1. Foundations of Hybrid Vigor and the Concept of Heterosis
The strategic deployment of heterosis, or hybrid vigor, remains the most potent tool in the global breeder’s arsenal for enhancing agricultural productivity. In commercial seed production, the transition from inbred line development to the creation of F1 hybrids is the fundamental mechanism for maximizing yield potential and phenotypic stability. By intentionally crossing genetically distinct parental lineages, we capture a level of vigor—characterized by superior growth, fertility, and biomass—that is unattainable in homozygous lines.

The conceptual evolution of this phenomenon has advanced significantly since Shull first coined “heterosis” in 1914 to replace the technically ambiguous “heterozygotic stimulation.” While often conflated, a professional distinction exists: “hybrid vigor” specifically denotes the phenotypic manifestation of increased biomass or yield, whereas “heterosis” describes the broader genetic superiority of the hybrid, including modern constructs like “defense heterosis.” This later term refers to the enhanced resilience against biotic stresses where the F1 outperforms even the most resistant parent.
The success of these programs depends on the inherent reproductive strategies of the target species. As categorized by Darwin and Simmonds, plants exhibit distinct tolerances to inbreeding based on their natural pollination mechanisms:
| Feature | Outbreeders (e.g., Maize, Alfalfa) | Inbreeders (e.g., Wheat, Soybean) |
|---|---|---|
| Reproductive Mechanism | Promotes cross-pollination (Monoecious, Dioecious, Self-incompatibility) | Promotes self-pollination (Cleistogamy, closed flowering) |
| Genetic State | Highly heterozygous across the population | Approaches complete homozygosity |
| Population Variability | Distributed across the population | Distributed primarily between component lines |
| Deleterious Recessives | Carries a heavy load of masked recessive alleles | Recessives are systematically eliminated (purged) |
| Inbreeding Tolerance | Severe depression; often intolerant of homozygosity | High tolerance; minimal vigor loss upon selfing |
For the genetic architect, the movement from a heterozygous F1 state toward homozygosity is not merely a biological shift but a high-stakes transition that requires precise modeling to prevent the collapse of commercial performance.
Conceptual Evolution: From Hybrid Vigor to Defense Heterosis
The trajectory of hybrid observations has transitioned from early phenotypic curiosity to the current era of molecular precision, establishing a technical foundation for modern breeding.
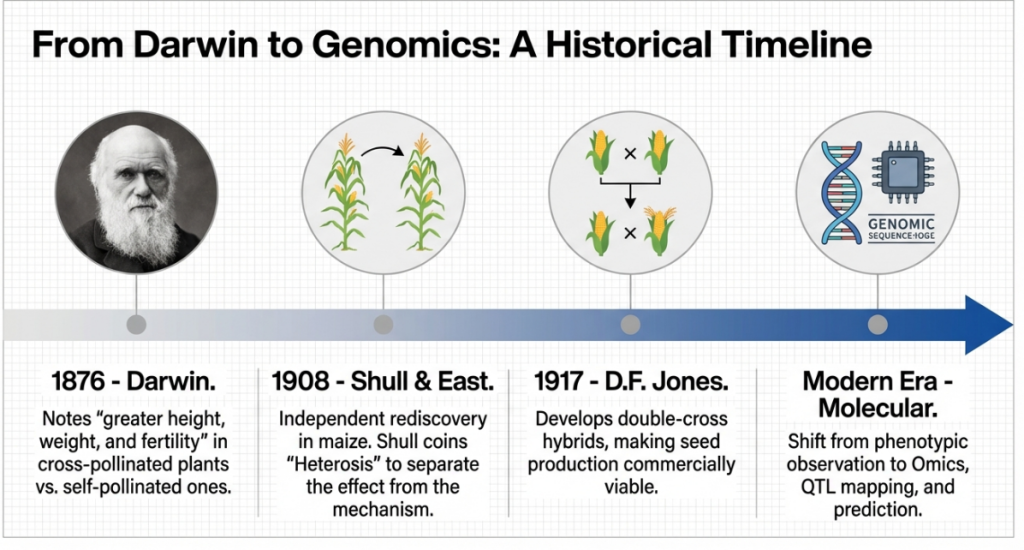
• 544 AD – 1877: Foundational Observations of Hybrid Superiority
◦ 544 AD: The Qimin Yaoshu provided the earliest documentation of hybrid vigor in mules; subsequent veterinary analysis confirmed these hybrids possessed significant disease-resistance advantages.
◦ 1766: Joseph Kölreuter demonstrated growth vigor in interspecific hybrids of tobacco and carnations.
◦ 1866: Gregor Mendel systematically recorded that F1 pea hybrids frequently exhibited stem lengths exceeding those of either parent.
◦ 1869: James Clark developed the “Magnum Bonum” potato hybrid, which serendipitously combined high yield with potent resistance to Potato Late Blight (Phytophthora infestans).
◦ 1877: Charles Darwin published The Effects of Cross and Self Fertilisation in the Vegetable Kingdom, providing the first quantitative distinctions between “outbreeders” (intolerant of inbreeding) and “inbreeders” (tolerant of self-pollination).
• 1909 – 1914: The Formalization of “Heterosis”
◦ 1909: Nilsson-Ehle documented that hybrid wheat progeny exhibited superior resistance to Yellow Rust (Puccinia striiformis) compared to parental lines.
◦ 1914: George Shull coined the term “Heterosis” to denote the superior performance of F1 hybrids. Critically, Shull broadened the concept to include resistance to biotic stresses and environmental rigors, establishing that hybrid advantage extends far beyond simple biomass.
• 2021 – Present: The Emergence of Defense Heterosis as a Framework
◦ The formalization of “Defense Heterosis” occurred following a landmark 2021 Arabidopsis thaliana study. This transition established a dedicated scientific framework to analyze immunity-linked hybrid advantage as a primary breeding objective rather than a secondary “byproduct” of general vigor.
