenesis, the number of mitochondria per cell can increase by 20 to 40 times.
- The Threshold Effect: Mitochondrial mutations that may have a subtle, undetectable effect in vegetative tissues become catastrophic under the high energy demands of the anther. This results in a localized molecular imbalance characterized by reduced ATP levels and a burst of reactive oxygen species (ROS), which ultimately triggers premature programmed cell death (PCD) in the tapetum cells.
2. Can environmental factors like temperature trigger a reversion to fertility?
Yes, environmental factors, most notably temperature, can trigger a reversion to fertility in plants that are expected to be male-sterile. This phenomenon is documented across various crops and occurs in both genic male sterility (GMS) and cytoplasmic male sterility (CMS) systems.
Temperature-Induced Reversion
Temperature is one of the most critical environmental factors influencing the stability of male sterility:
- Maize: In CMS maize lines (specifically C and S types), cool and humid conditions are associated with a higher frequency of fertility restoration, whereas sterility is better maintained in hot and dry conditions. Conversely, cold weather during panicle development can also affect fertility stability in maize.
- Rice and Wheat: These crops often utilize Thermo-sensitive Genetic Male Sterility (TGMS), where plants are sterile at one temperature range (e.g., high temperatures) and become fertile at another (e.g., lower temperatures). For instance, certain wheat mutants are male-sterile between 7.4–11.4°C but become fertile above 11.4°C.
- Rapeseed: The Polima-type CMS in rapeseed is known to revert to pollen fertility under high temperatures in certain genotypes.
- Sorghum and Pearl Millet: In sorghum, scorching temperatures of 42°C and higher can impact fertility stability, while in pearl millet, cold weather during development can trigger reversion.
Other Environmental Triggers
While temperature is primary, other factors also play a role:
- Photoperiod (Day Length): Some systems, known as Photoperiod-sensitive Genic Male Sterility (PGMS), switch between sterile and fertile states based on day length. In rice, for example, certain lines are sterile during long days and fertile during short days.
- Humidity and Water Stress: High humidity has been noted to affect fertility stability in pearl millet. In sorghum, water availability and drought conditions can influence the restoration of male fertility.
- Light Intensity and Solar Radiation: Variability in light intensity and solar radiation can positively or negatively influence the expression of male sterility 2 to 3 weeks before anthesis.
Underlying Mechanisms
The reversion to fertility due to environmental cues is often driven by complex molecular interactions:
- Environment-Sensitive Restorer Genes: Reversion can be caused by the presence of environment-sensitive restorer-of-fertility (Rf) genes. For example, the expression of the Rf9 gene in maize is directly influenced by temperature.
- Epigenetic Changes: Environmental factors like high temperature can disrupt DNA methylation patterns, which in turn causes microspore sterility or reversion depending on the plant’s sensitivity.
- Metabolic Signaling: The nuclear gene MSH1 acts as a suppressor of mitochondrial rearrangements. In non-pollination conditions, MSH1 expression declines, which can trigger spontaneous fertility reversion as a reproductive strategy for isolated plants to self-pollinate.
Practical Implications
For plant breeders, this environmental sensitivity is a “double-edged sword”. While unintended reversion (often called “breaking sterility”) can contaminate hybrid seed fields and compromise purity, it is also intentionally exploited to create “maintainer-less” breeding systems. In these systems, breeders can multiply the sterile line by simply growing it in an environment that triggers its fertile state, allowing it to self-pollinate.
3. How does cytoplasmic male sterility benefit hybrid seed production?
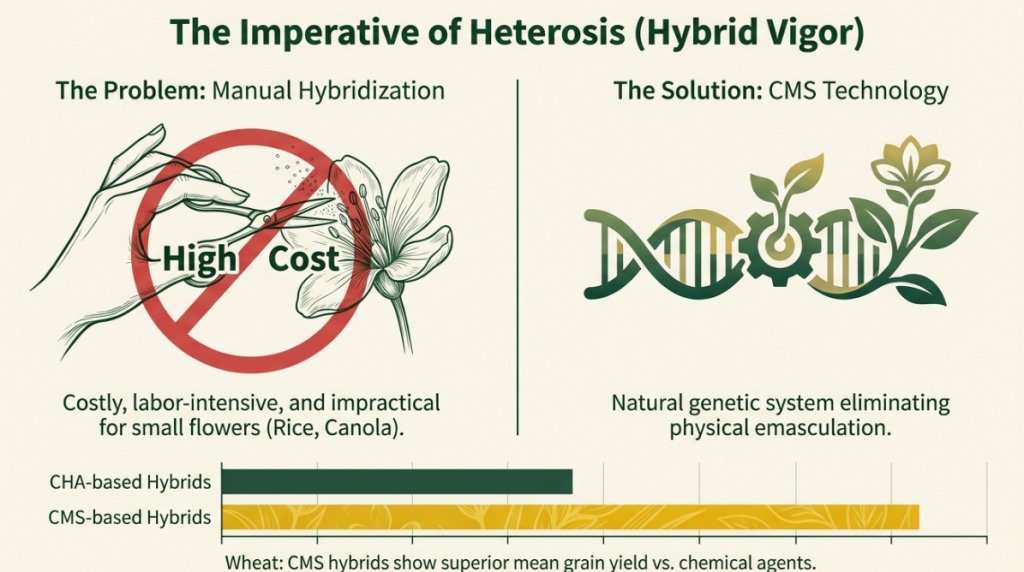
Cytoplasmic male sterility (CMS) provides several significant advantages for hybrid seed production, primarily by serving as an efficient and cost-effective pollination control mechanism.
The key benefits identified in the sources include:
1. Elimination of Manual and Mechanical Emasculation
The most prominent benefit of CMS is that it eliminates the need for manual or mechanical removal of male reproductive parts (such as detasseling in corn) from the female parent line. Traditional manual emasculation is:
- Labor-intensive and expensive: It requires hiring many skilled workers, which significantly drives up the cost of hybrid seeds.
- Time-consuming: In crops with small flowers, such as wheat or certain vegetables, manual emasculation is practically impossible on a commercial scale.
- Physically damaging: Manual or mechanical detasseling can injure the female plant, reducing its production potential.
2. Improved Hybrid Purity and Quality
CMS ensures higher genetic purity in the resulting seed lots by preventing “biological contamination” caused by the female inbred self-pollinating.
- It provides reassurance that the cross made is the intended one, rather than wondering if manual emasculation was performed too late.
- Studies in cabbage have shown that using CMS can increase the purity of commercial cultivars by 5–7% compared to other methods.
- CMS lines often produce higher quality seeds (larger size, better germination) because pollination is more controlled and uniform.
3. Increased Yield Potential
Using CMS can lead to higher yields through two main mechanisms:
- Energy Refocusing: Because sterile plants do not produce viable pollen, they have a reduced need for energy in their tassels/male organs. This energy can be refocused toward ear and seed development.
- Stress Reduction: Plants in CMS systems are not stressed by the physical injury of castration operations, resulting in yield increases of 3% to 19% compared to fertile versions of the same lines.
4. Reduced Need for Isolation Zones
CMS can reduce the requirements for isolation zones when dealing with regulated traits. Because sterile plants do not produce viable pollen, the risk of regulated traits traveling to and contaminating non-target species is significantly lowered.
5. Large-Scale Hybridization of Self-Pollinating Crops
CMS has revolutionized the seed industry by allowing the large-scale commercialization of hybrids in typically self-pollinating crops like rice, wheat, and cotton. For example, the use of CMS in hybrid rice has enabled yield increases of 15–20% over traditional cultivars, significantly contributing to global food security.
6. Simplified Breeding Systems
Because CMS is maternally inherited through the cytoplasm, it does not show segregation, which helps maintain population uniformity in cross-pollination systems. Once a line is converted to CMS through backcrossing, it provides a stable female parent for the three-line breeding system (consisting of the male-sterile A-line, the maintainer B-line, and the restorer R-line).
4. How does mitochondrial energy demand change during pollen development?
Pollen development is one of the most energetically demanding processes in a plant’s life cycle, requiring a massive increase in mitochondrial activity to support rapid cell division and maturation.
Mitochondrial energy demand changes during this period in the following ways:
1. Rapid Proliferation of Mitochondria
To meet the surge in metabolic needs, mitochondrial biogenesis accelerates dramatically during microsporogenesis. Research in crops like maize shows that the number of mitochondria per cell increases 20 to 40 times in the tapetal cells (which nourish the developing pollen) and the sporogenous cells. This rapid increase in organelle density is unique to the male reproductive tissues and has not been observed in other plant organs, such as developing ears.
2. Sharp Rise in ATP Requirements
The demand for adenosine triphosphate (ATP) rises sharply during the generation of pollen grains. The mitochondrial respiratory chain must operate at high flux to fuel cellular processes like meiosis, callose metabolism, and the synthesis of complex pollen wall structures. Because of this high flux, the mitochondrial population acts as a “stress sensor,” and any inability to maintain this level of energy production can lead to developmental arrest.
3. High Metabolic Flux in the Tapetum
The tapetum, the innermost layer of the anther wall, serves as a high-flux metabolic hub. It must provide a continuous supply of nutrients, enzymes, and precursors for sporopollenin (the main component of the pollen outer wall) to the developing microspores. This secretory function requires constant oxidative phosphorylation and ATP synthesis, making the tapetum highly sensitive to any mitochondrial dysfunction.
4. The “Energy Hypothesis” and Male Sterility
The extreme energy demand during this phase is central to the mechanism of cytoplasmic male sterility (CMS). According to the “energy hypothesis,” mitochondrial mutations that have negligible or undetectable effects in vegetative tissues (where energy demand is lower) become catastrophic during pollen development. When these mutated mitochondria cannot meet the soaring energy needs of the anther, it results in:
- Reduced ATP/ADP ratios and decreased membrane potential.
- A burst of reactive oxygen species (ROS) caused by an over-reduction of the ubiquinone pool.
- Premature programmed cell death (PCD) of the tapetal cells, which starves the developing pollen and leads to abortion.
5. Energy Refocusing in Sterile Plants
In plants that are successfully rendered male-sterile, the lack of pollen production significantly reduces the energy load on the tassel. This allows the plant to refocus its energy resources toward vegetative growth or female reproductive structures, often resulting in increased yield for ears or seeds in hybrid production systems.
5. What happens to mitochondrial DNA during spontaneous fertility reversion?
During spontaneous fertility reversion, a plant carrying male-sterile cytoplasm physically alters its mitochondrial DNA (mtDNA) to regain the ability to produce functional pollen. This process is not a change in the nuclear restorer genes but a rapid reorganization of the mitochondrial genome itself.
The primary changes to mitochondrial DNA during this reversion include:
1. Substoichiometric Shifting (SSS)
The most common mechanism for reversion is substoichiometric shifting. Plant mitochondria are often heteroplasmic, meaning they contain a primary, high-copy mitochondrial genome alongside rare, low-copy (substoichiometric) variants.
- Amplification and Suppression: During a reversion event, a rare “male-fertile” version of the mitochondrial genome is rapidly amplified to become the primary molecule, while the CMS-inducing genome is suppressed to very low levels or lost entirely.
- Speed of Transition: This shift can occur dramatically within a single generation, effectively “silencing” the sterility-associated sequence by making it a negligible part of the total mitochondrial population.
2. Genomic Recombination and Deletion
Spontaneous reversion is frequently characterized by de novo recombination events that physically alter the structure of the mtDNA.
- Loss of Sterility Genes: In some systems, the specific chimeric open reading frame (S-ORF) that causes sterility is physically deleted or lost through recombination. For example, in Texas-type (T) CMS maize, the CMS-inducing gene urf13 can be lost through recombination between repetitive sequences, resulting in a fertile revertant.
- Frameshift Mutations: Reversion can also occur if the mitochondrial ORF responsible for sterility suffers a frameshift mutation, rendering the resulting protein non-functional.
3. Failure of Nuclear Recombination Surveillance
The stability of mitochondrial DNA structure is normally maintained by nuclear “surveillance” genes, most notably MSH1 (MutS HOMOLOG 1).
- Suppression of Ectopic Recombination: MSH1 acts as a suppressor of abnormal (ectopic) recombination in the mitochondria.
- The Reversion Trigger: When MSH1 expression is mutated or suppressed (often by environmental signals or a “non-pollination” state), it allows for “illegitimate” recombination and substoichiometric shifting to occur. This instability provides the mitochondrial genome the flexibility to reorganize and revert to a fertile state as a survival strategy.
4. Examples in Specific Crops
- Maize: In S-type CMS maize, reversion arises through reorganization within the mitochondrial DNA or through new nuclear mutations.
- Common Bean: Reversion occurs at a rate of up to 1% and is associated with stoichiometric shifts that lead to the loss of the CMS-associated protein ORF239.
- Mustard Greens (B. juncea): Reversion involves shifting the copy number of the CMS-associated gene ORF220 until it is suppressed.
While these changes restore fertility, revertant plants are distinct from those with nuclear restorer (Rf) genes because they do not “restore” fertility when crossed back to the original CMS line; they have physically changed their cytoplasmic heritage rather than just masking it.
6. How does CMS improve seed quality and genetic purity?
Cytoplasmic male sterility (CMS) improves both genetic purity and seed quality by providing a stable, precision-based mechanism for pollination control that avoids the pitfalls of manual or mechanical methods.
Improvement of Genetic Purity
CMS ensures high genetic purity by systematically preventing biological contamination within hybrid seed production fields.
- Elimination of Self-Pollination: CMS deactivates the pollen-producing parts of the female parent, ensuring that the resulting seeds are the product of the intended cross rather than unwanted self-pollination.
- Avoidance of Human Error: Unlike manual emasculation (detasseling), which can be compromised if performed too late or incompletely, CMS provides a sense of “reassurance” that the cross is accurate.