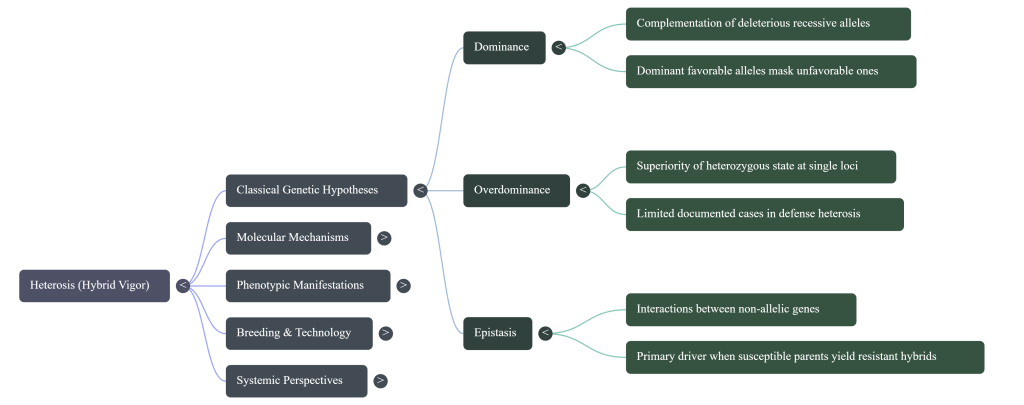
2. Genetic Mechanics: Dominance, Overdominance, and Epistasis Hypotheses
Understanding the genetic drivers of heterosis is essential for predicting specific combining ability and directing commercial selection. These hypotheses are not mutually exclusive; rather, they provide a multi-layered framework for interpreting how allelic interactions drive hybrid superiority.

2.1 The Dominance Hypothesis: The “Safety Net” Theory
The Dominance Hypothesis is the most widely accepted explanation for heterosis. Think of this model as a high-performing team where a specialized expert covers for a novice’s mistakes. Every plant carries “deleterious recessive alleles”—essentially genetic “glitches” that are usually hidden. Inbred parents are weak because they are homozygous at many loci, meaning there is no “expert” gene to mask those glitches.

In this model, the hybrid acts as a genetic safety net. It “collects” the favorable dominant alleles from both parents, ensuring that every “bad” recessive trait from Parent 1 is silenced by a “good” dominant trait from Parent 2.
Numerical Example: The Power of Masking
Consider a trait controlled by six genes. In this model, having even one dominant allele (N_) provides the full phenotypic benefit.
| Genotype State | Value Contribution |
|---|---|
| Base Value (all recessive: aa bb…) | 40 |
| Value of One Dominant Allele (N_) | +10 |
The Math of the Cross:
• Parent 1 (aa bb cc DD EE FF): 40+(3 genes×10)=70
• Parent 2 (AA BB CC dd ee ff): 40+(3 genes×10)=70
• F1 Hybrid (Aa Bb Cc Dd Ee Ff): 40+(6 genes×10)=100
The F1 is superior because it has accumulated the “best of both worlds,” using dominance to effectively hide the parental weaknesses.
2.2 The Overdominance Hypothesis: The “Synergy” Theory
But what if the hybrid’s strength isn’t just about hiding mistakes? The Overdominance Hypothesis suggests a “Heterozygote Advantage,” where the mixture itself possesses a unique power.
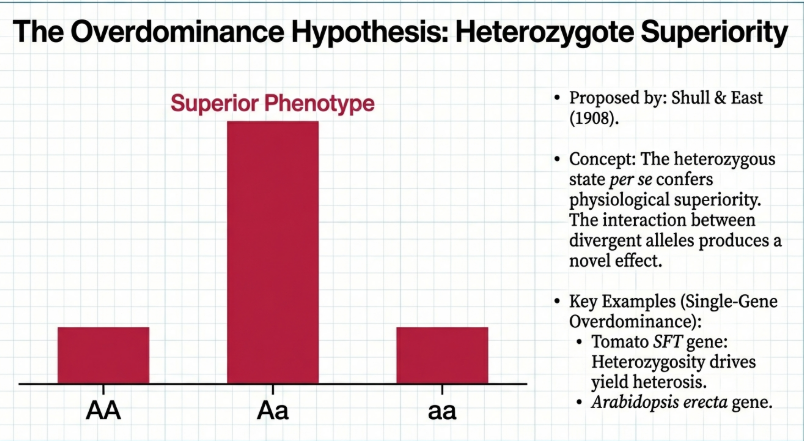
Think of this like alloyed metals or musical harmony. A single note is pure, but two different notes played together create a chord—a sound with a depth and resonance that neither note can produce alone. In this model, the heterozygous state (Aa) is physically superior to both homozygous states (AA or aa).
Numerical Example: The Synergy of the Mix
Here, the “mix” (Nn) actually performs better than the “pure” dominant (NN).
| Genotype State | Value Contribution |
|---|---|
| Base Value (all recessive: aa bb…) | 40 |
| Homozygous Dominant (NN) | +7 |
| Heterozygous (Nn) | +10 |
The Math of the Cross:
• Parent 1 (NN NN NN nn nn nn): 40+(3 genes×7)=61
• Parent 2 (nn nn nn NN NN NN): 40+(3 genes×7)=61
• F1 Hybrid (Nn Nn Nn Nn Nn Nn): 40+(6 genes×10)=100
In this synergy model, the hybrid reaches the same peak value of 100, but it does so through a mechanism that “pure” lines simply cannot reach.