- Uniformity through Maternal Inheritance: Because CMS is maternally inherited through the cytoplasm, it does not show segregation, which helps maintain the uniformity of the population in cross-pollination systems.
- Statistical Purity Gains: Studies have demonstrated that using CMS can increase the purity of commercial cultivars by 5–7% compared to other systems like self-incompatibility.
- Precision and Reliability: CMS has transformed breeding into a precision system that produces vigorous, genetically pure hybrids by serving as a robust mechanism to block self-fertilization.
Improvement of Seed Quality
Beyond purity, CMS contributes to the physical and physiological quality of the seeds produced.
- Superior Fecundation: The quality of fecundation in CMS systems is often superior, leading to a higher percentage of large seeds with a high Thousand Kernel Weight (TKW).
- Enhanced Vitality: Seeds produced via CMS often exhibit improved germination rates and a significant reduction in waste products, such as small round or broken seeds.
- The “Female Advantage”: CMS hybrids may exhibit enhanced reproductive capacity and seed vitality because resources are reallocated away from male functions and toward the development of the ear and seeds.
- Reduced Plant Stress: Because CMS eliminates the need for mechanical detasseling, the female plants are not subjected to the physical injury and stress of castration operations, which can otherwise decrease the production potential and seed quality of the parent line.
- Consistent Performance: CMS-based hybrids ensure controlled and more uniform pollination, which is essential for obtaining high-quality seeds with the desired genetic characteristics.
7. Can CMS increase crop yields by refocusing plant energy?
Yes, cytoplasmic male sterility (CMS) can increase crop yields by refocusing plant energy from male reproductive functions to female reproductive structures. Because CMS plants do not produce viable pollen, the energy that would normally be consumed by the male organs—such as the tassel in maize—is significantly reduced.
The specific ways this energy refocusing benefits yield include:
- Ear and Seed Development: In crops like maize, the energy saved by having a sterile tassel can be refocused directly toward ear development, leading to higher grain output. Studies have found yield increases ranging from 3% to 19% when comparing CMS lines to their fertile counterparts.
- The “Female Advantage”: This phenomenon is often referred to as a “female advantage,” where resource allocation is shifted away from male functions to enhance female reproductive capacity and seed vitality.
- Reduced Plant Stress: High yields are also maintained because CMS eliminates the need for manual or mechanical emasculation (detasseling). In fertile plants, these operations can physically damage or stress the plant, removing upper leaves and reducing its overall production potential.
- Extended Pollination Window: In some CMS maize cultivars, the plants exhibit slow senescence of the silk. This decelerated aging extends the duration for potential pollination, which increases the probability of effective seed development and enhanced yield.
- Heterosis Exploitation: Beyond simple energy refocusing, CMS is the primary tool for producing hybrid seeds on a commercial scale, allowing farmers to take advantage of heterosis (hybrid vigor). In rice, for example, the use of CMS-based hybrids has contributed to yield increases of 15% to 20% over traditional varieties.
8. What are the economic benefits of eliminating manual detasseling?
The elimination of manual detasseling through the use of cytoplasmic male sterility (CMS) provides several significant economic benefits, ranging from direct labor savings to substantial increases in crop yield and quality.
1. Direct Reduction in Labor and Resource Costs
Manual detasseling is a labor-intensive, expensive, and tedious process that requires hiring numerous skilled laborers.
- Labor Savings: CMS eliminates the need for large crews to manually remove male reproductive parts, which significantly reduces the cost of producing hybrid seeds.
- Machinery Costs: In traditional maize production, mechanical detasseling requires specialized machinery and is often followed by manual labor for verification; CMS removes the need for both.
- Scalability: Many vegetable and small-grain crops have flowers too tiny for manual emasculation to be economically feasible at a commercial scale; CMS makes large-scale hybridization of these crops possible.
2. Significant Yield Increases
Using sterile female parents leads to higher yields through the prevention of physical stress and the reallocation of cellular energy.
- Avoiding Plant Stress: Traditional detasseling often results in physical damage to the female plant, such as the accidental removal of upper leaves. Plants in CMS systems are not stressed by these operations, resulting in yield increases of 3% to 19% compared to fertile versions of the same lines.
- Energy Refocusing: Because sterile tassels do not produce viable pollen, the plant has a reduced demand for metabolic energy in its male organs. This energy can be refocused toward ear and seed development, further boosting productivity.
3. Improved Seed Quality and Purity
The use of CMS enhances the financial value of the final seed product by ensuring genetic integrity and better physical characteristics.
- Genetic Purity: CMS serves as a robust pollination control mechanism that prevents “biological contamination” caused by the female parent self-pollinating. This provides a higher guarantee that the resulting seeds are the intended hybrid cross.
- Superior Seed Traits: Seeds produced using CMS lines often exhibit a higher percentage of large seeds, higher 1,000-grain weight, and improved germination rates.
- Reduced Waste: Higher quality fecundation in CMS systems leads to a reduction in processing waste (such as broken or small round seeds) in final seed lots.
4. Operational Efficiencies
- Land Use Optimization: Because sterile plants do not produce viable pollen that can travel to non-target species, the requirements for isolation zones (especially when dealing with regulated traits) are reduced. This allows a higher percentage of the field to be utilized for actual production.
- Streamlined Breeding: CMS systems (particularly the “three-line” system) provide a stable, maternally inherited platform that ensures population uniformity and simplifies the maintenance of parent lines.
9. What is the three-line breeding system in hybrid seed production?
The three-line breeding system is a commercially robust pollination control mechanism used to produce hybrid seeds by utilizing cytoplasmic male sterility (CMS). This system relies on the interaction between three distinct plant lines: the A-line (male-sterile), the B-line (maintainer), and the R-line (restorer).

1. The Male-Sterile Line (A-line)
The A-line serves as the female or seed parent for hybrid production.
- Genetics: It possesses sterile (S) cytoplasm (mitochondrial DNA) and recessive nuclear fertility alleles (rfrf).
- Function: Because it cannot produce functional pollen, it is physically unable to self-pollinate. This allows it to be mass-pollinated by a chosen male parent without the need for labor-intensive manual emasculation.
2. The Maintainer Line (B-line)
The B-line is used specifically to multiply and propagate the A-line, which cannot reproduce on its own.
- Genetics: The B-line is nuclear-isogenic to the A-line, meaning their nuclear DNA is nearly identical, including the recessive rfrf alleles. However, the B-line contains normal fertile (F or N) cytoplasm.
- Function: When the A-line is crossed with the B-line (A × B), the resulting progeny inherit the sterile cytoplasm from the A-line and the recessive nuclear genes from both parents. This ensures the entire offspring population remains male-sterile, effectively creating more A-line seeds for future use.
3. The Restorer Line (R-line)
The R-line is the male parent used in the final step to create the commercial F1 hybrid seed sold to farmers.
- Genetics: It carries dominant fertility-restorer (Rf) genes in its nucleus.