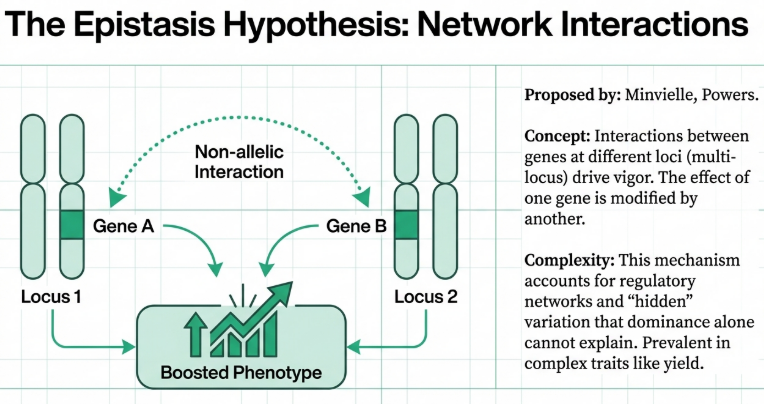
2.3 The Epistasis Hypothesis and Transcriptional Reprogramming
Beyond single-locus effects, the Epistasis Hypothesis emphasizes non-additive interactions between genes at different loci. Modern breeding programs utilize Genome-Wide Association Studies (GWAS) and expression Quantitative Trait Locus (eQTL) mapping to identify these complex, “context-dependent” regulatory relationships. By treating gene expression as a molecular phenotype, breeders can construct epistatic maps that reveal how trans-eQTLs and SNP-SNP interactions reshape transcriptional networks in the hybrid. This perspective suggests that heterosis is not just about allele presence, but about the unique genomic environment created when two divergent regulatory systems are integrated.
2.4 Comparative Analysis: Allele Roles and Predictions
While both models can result in a high-performing hybrid, they offer very different insights into the future of a breeding program.
Dominance vs. Overdominance: At a Glance
| Feature | Dominance Hypothesis | Overdominance Hypothesis |
|---|---|---|
| Core Mechanism | Favorable dominant alleles mask harmful recessives. | The heterozygous state (Aa) is superior to AA and aa. |
| Role of Recessives | Deleterious/Harmful; they decrease fitness. | A necessary component of the superior “mix” (Aa). |
| Is AA equal to Aa? | Yes (or nearly so). | No; Aa is significantly better than AA. |
| Genetic Fixation | Theoretically possible to “fix” all AA traits in a pure line. | Vigor is permanently tied to the heterozygous state. |
| Predictive Math | Hybrid (100) vs. Parents (70). | Hybrid (100) vs. Parents (61). |
The “So What?” for Breeders In reality, nature rarely chooses just one path. A landmark molecular study by Swanson-Wagner on maize mRNA found that while 78% of gene action was additive, 15% showed either high-parent or low-parent dominance, and 3% showed either overdominance or underdominance. When we add Epistasis (complex interactions between different genes) and epigenetic layers—such as the role of DDM1 in chromatin remodeling—it becomes clear that heterosis is a multi-engine machine.
2.5 Case Study: Defense Heterosis
When we apply these theories to immunity, we enter the field of Defense Heterosis. This is where hybrids exhibit superior resistance to pathogens compared to their parents.
• Dominance in Action (Gene Pyramiding): In rice breeding, we exploit dominance by accumulating multiple dominant Resistance (R) genes into a single line. For example, by introducing the blast resistance genes Pi-1, Pi-9, and Pi-kh into the high-yield restorer line Fuhui 676, breeders create a “pyramid” of protection. The hybrid inherits a robust shield that masks any susceptibility alleles from the other parent.
• Molecular Synergy and Timing: Hybrids often show an “above-high-parent” surge in Salicylic Acid (SA), the key immune hormone. But more SA isn’t always better; too much can stunt growth. The “aha!” moment here is timing. The core circadian factor CCA1 ensures that this SA surge happens rhythmically—usually at night or during an infection. This allows the plant to boost its defense without interfering with growth-related genes during the day, effectively solving the “growth-defense trade-off.”
• The Epigenetic Layer: Modern research shows that hybrids often possess higher “DNA methylation heterozygosity.” Epigenetic modifications can “lock” the parental SA baseline into an optimal range, ensuring the hybrid is always ready but never over-extended.
3. The Mechanics and Consequences of Inbreeding Depression
Inbreeding depression is the functional inversion of heterosis, manifesting as reduced survival and fertility. From a line development perspective, inbreeding serves as the primary mechanism to drive autozygosity and fix favorable alleles, despite the concomitant risk of exposing deleterious recessives.
Autozygous vs. Allozygous Genotypes
• Autozygous (Identical by Descent): Genotypes where alleles are replicates of a single ancestral gene, brought together through the mating of related individuals.
• Allozygous (Alike in State): Genotypes where alleles have the same function but descend from different ancestral copies.
Inbreeding systematically increases the probability of autozygosity. As a population moves toward homozygosity, the emergence of recessive states leads to measurable phenotypic decline: reduced stature, decline in pollen production, and suppressed seed quality.
The Strategic “So What?” Layer
For commercial operations, the symptoms of inbreeding depression are more than physiological—they are operational barriers. Severe depression in outbreeding species like alfalfa can result in genotypes that fail to survive, making the maintenance of homozygous parent lines nearly impossible. Furthermore, inbreeding can cause the collapse of male-sterile systems or the failure of self-incompatible lines, which are critical for controlled F1 seed production at scale.
Mathematical Realities of Population Mean Drop
The mathematical link between heterosis and inbreeding is demonstrated by the population mean. If we self-pollinate an F1 hybrid (AaBbCcDdEeFf) valued at 100, both the Dominance and Overdominance models predict a significant drop in the overall population mean to 85. This drop represents the quantifiable “depression” across generations as heterozygosity is lost and deleterious states are fixed.
4. Quantitative Benchmarks: Measuring Heterosis and Inbreeding
To evaluate breeding progress, we employ standardized metrics that quantify hybrid superiority and the degree of allelic fixation.
Core Metrics for Hybrid Evaluation
1. Mid-Parent Heterosis (MP%): Measures hybrid performance against the parental average.

2. High-Parent Heterosis (HP%): The commercial standard for determining if a hybrid exceeds the best existing inbred parent.
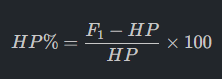
3. Inbreeding Coefficient (F): The proportional reduction in heterozygosity.
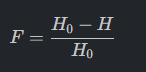
4. Effective Population Size (Ne): Determines the number of breeding individuals required to maintain genetic diversity.
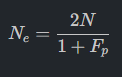
The Challenge of Polygenic Homozygosity
Reaching complete homozygosity in polygenic traits is a massive multi-generational undertaking. The frequency of completely homozygous individuals across generations (F2 to F8) drops precipitously as the number of segregating loci increases:
• 1 Locus: F2=50.00% → F8=99.22%
• 10 Loci: F2=0.01% → F8=92.45%
• 1000 Loci: F2≈0% → F8=0.04%
This reinforces why breeding timelines are governed by the speed of the mating system. For example, at the 6th generation, a full-sib mating system reaches 87% homozygosity, whereas a half-sib system only reaches 68%.