- Function: When the A-line is crossed with the R-line (A × R), the resulting F1 hybrid inherits the dominant Rf gene. This gene suppresses the effects of the sterile cytoplasm, restoring male fertility to the hybrid plants. This restoration is essential for crops where the end product is a seed or fruit, such as maize, rice, or wheat, as the farmer’s crop must be able to produce its own pollen to set grain.
Operational and Economic Importance
The three-line system has revolutionized the seed industry by allowing for the large-scale production of genetically pure hybrids.
- Efficiency: It serves as a robust alternative to manual or mechanical detasseling, significantly reducing labor, time, and costs associated with seed production.
- Yield Gains: CMS-based hybrids often display heterosis (hybrid vigor), producing yields 15% to 50% higher than traditional inbred varieties.
- Widespread Use: This system is the backbone of hybrid production for major staples, including maize (T, C, and S types), rice (WA, BT, and HL types), sunflower (PET-1 type), and pearl millet (A1 type).
Despite its success, the system is complex to establish because developing stable restorer lines and maintaining isogenic A and B lines requires extensive backcrossing and precise genetic mapping. Additionally, reliance on a single CMS source can create genetic vulnerability, as seen in the 1970s when the T-cytoplasm in maize led to a massive outbreak of Southern Corn Leaf Blight.
10. Can CRISPR or mitoTALENs be used to permanently fix sterility?
Yes, both CRISPR and mitoTALENs can be used to permanently fix sterility, although they function by targeting different parts of the plant’s genetic system. While CRISPR is primarily used to manipulate nuclear genes (often for genic male sterility), mitoTALENs are the specialized “gold standard” for permanently curing cytoplasmic male sterility (CMS) by editing the mitochondrial genome.
mitoTALENs: Permanently Fixing CMS
mitoTALENs (mitochondria-targeted Transcription Activator-Like Effector Nucleases) are specifically designed to overcome the challenge of importing genetic material into mitochondria, which makes the standard CRISPR/Cas9 system difficult to use for mitochondrial DNA (mtDNA).
- Mechanism of Permanent Restoration: mitoTALENs introduce double-strand breaks (DSBs) directly into the specific CMS-associated open reading frame (S-ORF) that causes sterility. The mitochondrial genome then undergoes a homologous recombination repair process that physically deletes or removes the sterility-inducing gene from the mitogenome.
- Proven Success in Crops: This technology has been successfully used to restore full fertility in several crops by knocking out their specific sterility genes:
- Rice: Targeted disruption of orf79 (BT-CMS) and orf352 (WA-CMS) successfully restored fertility.
- Tomato: Disruption of orf137 resulted in transgenic lines that produced mature fruits full of seeds, with fertility restoration persisting into subsequent generations.
- Rapeseed and Broccoli: mitoTALENs were used to validate and disrupt orf125 and orf138, recovering fertility in these lines.
- Heritage and Stability: Once the CMS-causing gene is deleted via mitoTALENs, the fertility restoration is permanent and heritable; the plants remain fertile in subsequent generations even after the mitoTALEN expression cassette is no longer present.
CRISPR/Cas9: Fixing and Creating Genic Sterility
CRISPR/Cas9 is the primary tool for managing Genic Male Sterility (GMS), which is controlled by nuclear genes rather than mitochondrial ones.
- Restoring Fertility: CRISPR has been used to restore fertility in specific CMS systems by targeting the nuclear regulatory pathways that the mitochondria use to trigger sterility. For example, in CW-CMS rice, CRISPR/Cas9 was used to introduce mutations into the promoter of the nuclear RMS gene; reducing the expression of this gene permanently restored pollen fertility.
- Fixing Hybrid Vigor (Synthetic Apomixis): Genetic engineering is being explored to “fix” sterility and hybrid vigor simultaneously through synthetic apomixis. By using CRISPR to edit genes involved in meiosis and fertilization, researchers have enabled hybrid rice to reproduce asexually through seeds, allowing the permanent inheritance of hybrid vigor without the need for manual pollination control in every generation.
- Creating One-Step Breeding Systems: CRISPR/Cas9 is also used to create “one-step” systems that produce both sterile and maintainer lines simultaneously (such as with the ZmMS26 gene in maize), which simplifies the breeding process and eliminates the need for manual emasculation.
Summary of Differences
| Feature | mitoTALENs | CRISPR/Cas9 |
|---|---|---|
| Primary Target | Mitochondrial DNA (mtDNA) | Nuclear DNA |
| Type of Sterility | Cytoplasmic Male Sterility (CMS) | Genic Male Sterility (GMS) |
| Method of “Fix” | Physically deletes the S-ORF sterility gene | Edits nuclear regulators or sterility genes |
| Key Advantage | Permanently changes the cytoplasmic heritage | Can enable asexual seed production (apomixis) |
11. Can CRISPR create hybrid seeds that reproduce asexually?
Yes, CRISPR technology can be used to create hybrid seeds that reproduce asexually, a breakthrough process known as synthetic apomixis.
This technique has already been successfully demonstrated in hybrid rice, where researchers used gene editing to bypass the standard requirements for sexual reproduction.
How the Process Works
The creation of asexual hybrid seeds involves several key genetic manipulations:
- Targeting Reproductive Genes: Scientists use CRISPR/Cas9 to precisely edit genes involved in meiosis and fertilization pathways.
- Engineering Meiotic Recombination: By altering meiotic recombination, the system avoids the normal fertilization process.
- Producing Clonal Seeds: The result is the production of clonal seeds—seeds that are genetically identical to the parent plant—without the need for male and female gametes to fuse.
The Primary Benefit: Fixing Hybrid Vigor
The most significant advantage of this technology is the fixation of hybrid vigor (heterosis).
- The Problem: In traditional hybrid breeding, the superior traits (high yield, stress resistance) of the F1 generation are lost in the F2 generation due to genetic segregation during sexual reproduction.
- The Solution: Reproducing asexually through seeds allows the hybrid vigor to be stably inherited by the next generation. This means a farmer could potentially save and replant seeds from a hybrid crop while maintaining its high-performance characteristics.
Future Applications
While the most prominent breakthroughs have occurred in rice, researchers are currently exploring apomixis to reshape hybrid breeding systems in other crops, such as Chinese cabbage and various other plants. This approach is expected to become a core part of “molecular design hybrid breeding” in the future.
12. What is the difference between GMS and CMS breeding?
Breeding systems utilizing male sterility are primarily categorized into Genic Male Sterility (GMS) and Cytoplasmic Male Sterility (CMS), distinguished by their genetic origin, inheritance patterns, and the breeding methods required to produce hybrid seeds.
1. Genetic Control and Inheritance
- GMS: Is strictly governed by nuclear genes. It typically follows Mendelian inheritance, with the sterility trait often being monogenic and recessive.
- CMS: Is caused by mutations in the mitochondrial genome (cytoplasm) that interact with nuclear genes. It is passed on maternally through the female cytoplasm and follows non-Mendelian inheritance.
2. Breeding Systems
- Three-Line System (CMS): This is the standard commercial system for CMS, consisting of:
- A-line (Male-sterile): The female parent carrying sterile cytoplasm and recessive nuclear genes.