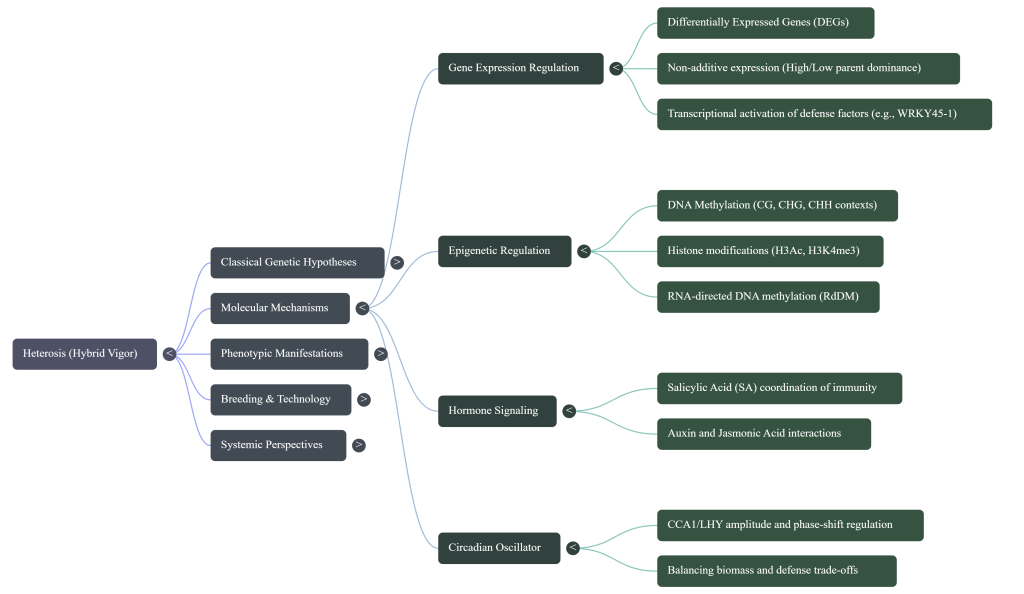
5. Molecular and Epigenetic Drivers of Hybrid Performance
High-throughput sequencing has transitioned our understanding from phenotypic observation to molecular regulation. Heterosis is increasingly viewed as a result of non-additive gene expression patterns and heritable epigenetic layers.
Differentially Expressed Genes (DEGs)
Phenotypic shifts in hybrids are categorized by four expression patterns: High parent, Above high parent (vital for defense), Low parent, and Below low parent. Integrated transcriptomic analysis shows that yield heterosis often results from the repression of defense genes, explaining why many high-yield hybrids lack concurrent disease resistance.
The Growth-Defense Balance and DDM1
Defense heterosis is mediated by the immune hormone Salicylic Acid (SA). While constitutive SA accumulation is detrimental to biomass, a pathogen-induced “above-high-parent” surge drives resistance. This balance is managed by:
• The Circadian Clock (CCA1): Rhythmically coordinates immunity genes to overexpress before dawn while repressing them during the day to maximize photosynthesis.
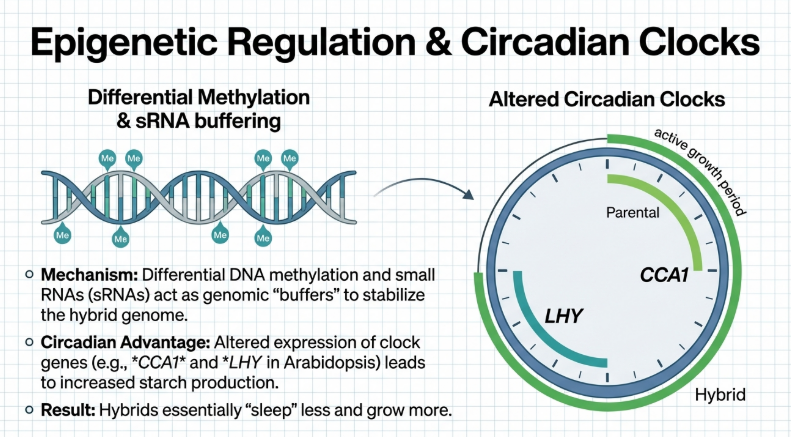
• DDM1 (Decreased DNA Methylation 1): Acts as a molecular “lock,” maintaining the parental SA baseline within an optimal range to prevent yield drag.
Epigenetic marks, such as histone H3 acetylation (H3Ac) at SA biosynthetic loci, allow the hybrid to dynamically activate defense modules without altering the underlying DNA sequence.
The Salicylic Acid (SA) Balancing Act: Coordinating Growth and Defense
The strategic challenge in crop protection is the metabolic cost of defense. Constitutive immunity is detrimental to yield; thus, the hybrid must manage hormone levels with extreme precision.
• The Yield-Defense Trade-off: High baseline levels of Salicylic Acid (SA) are incompatible with biomass heterosis. Hybrids maintain a low-parent SA baseline under normal conditions, a state managed by chromatin remodeling via DDM1 (Decreased DNA Methylation 1).
• The Hybrid Solution: Upon pathogen challenge, F1 hybrids exhibit a timely, “Above-High-Parent” surge in SA. This surge is indispensable for the resistance advantage and is temporally regulated to avoid growth interference.
• Hormonal Crosstalk: The SA pathway interacts with Jasmonic Acid (JA) in a complex manner. While often antagonistic, SA can activate JA signaling through receptors NPR3 and NPR4 in a noncanonical manner, promoting effector-triggered immunity.
These biological mechanisms culminate in observable resistance across the world’s most vital agricultural crops.