- B-line (Maintainer): Isonuclear to the A-line but with fertile cytoplasm; crossing A × B reproduces more A-line seeds.
- R-line (Restorer): The male parent carrying dominant restorer-of-fertility (Rf) genes that suppress the mitochondrial sterility, ensuring the F1 hybrid is fertile.
- Two-Line or One-Step Systems (GMS): GMS traditionally requires separating 50% fertile and 50% sterile plants in a segregating population, which is labor-intensive and costly. Modern breeding overcomes this using Environment-sensitive Genic Male Sterility (EGMS)—where plants are sterile in one environment (e.g., high temperature) and fertile in another (e.g., low temperature)—or through “one-step” systems created via CRISPR/Cas9.
3. Stability and Environmental Sensitivity
- CMS Sensitivity: Some CMS systems, particularly the S-type in maize, are sensitive to environmental conditions such as temperature and humidity, which can cause the plants to “break sterility” and produce unwanted pollen.
- GMS Stability: Nuclear genetic male sterility is generally considered highly stable and less sensitive to environmental changes than CMS, reducing the risk of accidental self-pollination.
4. Advantages and Limitations
| Feature | Genic Male Sterility (GMS) | Cytoplasmic Male Sterility (CMS) |
|---|---|---|
| Pollen Control | Highly effective; prevents selfing without manual detasseling. | Eliminates manual/mechanical emasculation, significantly reducing labor costs. |
| Cytoplasmic Effects | No adverse “pleiotropic penalties” or negative cytoplasmic effects. | May cause undesirable side effects like reduced growth, yield drag, or disease susceptibility (e.g., CMS-T and leaf blight). |
| Fertility Restoration | Easy; any wild-type line can restore fertility. | Complex; requires finding or developing specific, stable restorer lines with compatible Rf genes. |
| Commercial Use | Historically limited by separation costs; increasing with EGMS and gene editing. | Broadly used in major crops like maize, rice, and cotton for decades. |
13. How are genome-editing tools like CRISPR-Cas9 used in hybrid breeding?
Genome-editing tools like CRISPR-Cas9 are transforming hybrid breeding by enabling the precise creation and manipulation of male sterility systems, functionally validating candidate genes, and fixing hybrid vigor.
1. Creating De Novo Male Sterile Lines
CRISPR-Cas9 is primarily used to knock out specific nuclear genes essential for male gamete formation, bypassing the need to search for rare natural mutations.
- Maize: The technique has been used to precisely modify the ZmMS26 gene, resulting in complete male sterility while maintaining normal vegetative growth and grain production.
- Wheat: Researchers have targeted the TaNP1 gene and the TaTDF (Tapetal Development and Function1) gene; knocking out these homologs successfully induced male sterility.
- Tomato and Soybean: CRISPR has been used to target the SlMS10 gene in tomato to create male-sterile plants without exogenous vectors and the GmAMS1 gene in soybean to produce stable sterile lines.
2. Developing “One-Step” Breeding Systems
One of the most significant applications is the creation of a “one-step” hybrid seed production system. This technology uses CRISPR-Cas9 to edit genes responsible for recessive nuclear sterility to simultaneously produce both sterile and maintainer lines. This eliminates the labor-intensive requirement for manual or mechanical emasculation (detasseling) during the seed production process, thereby reducing costs.
3. Restoring and Engineering Fertility
CRISPR-Cas9 is used to investigate and manipulate the pathways that restore fertility to sterile plants:
- Artificial Restorer Lines: In Chinese wild rice-type (CW-CMS) rice, CRISPR/Cas9 was used to introduce mutations into the promoter of the nuclear RMS (Retrograde-Regulated Male Sterility) gene. By reducing the expression of this gene, researchers successfully restored pollen fertility, providing a new method to design artificial restorer lines.
- Functional Validation: The tool allows scientists to conclusively prove if a specific gene is responsible for sterility or restoration by knocking it out and observing the phenotypic change.
4. Synthetic Apomixis (Fixing Hybrid Vigor)
CRISPR-Cas9 is being used to “fix” hybrid vigor (heterosis) so it can be inherited across generations. This involves simultaneous genome engineering of meiosis and fertilization genes to enable hybrid rice to reproduce asexually through seeds (apomixis). This breakthrough could allow farmers to replant seeds from their hybrid crops while maintaining the high-yield benefits of the first generation.
5. Haploid Induction and Cyto-swapping
Genetic editing is used to create innovative breeding tools like haploid inducer lines.
- CENH3 Mutants: In crops like wheat, maize, and Brassica oleracea, CRISPR-Cas9-mediated mutagenesis of the CENH3 gene has been used to create paternal haploid inducers.
- Cyto-swapping: In maize, these haploid inducers have been used in a “cyto-swapping” method to rapidly move a desired nucleus into a male-sterile cytoplasm in just a few generations, significantly accelerating the breeding timeline.
6. Multiplex Editing and Transgene-Free Progeny
CRISPR-Cas9 allows for multiplex editing, where several genes for different traits—such as aroma, disease resistance, and flowering time—are targeted simultaneously to produce high-quality hybrid parents. Furthermore, technologies like the Transgene Killer CRISPR (TKC) system enable the CRISPR construct to self-eliminate after the desired edit is made, ensuring the final hybrid seeds are transgene-free.
14. What are the risks of using single CMS sources?
The use of a single source of cytoplasmic male sterility (CMS) across large-scale commercial hybrid production carries several critical risks, primarily centered on genetic vulnerability and the potential for catastrophic crop failure.
The key risks identified in the sources include:
1. Increased Susceptibility to Diseases (Biotic Stress)
The most significant risk of relying on a single CMS source is the potential for specialized pathogens to target that specific cytoplasm.
- The T-Cytoplasm Disaster: In the 1970s, the U.S. maize industry heavily relied on CMS-T, which was used in 85% of all hybrids.
- Molecular Cause: The mitochondrial gene responsible for sterility, urf13, also produced a protein that acted as a receptor for the toxin produced by Southern Corn Leaf Blight (Bipolaris maydis race T).
- Epidemic Consequence: This genetic uniformity allowed the blight to spread rapidly through cornfields in 1969 and 1970, resulting in an 80% yield loss and economic damage totaling approximately $1.0 billion.

2. Genetic Vulnerability and Fragility
Reliance on a narrow set of cytoplasm sources creates a narrow genetic base, making the entire hybrid population “genetically fragile”.
- When a single CMS source is disseminated globally to various sterile lines (as was done with WA-CMS in rice), it creates a dangerous uniformity that can be exploited by evolving pests or diseases.
- Diversity is required to mitigate the risk of catastrophic losses from both abiotic and biotic stresses.
3. Pleiotropic Penalties
Single CMS sources often carry undesirable pleiotropic effects—secondary negative traits that are inherited along with the sterile cytoplasm.
- Maize CMS-T: Caused reductions in plant height, leaf number, and overall yield (up to 4%), along with non-synchronous maturity of reproductive organs.
- Wheat T-CMS: The T. timopheevii cytoplasm has been associated with shriveled grains and low germination rates in hybrids.
- Brassica ogura CMS: Original versions induced chlorosis (yellowing of leaves) at low temperatures because the radish chloroplasts were incompatible with the Brassica nucleus.
4. Environmental Instability and Reversion
Single sources may be highly sensitive to environmental fluctuations, leading to “breaking sterility” (unintended reversion to fertility).
- Instability: Systems like CMS-S in maize are prone to partial fertility restoration under certain conditions, such as cold weather during panicle development.
- Contamination Risks: If a single, unstable source reverts to fertility unnoticed, it can lead to biological contamination of the entire hybrid seed field with unwanted pollen from the female parent.
5. Lack of Restoration Options
Relying on a single source limits the potential for hybrid screening because there may be limited fertility-restorer (Rf) genes available for that specific cytoplasm.
- In sunflowers, commercial production is heavily dependent on the PET1 source and its corresponding Rf1 gene, which lacks genetic diversity and leaves the crop vulnerable to environmental challenges.
- Developing strong restorer lines for specific cytoplasms (like CMS-C in maize) is often difficult, further narrowing the breeder’s choices.
15. Could CRISPR be used to engineer new types of sterility?
Yes, CRISPR technology can be used to engineer new types of male sterility, primarily by precisely knocking out nuclear genes essential for reproduction or by manipulating epigenetic pathways. This capability allows breeders to create sterile lines in crops that lack naturally occurring sterility systems.
Ways CRISPR is being used to engineer new types of sterility include:
1. Creating De Novo Genic Male Sterility (GMS)
CRISPR/Cas9 is used to knock out specific nuclear genes required for male gamete formation, bypassing the need to find rare natural mutations.
- Maize: Precision modification of the ZmMS26 gene resulted in complete male sterility while maintaining normal vegetative growth and grain production.
- Wheat: Researchers modified three homoallelic variants of the TaNP1 gene; homozygous mutation of all three led to total male sterility.
- Rapeseed: Concurrent mutation of two OPR3 gene homologs via CRISPR/Cas9 introduced complete male sterility.
- Soybean and Tomato: CRISPR has been used to target GmAMS1 in soybean and SlMS10 in tomato to generate stable male-sterile lines.
2. “One-Step” Hybrid Breeding Systems
One of the most innovative applications is the “one-step” system, where CRISPR is used to edit genes responsible for recessive nuclear sterility to simultaneously produce both sterile and maintainer lines. This approach eliminates the need for manual or mechanical detasseling during hybrid seed production, significantly reducing costs.
3. Engineering Artificial Restorer Lines
In rice types like CW-CMS, CRISPR/Cas9 has been used to introduce mutations into the promoter of the nuclear RMS (Retrograde-Regulated Male Sterility) gene. By reducing the expression of this gene, researchers successfully restored pollen fertility, demonstrating a method to design artificial restoration factors for existing or new sterility systems.
4. Tissue-Specific and Inducible Sterility
For genes that have essential functions in other parts of the plant (pleiotropic genes), CRISPR can be used to target them specifically in reproductive tissues.
- Tissue-Specific Promoters: Researchers can drive Cas9 expression using pollen- or ovule-specific promoters to target essential genes only in those organs, avoiding developmental defects in the rest of the plant.
- Inducible Systems: CRISPR can facilitate the creation of systems where sterility is triggered by external factors like chemicals or herbicides, allowing the plant to self-pollinate under normal conditions but remain sterile during hybrid seed production.
5. Epigenetic Sterility Engineering
The CRISPR-deactivated Cas9 (dCas9) system allows for targeted DNA methylation without changing the underlying DNA sequence. By directing methylation machinery to specific loci, researchers can silence or downregulate fertility-related genes to induce artificial sterility, which may peak at specific developmental stages like the tetrad or binucleate stages.
6. Synthetic Apomixis
CRISPR-Cas9 is being utilized to engineer meiosis and fertilization pathways to enable synthetic apomixis—where hybrid plants reproduce asexually through seeds. This allows for the permanent inheritance of hybrid vigor (heterosis) across subsequent generations, which is impossible with traditional sexual hybrid production.
7. Future Mitochondrial Engineering
While editing mitochondrial DNA (mtDNA) has traditionally been the domain of mitoTALENs, recent advancements like mitoCRISPR have begun to modify mitochondrial genes such as mtatp9 in tobacco, resulting in aborted stamens and no seed setting. This opens the door to designing and engineering entirely novel CMS-causing lesions in the mitochondrial genome.
16. How does synthetic apomixis allow for clonal hybrid seed production?
Synthetic apomixis allows for clonal hybrid seed production by utilizing gene-editing technology to bypass the standard processes of sexual reproduction, specifically meiosis and fertilization. This breakthrough method enables hybrid plants to produce seeds that are genetically identical to the parent, effectively “fixing” the genetic state of the hybrid.
The mechanism operates through several key steps:
- Engineering Meiotic Recombination: Scientists use genome-editing tools like CRISPR-Cas9 to target and modify reproductive regulation genes involved in the meiosis pathway. By engineering meiotic recombination, the system avoids the genetic shuffling that typically occurs when gametes are formed.
- Bypassing Fertilization: The system is designed to enable the plant to reproduce asexually through seeds without the need for fertilization. In normal sexual reproduction, the fusion of male and female gametes causes genetic segregation, which leads to the loss of superior traits in the next generation. Synthetic apomixis produces clonal seeds that retain the exact 100% nuclear DNA of the hybrid parent.
- Fixation of Hybrid Vigor (Heterosis): The primary advantage of this technique is the stable inheritance of hybrid vigor. In traditional hybrid breeding, the 15–50% yield increase seen in F1 hybrids is lost in the F2 generation due to segregation. Synthetic apomixis allows this high-performance phenotype to be passed down through generations without degradation.
- Applications in Breeding: While this technology has been most notably demonstrated in hybrid rice, research is ongoing to apply apomixis to other crops, such as Chinese cabbage, to reshape modern molecular design hybrid breeding systems.
By enabling hybrid seeds to produce clonal offspring, synthetic apomixis offers a path toward a more efficient seed production system where farmers could potentially save and replant seeds from high-yielding hybrid crops while maintaining their original vigor.