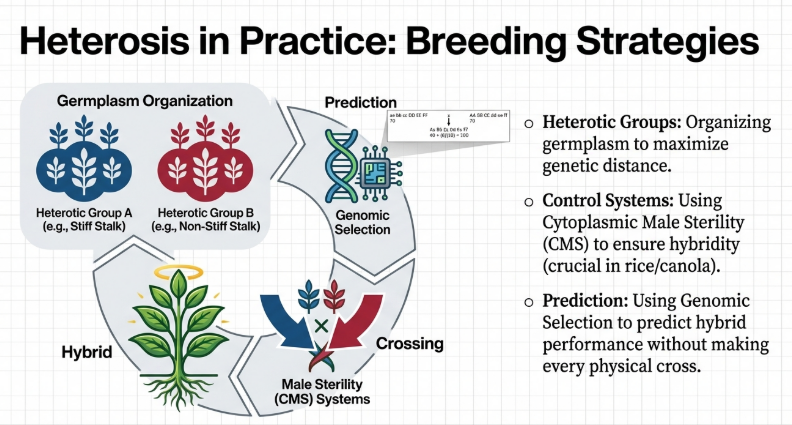
6. Commercial Application: Optimizing Hybrid Cultivar Performance
The roadmap for modern breeding involves leveraging genetic and molecular insights to overcome the traditional yield-defense trade-off.
Strategic Selection of Mating Systems
Breeders must match the mating system to the species’ reproductive biology and the required speed of allelic fixation:
| Mating System | Description | Primary Application | Speed to Homozygosity (Gen 6) |
|---|---|---|---|
| Self-pollination | An individual mated with itself | Standard for pure line development | ~97-99% (Rapid) |
| Full-sib Mating | An individual mated with itself | Self-incompatible or dioecious species | 87% (Moderate) |
| Half-sib Mating | Crossing plants with one parent in common | Maintaining wider genetic variability | 68% (Slow) |
| Backcrossing | Hybrid mated to recurrent parent | Incorporating specific traits into elite backgrounds | High for recurrent loci |
Integrated Framework for Future Commercial Success
To achieve durable resistance and stable yields, we are moving toward an AI-driven, multi-omics approach:
1. Multi-omics Integration: Large-scale gathering of phenomic, genomic, and transcriptomic data to predict specific combining ability.
2. Precision Engineering: Utilizing CRISPR/Cas9 to fix favorable heterozygous alleles or engineer the DDM1/CCA1 modules to fine-tune hormone dynamics.
3. Ecological Management: Utilizing LysM receptors and managing rhizosphere microbiota to enhance defense heterosis through beneficial symbionts.
By synthesizing classical dominance hypotheses with precision molecular tools and ecological insights, we provide a definitive roadmap for the next generation of high-performance commercial hybrids.
7. Case Studies: Defense Heterosis Across Major Crop Species
Defense heterosis manifests with high variability depending on the specific crop-pathogen interaction system:
• Maize: Griffing’s diallel studies against Exserohilum turcicum (Northern Leaf Blight) and Bipolaris maydis (Southern Leaf Blight) demonstrate that even highly susceptible parents can produce resistant F1 hybrids. This confirms that overdominance and epistasis drive traits that are not phenotypically visible in the inbred parents.
• Rice: The “Gene Pyramiding” strategy has successfully introduced broad-spectrum resistance genes (Pi-9, Pi-I, Pi-kh) into high-yield restorer lines like Fuhui 676. The resulting hybrids achieve a dual benefit of superior yield and potent blast resistance.
• Wheat: As an allohexaploid, wheat exhibits “fixed” heterosis. Studies confirm that nascent allohexaploids display Expression Level Dominance (ELD) favoring the Aegilops tauschii allele, providing robust resistance against Powdery Mildew. This is further regulated by the non-additive repression of miRNAs (e.g., miR9009, miR167, miR319), leading to the derepression of NBS-LRR and chitinase genes.
• Potato/Pepper: Mating designs have shown that specific combining ability—gene interactions unique to particular crosses—is the primary driver of resistance against Potato Late Blight and Chilli Leaf Curl Virus.
8. Conclusion: Shaping the Future of Food Security
The integration of these classical theories with modern tools like GWAS (Genome-Wide Association Studies) and CRISPR gene editing is transforming crop science. We are moving from observing “lucky” hybrids to active engineering.
By understanding whether a trait is driven by the “Safety Net” of dominance or the “Harmony” of overdominance, we can precisely edit genomes to achieve durable resistance and stable yield. Defense Heterosis is no longer a byproduct; it is a precision tool. As we fine-tune these genetic engines, we secure a future where our crops are not just more productive, but naturally fortified against the challenges of a changing world.

Image Summary

Questions/Answers
1. What are the three classical genetic theories that explain heterosis?
The three classical genetic theories that explain heterosis are the dominance hypothesis, the overdominance hypothesis, and the epistasis hypothesis.
1. Dominance Hypothesis
Proposed by Bruce in 1910 and later refined by Jones in 1917, this theory is currently the most widely accepted explanation for heterosis.
• Core Premise: The hybrid’s superior performance results from favorable dominant alleles from one parent masking or complementing deleterious recessive alleles from the other parent.
• Mechanism: During hybridization, the F1 hybrid accumulates these superior dominant alleles at multiple loci, leading to increased vigor.
• Prediction: This model suggests that if all deleterious alleles could be removed through recombination and selection, it would be possible to create an inbred line with performance equal to the hybrid.
2. Overdominance Hypothesis
Proposed independently by Shull and East in 1908, this theory attributes hybrid vigor to the heterozygous state itself.
• Core Premise: A heterozygous locus is inherently superior to either of its homozygous states.
• Mechanism: Synergistic allelic interactions at a single locus result in greater vigor, size, or productivity than that of either parent.
• Prediction: Unlike the dominance theory, this hypothesis predicts that an inbred line cannot be developed that equals the performance of the F1 hybrid.
3. Epistasis Hypothesis
While the first two theories focus on interactions at a single locus, the epistasis hypothesis addresses interactions between multiple genes.
• Core Premise: Heterosis arises from interactions between non-allelic genes at two or more different genetic loci.
• Mechanism: These complex, non-additive interactions (such as additive-by-additive or dominance-by-dominance) create favorable gene combinations in the hybrid that are not present in either parent.