References
Abdullah, M., Sheraz, U., Ain, A. T., Nasir, B., Hammad, S., & Shokat, S. (2025). Exploring the Strategies of Male Sterility for Hybrid Development in Hexaploid Wheat: Prevailing Methods and Potential Approaches. In Rice (Vol. 18, Number 1). Springer. https://doi.org/10.1186/s12284-025-00807-2
Ali, A., Bakhsh, M. Z. M., Li, S., Zhang, X., Tu, J., & Yi, B. (2025). Cytoplasmic male sterility: Sterility induction, fertility restoration and gene-for-gene interaction of CMS/Rf genes. In Plant Science (Vol. 360). Elsevier Ireland Ltd. https://doi.org/10.1016/j.plantsci.2025.112721
Behera, L., Samal, K. C., Mishra, A., Sahoo, J. P., Dash, M., & Mishra, A. (2026). CRISPR-driven innovations in rice (Oryza sativa L.) breeding: precision development of male sterile lines. In Molecular Biology Reports (Vol. 53, Number 1). Springer Science and Business Media B.V. https://doi.org/10.1007/s11033-025-11269-6
Bhattacharya, J., Nitnavare, R. B., Bhatnagar-Mathur, P., & Reddy, P. S. (2024). Cytoplasmic male sterility-based hybrids: mechanistic insights. In Planta (Vol. 260, Number 4). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00425-024-04532-w
Bohra, A., Tiwari, A., Pareek, S., Joshi, R., Satheesh Naik, S. J., Kumari, K., Verma, R. L., Parihar, A. K., Patil, P. G., & Dixit, G. P. (2025). Past and future of cytoplasmic male sterility and heterosis breeding in crop plants. In Plant Cell Reports (Vol. 44, Number 2). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00299-024-03414-5
Braun, C. 1, Brown, G. G., & Levings Iiil, C. S. (1991). Cytoplasmic Male Sterility. In Cell Organelles.
Breton, S., Stewart, D. T., Brémaud, J., Havird, J. C., Smith, C. H., & Hoeh, W. R. (2022). Did doubly uniparental inheritance (DUI) of mtDNA originate as a cytoplasmic male sterility (CMS) system? BioEssays, 44(4). https://doi.org/10.1002/bies.202100283
Chapagain, S. (2024). A COMPREHENSIVE REVIEW ON-APPLICATION AND FUTURE PROSPECT OF CYTOPLASMIC MALE STERILITY. Acta Scientifica Malaysia, 8(1), 21–27. https://doi.org/10.26480/asm.01.2024.21.27
Chase, C. D. (2007). Cytoplasmic male sterility: a window to the world of plant mitochondrial-nuclear interactions. In Trends in Genetics (Vol. 23, Number 2, pp. 81–90). https://doi.org/10.1016/j.tig.2006.12.004
Chen, X., Li, Y., Sun, H., An, X., & Tang, J. (2024). Molecular Mechanisms of Male Sterility in Maize. In Plant Molecular Biology Reporter (Vol. 42, Number 3, pp. 483–491). Springer. https://doi.org/10.1007/s11105-024-01441-w
Chen, Z., Wu, J., Deng, X. W., & Tang, X. (2023). Establishment and Advances of Third-Generation Hybrid Rice Technology: A Review. In Rice (Vol. 16, Number 1). Springer. https://doi.org/10.1186/s12284-023-00670-z
Cheng, Z., Song, W., & Zhang, X. (2023). Genic male and female sterility in vegetable crops. In Horticulture Research (Vol. 10, Number 1). Oxford University Press. https://doi.org/10.1093/hr/uhac232
Chikh-Rouhou, H., Singh, S., Priyadarsini, S., & Mallor, C. (2025). Onion Male Sterility: Genetics, Genomics and Breeding. In Horticulturae (Vol. 11, Number 5). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/horticulturae11050539
Chu, X., Li, L., & Zhu, G. (2025). A review of the molecular mechanisms of the crop cytoplasmic genome and maternal genetic effects. Resources Data Journal, 4, 357–372. https://doi.org/10.50908/rdj.4.0_357
Coman, S., Szilagyi, L., & Ciontu, C. (2025). BREAKING CYTOPLASMIC MALE STERILITY IN INBRED CORN LINES (Zea mays L.): A REVIEW. Scientific Papers. Series A. Agronomy, LXVIII(2).
Dharsini, V. D., Subramanian, A., Premalatha, N., Boopathi, N. M., Djanaguiraman, M., & Santhanakrishnan, V. P. (2024). Fertile grounds: exploring male sterility in cotton and its marker development. In Molecular Biology Reports (Vol. 51, Number 1). Springer Science and Business Media B.V. https://doi.org/10.1007/s11033-024-09893-9
Dhillon, H. K., Sharma, M., Dhatt, A. S., Meena, O. P., Khosa, J., & Sidhu, M. K. (2024). Insights into cellular crosstalk regulating cytoplasmic male sterility and fertility restoration. In Molecular Biology Reports (Vol. 51, Number 1). Springer Science and Business Media B.V. https://doi.org/10.1007/s11033-024-09855-1
Eckardt, N. A. (2006). Cytoplasmic Male Sterility and Fertility Restoration. The Plant Cell, 18. http://www.fao.org/
Farinati, S., Draga, S., Betto, A., Palumbo, F., Vannozzi, A., Lucchin, M., & Barcaccia, G. (2023). Current insights and advances into plant male sterility: new precision breeding technology based on genome editing applications. In Frontiers in Plant Science (Vol. 14). Frontiers Media SA. https://doi.org/10.3389/fpls.2023.1223861
Farooq, A., Khan, U. M., Khan, M. A., Ali, Z., Maqbool, R., & Sajjad, M. (2024). Male sterility systems and their applications in hybrid wheat breeding. In Cereal Research Communications (Vol. 52, Number 1, pp. 25–37). Akademiai Kiado ZRt. https://doi.org/10.1007/s42976-023-00376-4
Gautam, R., Shukla, P., & Kirti, P. B. (2023). Male sterility in plants: an overview of advancements from natural CMS to genetically manipulated systems for hybrid seed production. In Theoretical and Applied Genetics (Vol. 136, Number 9). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00122-023-04444-5
Horn, R., Gupta, K. J., & Colombo, N. (2014). Mitochondrion role in molecular basis of cytoplasmic male sterility. In Mitochondrion (Vol. 19, Number PB, pp. 198–205). Elsevier. https://doi.org/10.1016/j.mito.2014.04.004
Islam, M. S., Studer, B., Møller, I. M., & Asp, T. (2014). Genetics and biology of cytoplasmic male sterility and its applications in forage and turf grass breeding. Plant Breeding, 133(3), 299–312. https://doi.org/10.1111/pbr.12155
Karki, A., Gyawali, A., Gahatraj, A., & Tiwari, S. (2026). Cytoplasmic Male Sterility: A Review On Its Role As A Genetic Innovation Driving Hybrid Breeding And Crop Improvement. Acta Scientifica Malaysia (ASM), 9(2), 104–109. https://doi.org/10.26480/asm.02.2025.104.109
Kitazaki, K., Oda, K., Akazawa, A., & Iwahori, R. (2023). Molecular genetics of cytoplasmic male sterility and restorer-of-fertility for the fine tuning of pollen production in crops. In Theoretical and Applied Genetics (Vol. 136, Number 7). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00122-023-04398-8
Liu, D., Shi, J., Liang, W., & Zhang, D. (2023). Molecular mechanisms underlying plant environment-sensitive genic male sterility and fertility restoration. In Seed Biology (Vol. 2). Maximum Academic Press. https://doi.org/10.48130/SeedBio-2023-0013
Liu, W., Ni, J., Ma, C., Yang, J., Wang, S., & Xu, D. (2026). Unraveling Advances in Rice Male Sterility Systems: From Genetic Basis to Hybrid Breeding Innovation. In Plants (Vol. 15, Number 3). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/plants15030507
Luo, D., Xu, H., Liu, Z., Guo, J., Li, H., Chen, L., Fang, C., Zhang, Q., Bai, M., Yao, N., Wu, H., Wu, H., Ji, C., Zheng, H., Chen, Y., Ye, S., Li, X., Zhao, X., Li, R., & Liu, Y. G. (2013). A detrimental mitochondrial-nuclear interaction causes cytoplasmic male sterility in rice. Nature Genetics, 45(5), 573–577. https://doi.org/10.1038/ng.2570
Martín, A. C., Castillo, A., Atienza, S. G., & Rodríguez-Suárez, C. (2018). A cytoplasmic male sterility (CMS) system in durum wheat. Molecular Breeding, 38(7). https://doi.org/10.1007/s11032-018-0848-4
Nie, H., Cheng, C., Kong, J., Li, H., & Hua, J. (2023). Plant non-coding RNAs function in pollen development and male sterility. In Frontiers in Plant Science (Vol. 14). Frontiers Media S.A. https://doi.org/10.3389/fpls.2023.1109941
Priyadarsini, S., Singh, S., & Nandi, A. (2024). Male sterility systems in the genomics era for expediting vegetable breeding. In Scientia Horticulturae (Vol. 338). Elsevier B.V. https://doi.org/10.1016/j.scienta.2024.113774
Priyadarsini, S., Singh, S., & Nandi, A. (2025). Molecular advances in research and applications of male sterility systems in tomato. In Plant Physiology and Biochemistry (Vol. 220). Elsevier Masson s.r.l. https://doi.org/10.1016/j.plaphy.2025.109503
Rahman, A., Rahman, M. H. S., Uddin, M. S., Sultana, N., Akhter, S., Nath, U. K., Begum, S. N., Islam, M. M., Naznin, A., Amin, M. N., Ahmed, S., & Hossain, A. (2024). Advances in DNA methylation and its role in cytoplasmic male sterility in higher plants. In Journal of Integrative Agriculture (Vol. 23, Number 1, pp. 1–19). Editorial Department of Scientia Agricultura Sinica. https://doi.org/10.1016/j.jia.2023.04.045
Ramlal, A., Nautiyal, A., Baweja, P., Kumar Mahto, R., Mehta, S., Pujari Mallikarunja, B., Vijayan, R., Saluja, S., Kumar, V., Kumar Dhiman, S., Lal, S. K., Raju, D., & Rajendran, A. (2022). Harnessing heterosis and male sterility in soybean [Glycine max (L.) Merrill]: A critical revisit. In Frontiers in Plant Science (Vol. 13). Frontiers Media S.A. https://doi.org/10.3389/fpls.2022.981768
Ren, W., Si, J., Chen, L., Fang, Z., Zhuang, M., Lv, H., Wang, Y., Ji, J., Yu, H., & Zhang, Y. (2022). Mechanism and Utilization of Ogura Cytoplasmic Male Sterility in Cruciferae Crops. In International Journal of Molecular Sciences (Vol. 23, Number 16). MDPI. https://doi.org/10.3390/ijms23169099
Shahbazi, M., Sharbrough, J., Knerova, J., Wendel, J. F., & Kopecky, D. (2026). Causes and Consequences of Cytonuclear Incompatibility in Hybrids of Flowering Plants 1 2. https://doi.org/10.1093/jxb/erag075/8475166
Sohail, A., Lu, C., & Xu, P. (2025). Genetic and molecular mechanisms underlying the male sterility in rice. In Journal of Applied Genetics (Vol. 66, Number 2, pp. 251–265). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s13353-024-00923-7
Toriyama, K. (2021). Molecular basis of cytoplasmic male sterility and fertility restoration in rice. In Plant Biotechnology (Vol. 38, Number 3, pp. 285–295). Japanese Society for Plant Cell and Molecular Biology. https://doi.org/10.5511/plantbiotechnology.21.0607a
Vasupalli, N., Mogilicherla, K., Shaik, V., Rao, K. R. S. S., Bhat, S. R., & Lin, X. (2025). Advances in plant male sterility for hybrid seed production: an overview of conditional nuclear male sterile lines and biotechnology-based male sterile systems. In Frontiers in Plant Science (Vol. 16). Frontiers Media SA. https://doi.org/10.3389/fpls.2025.1540693
Xu, F., Yang, X., Zhao, N., Hu, Z., MacKenzie, S. A., Zhang, M., & Yang, J. (2022). Exploiting sterility and fertility variation in cytoplasmic male sterile vegetable crops. In Horticulture Research (Vol. 9). Oxford University Press. https://doi.org/10.1093/hr/uhab039
Xu, Y., Yu, D., Chen, J., & Duan, M. (2023). A review of rice male sterility types and their sterility mechanisms. Heliyon, 9(7). https://doi.org/10.1016/j.heliyon.2023.e18204
Yang, H., Xue, Y., Li, B., Lin, Y., Li, H., Guo, Z., Li, W., Fu, Z., Ding, D., & Tang, J. (2022). The chimeric gene atp6c confers cytoplasmic male sterility in maize by impairing the assembly of the mitochondrial ATP synthase complex. Molecular Plant, 15(5), 872–886. https://doi.org/10.1016/j.molp.2022.03.002
Yu, H., & Xiong, X. (2025). Advances in the Research of Genic Male Sterility in Chinese Cabbage (Brassica rapa). In Plant Molecular Biology Reporter (Vol. 43, Number 4, pp. 1721–1731). Springer. https://doi.org/10.1007/s11105-025-01585-3
Zhang, H., Zhang, X., Wang, P., Jin, X., & Wu, X. (2022). Research Advances on Cytoplasmic Male Sterility and Restorer Genes in Maize. Guangdong Agricultural Sciences, 49(5), 1–9. https://doi.org/10.16768/j.issn.1004-874X.2022.05.001
Zhang, Y., Li, P., Yi, B., & He, D. (2026a). Current advances in plant mitochondria: Application revolution of cytoplasmic male sterility. In New Crops (Vol. 3). KeAi Communications Co. https://doi.org/10.1016/j.ncrops.2025.100081
Zhang, Y., Li, P., Yi, B., & He, D. (2026b). Current advances in plant mitochondria: Application revolution of cytoplasmic male sterility. In New Crops (Vol. 3). KeAi Communications Co. https://doi.org/10.1016/j.ncrops.2025.100081
Zhong, W., Zhang, S., Gu, X., Zhao, Y., Wu, Z., Liu, D., & Xing, W. (2025). Advances in Cytoplasmic Male Sterility in Sugar Beet from Mitochondrial Genome Structural Dynamics and Nuclear-Cytoplasmic Coordination. In International Journal of Molecular Sciences (Vol. 26, Number 20). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/ijms262010175