• Significance: This theory is often used to explain complex quantitative traits, like grain yield, where multiple underlying acting traits are controlled by numerous genetic loci.
2. How do molecular mechanisms like DNA methylation influence hybrid performance?
DNA methylation influences hybrid performance by acting as a heritable layer of epigenetic information that regulates gene expression patterns without altering the underlying DNA sequence. These modifications are genotype, tissue, and developmental stage-specific and are significantly related to the manifestation of heterosis (hybrid vigor) in various crops.
Regulation of Gene Expression and Transcriptome Profiles
DNA methylation primarily influences hybrid performance by altering the transcriptome profile, leading to the differential expression of genes that govern growth, fitness, and metabolic pathways.
• Non-additive Expression: Methylation variations in hybrids can cause gene expression levels to deviate from the average of the parents (mid-parent value), resulting in superior phenotypic traits.
• Differential Methylation Patterns: Distinct methylation patterns are frequently observed between F1 hybrids and their parents. In some species, such as maize, hybrids have been reported to be less methylated than inbred lines. Conversely, studies in Arabidopsis, rice, and pigeonpea have shown that hybrids can exhibit significantly higher methylation levels than their corresponding parents.
• Methylation Reprogramming: The process of hybridization can trigger immediate epigenetic reprogramming, creating new non-parental epialleles in the F1 generation.
Influence on Specific Performance Traits
Molecular mechanisms involving DNA methylation help coordinate the balance between different performance traits, such as growth and immunity.
• Defense Heterosis: In some hybrids, elevated DNA methylation leads to the repression of defense-related genes (e.g., PR1, PR2, or Xa1). This downregulation can attenuate basal defenses, which may facilitate biomass heterosis by reallocating resources toward growth.
• Stress Tolerance: Specific methylation levels regulate genes involved in stress responses. For example, lower levels of DNA methylation in Hibiscus cannabinus hybrids have been linked to enhanced cadmium tolerance.
• Circadian Regulation: Methylation at CHH sites in the promoter regions of key circadian clock genes, such as CCA1, can regulate their activity. These altered rhythms influence photosynthesis and starch accumulation, contributing to increased hybrid biomass.
Key Molecular Actors
The influence of DNA methylation on hybrid performance is driven by specific molecular pathways and proteins:
• DDM1 (Decreased DNA Methylation 1): This chromatin remodeling factor is considered a major regulator of heterosis. F1 hybrids with homozygous ddm1 mutations show significantly reduced vegetative heterosis, demonstrating that DDM1-mediated methylation is essential for the full manifestation of hybrid vigor.
• RdDM (RNA-directed DNA Methylation): This pathway uses 24-nucleotide small interfering RNAs (siRNAs) to target DNA methylation to specific genomic regions. In hybrids, siRNAs from one parent can modify the epigenetic state of the other parent’s allele through trans-chromosomal methylation (TCM), further contributing to the unique gene expression patterns seen in heterotic offspring.
• METHYLTRANSFERASE-1 (MET1): While MET1 is essential for maintaining methylation in the CG context, some studies in Arabidopsis suggest it may not be directly involved in the establishment of vegetative heterosis in certain hybrids. However, other studies indicate that reduced MET1 activity can correlate with a decline in overall heterosis.
3. In what ways does heterosis improve disease resistance and yield?
Heterosis, or hybrid vigor, refers to the superior performance of an F1 hybrid compared to its parents in traits such as biomass, speed of development, fertility, yield, and stress resistance. Historically, it has been a cornerstone of modern agriculture, responsible for yield increases between 15% and 50% depending on the crop.
Heterosis improves disease resistance and yield through a combination of genetic mechanisms, physiological optimizations, and molecular signaling networks:
1. Improving Yield
Heterosis enhances yield by optimizing the plant’s biological efficiency and structural components:
• Physiological Efficiency: Heterotic hybrids often exhibit higher photosynthetic efficiency and lower respiration rates compared to inbred lines. They also show increased leaf area index and cumulative growth rates, allowing them to accumulate more biomass over time.
• Metabolic Balance: Inbred parents often have unbalanced metabolic systems with “physiological bottlenecks” where specific enzyme reactions limit growth. Hybridization can overcome these limitations by bringing together complementary inbred parents, resulting in a better-balanced metabolic system in the hybrid.
• Transcriptional and Genomic Complementation: Hybrids can express hundreds of additional genes through a mechanism called Single-Parent Expression (SPE), where genes silent in one parent are active in the hybrid. This global expression complementation is significantly related to phenotypic heterosis for traits like plant height and ear size.
• Structural Components: Heterosis directly impacts specific yield-contributing traits. In maize, this includes increased kernel number, ear weight, and cob size. In vegetables like tomato, single overdominant genes such as SFT can increase yield by up to 60% by optimizing shoot architecture and flower production.
2. Improving Disease Resistance (Defense Heterosis)
Hybrids often display enhanced immunity against pathogens compared to their parental lines, a phenomenon specifically termed “defense heterosis”.
• Strategic Gene Pyramiding: Breeding strategies utilize heterosis to accumulate multiple dominant resistance (R) genes from genetically distinct parents into a single hybrid. This complementation of dominant alleles masks deleterious recessive alleles from the other parent, providing broad-spectrum resistance.
• Hormonal Regulation (Salicylic Acid): Salicylic acid (SA) is a key immune hormone. In hybrid combinations exhibiting defense heterosis, the progeny accumulate SA to levels exceeding those of both parents following a pathogen challenge. This above-high-parent SA surge is indispensable for the observed resistance advantage.
• Activation of Defense Transcription Factors: Certain transcription factors, such as WRKY45-1, are specifically activated in hybrids. These factors regulate programmed cell death and defense gene expression, making the hybrid’s defense signaling pathways more efficient than those of its parents.
• Epigenetic Amplification: Epigenetic mechanisms, including DNA methylation and histone modifications, can de-repress defense-related genes or amplify defense signals. For instance, small RNAs (miRNAs) in hybrids may exhibit non-additive repression, leading to the increased expression of their target resistance genes.
3. Balancing Growth and Defense
One of the most significant ways heterosis improves plant performance is by managing the trade-off between yield and defense.
• Circadian Coordination: The core circadian transcription factor CCA1 rhythmically coordinates growth and immunity genes. It ensures that growth-related genes are overexpressed during the day to maximize biomass, while immunity genes (via SA accumulation) are activated at night or following infection, allowing the hybrid to achieve both high yield and strong resistance simultaneously.
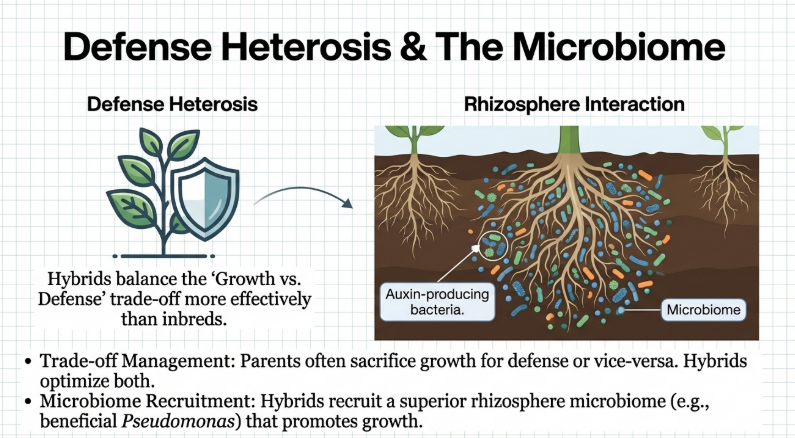
• Microbiome Interaction: Heterosis also influences the composition of the rhizosphere microbiome. Hybrids may recruit specific beneficial soil-borne microbes that improve nutrient uptake and inhibit pathogens more effectively than those recruited by inbred parents.
4. How do metabolic bottlenecks explain hybrid vigor?
Metabolic bottlenecks (also referred to as physiological bottlenecks or limiting factors) explain hybrid vigor through the metabolic balance concept, which suggests that the superior performance of hybrids results from a more efficient and better-equilibrated metabolic system than that of their inbred parents.
The following points detail how these bottlenecks contribute to heterosis:
• Unbalanced Systems in Inbreds: Inbred parental lines tend to have “unbalanced” metabolic systems where various enzymes are controlled at highly variable activity levels. Because they are highly homozygous, they often possess specific enzyme reactions that are severely deficient or under-active.
• Limiting Growth: These specific deficiencies act as physiological bottlenecks, where a single sluggish reaction or “limiting factor” restricts the entire metabolic pathway and, consequently, the plant’s overall growth and productivity.
• Complementation of Bottlenecks: These limiting factors usually reside at different genetic loci in different inbred lines. When two such lines are hybridized, the hybrid inherits a “complementary” set of functional alleles. The favorable allele from one parent can “mask” or overcome the specific metabolic bottleneck of the other parent.
• Metabolic Establishment: This complementation results in a better-balanced metabolic system in the hybrid. By removing individual parental constraints, the hybrid achieves higher metabolic efficiency, leading to the increased biomass, speed of development, and yield characteristic of hybrid vigor.
• Systemic Resource Exploitation: From a systems biology perspective, this allows the hybrid to effectively “do more with less”. A hybrid can often exceed the performance of its best parent while using a lower total enzyme concentration, as its resources are distributed in a way that is closer to an optimal metabolic distribution.
5. How does Salicylic Acid coordination drive defense heterosis in hybrids?
Salicylic acid (SA) coordination drives defense heterosis through a complex interplay of temporal regulation, epigenetic modifications, and specific gene activation that allows hybrids to surge past their parents’ immune responses without sacrificing growth vigor.
The “Above-High-Parent” SA Surge
In hybrid combinations exhibiting defense heterosis, the F1 progeny display a unique hormonal response following a pathogen challenge. While typical hybrids may maintain lower SA levels to promote growth, those specialized for defense accumulate SA to levels exceeding those of both parents. This “above-high-parent SA surge” is indispensable for superior resistance; if SA biosynthesis is genetically blocked, the hybrid’s defense advantage vanishes.
Coordination via the Circadian Clock
To avoid the biological “cost” of high immunity—which often inhibits growth—hybrids use the circadian oscillator (specifically the transcription factor CCA1) to coordinate the timing of defense.
• Temporal Separation: CCA1 ensures that the high-parent activation of growth-related genes occurs during the daytime to maximize photosynthesis and biomass.
• Defense Activation: Conversely, it triggers the surge of SA-related immune genes at night or specifically after pathogen infection. This rhythmic regulation enables the hybrid to achieve both high yield and robust resistance simultaneously.
Epigenetic and Molecular Drivers
Several molecular mechanisms facilitate this coordinated SA response:
• Histone Acetylation (H3Ac): In Arabidopsis hybrids, H3Ac levels are significantly enhanced at the promoter regions of key SA biosynthetic genes. This modification, predominantly regulated by CCA1, de-represses these loci to allow for increased SA synthesis.
• Chromatin Remodeling (DDM1): The factor DDM1 (Decreased DNA Methylation 1) acts as a major regulator by locking the parental SA baseline into an optimal range, preventing constitutive SA accumulation that would otherwise be detrimental to biomass heterosis.
• Gene Activation Pathways: In specific cases, such as certain Arabidopsis and rice hybrids, the SA surge is supported by the activation of transcription factors like WRKY45-1 or genes like PAD4, which function upstream of the SA biosynthesis pathway to amplify the immune signal.
Hormone Crosstalk
SA-mediated defense heterosis is further shaped by its interactions with other hormones, particularly jasmonic acid (JA). While traditionally viewed as antagonistic, in heterotic hybrids, SA can activate JA signaling via specific receptors (NPR3/NPR4) to promote effector-triggered immunity, creating a multifaceted defense network that is more efficient than that found in either inbred parent.
6. How do hybrids use the circadian clock to manage trade-offs?
Hybrids use the circadian clock—specifically the core transcription factor CIRCADIAN CLOCK ASSOCIATED 1 (CCA1)—as a master regulator to coordinate the competing resource demands of growth and immunity. This temporal coordination allows the hybrid to achieve both biomass heterosis and defense heterosis simultaneously without the typical “vigor costs” associated with a heightened immune system.
Temporal Separation of Growth and Defense
The primary way the circadian clock manages these trade-offs is through temporal separation, ensuring that energy-intensive processes do not interfere with one another:
• Daytime Growth: CCA1 ensures that the “above-high-parent” expression of growth-related genes is prioritized during the daytime. This maximizes photosynthesis, metabolic flux, and starch accumulation when sunlight is available.
• Nighttime/Induced Defense: Conversely, the clock triggers the surge of salicylic acid (SA) and immune-related genes at night or specifically following a pathogen challenge. By restricting high levels of SA to these specific windows, the hybrid avoids the growth-inhibitory effects of constitutive immune activation.
Molecular and Epigenetic Mechanisms
Several molecular “switches” controlled by the clock facilitate this balance:
• Histone Acetylation (H3Ac): CCA1 regulates the rhythmic activation of SA biosynthetic genes by enhancing histone H3 acetylation levels at their promoter regions. This allows for a rapid surge in immunity when needed while keeping the genes repressed during peak growth hours.
• Chromatin Remodeling (DDM1): The chromatin remodeling factor DDM1 works in tandem with the clock to lock the hybrid’s baseline SA levels into an optimal range, preventing excessive accumulation that would be detrimental to biomass.
• DNA Methylation: In Arabidopsis hybrids, the parent-of-origin effect on CHH methylation in the CCA1 promoter region affects the amplitude of its expression. Higher methylation levels at these sites correlate with lower CCA1 peaks, which in turn leads to increased starch accumulation and biomass.
Species-Specific Strategies
The circadian clock manages these trade-offs differently depending on the species:
• Arabidopsis (Amplitude Model): In Arabidopsis hybrids and allotetraploids, the amplitude of the circadian rhythm is altered, but the phase remains the same. This increased amplitude up-regulates downstream genes in the chlorophyll and starch metabolic pathways.
• Maize (Phase-Shift Model): In maize, hybrids exhibit a temporal phase-shift in ZmCCA1 binding. The binding of ZmCCA1 to its target genes shifts to the early morning, causing non-additive expression of photosynthetic and metabolic genes that enhances carbon fixation efficiency.
7. Could you explain the gene balance hypothesis in polyploid plants?
The gene balance hypothesis, primarily developed by James Birchler and Reiner Veitia, provides a molecular framework to explain phenotypic patterns in polyploid plants that classical genetic theories (like simple dominance) cannot fully account for.
Core Premise: Dosage Sensitivity
The hypothesis suggests that regulatory genes—such as transcription factors, signal transduction components, and members of chromatin-remodeling complexes—often function as part of multisubunit macromolecular complexes. The proper functioning of these complexes depends on the stoichiometric balance (the specific ratio) of their different subunits.
• Sensitivity to Dosage: Because these regulatory components are dosage-sensitive, any change in the relative abundance of one subunit compared to others can alter the kinetics or assembly of the entire complex.
• Regulatory Cascades: Since these complexes often sit at the top of hierarchical regulatory cascades, a shift in their balance can trigger a “ripple effect,” changing the expression of a large number of downstream target genes.
Application to Polyploidy and Heterosis
The gene balance hypothesis is particularly useful for explaining observations in polyploids, such as progressive heterosis and allelic dosage effects:
• Progressive Heterosis: This refers to the phenomenon where hybrid vigor increases as more distinct genomes are added to a polyploid (e.g., a double-cross tetraploid ABCD performing better than a single-cross tetraploid AABB). Simple dominance models struggle to explain this, as the ratio of favorable to unfavorable alleles shouldn’t change with ploidy, but the gene balance hypothesis suggests that hybrids achieve a more favorable gene dosage balance across complex regulatory networks as genomic diversity increases.
• Dosage Components: Studies in tobacco and maize show that the magnitude of heterosis is sensitive to the number of copies of a genome (e.g., AABB plants are more vigorous than AAB plants). The hypothesis posits that hybrids may possess an optimal “dosage” of regulatory gene products that allows them to surpass the performance of their parents.
• Evolutionary Retention: This hypothesis is supported by the observation that regulatory genes and members of macromolecular complexes are preferentially retained following whole-genome duplication events to maintain proper dosage balance, whereas they are often depleted in small-scale copy-number variations.
In summary, while classical theories focus on specific “good” or “bad” alleles, the gene balance hypothesis emphasizes the systemic coordination of gene products, suggesting that superior hybrid performance in polyploids stems from a more equilibrated and efficient regulatory network.
8. Can hybrids maintain superior defense across generations?
In general, hybrids cannot maintain superior defense across generations through standard sexual reproduction because heterosis is maximal in the F1 generation and typically breaks down in the F2 and subsequent generations due to genetic segregation.
However, the sources outline several advanced biological and technical strategies that allow for the “fixation” or maintenance of these superior traits over time:
1. Synthetic Apomixis
This is a transformative approach that involves engineering plants to reproduce asexually through seeds.
• Mechanism: By mimicking a process called diplospory (producing embryos without meiosis or fertilization), offspring become genetically identical clones of the F1 parent.
• Outcome: This allows the superior high-yielding and disease-resistant traits of the hybrid to be maintained indefinitely across generations, offering a cost-effective alternative to purchasing new F1 seeds every season.
2. Polyploidy and Fixed Heterosis
Certain complex organisms, such as allohexaploid wheat, are considered to have “permanently fixed heterosis”.
• Mechanism: In nascent allohexaploid wheat, robust and heritable disease resistance (e.g., against powdery mildew) is maintained through specific regulatory circuits, such as expression-level dominance (ELD) of resistance genes and the non-additive repression of small RNAs (miRNAs).
• Outcome: These molecular mechanisms amplify defense signaling in a way that is stably transmitted during mitotic and meiotic divisions.
3. Hybrid Mimics
“Hybrid mimics” are pure-breeding lines developed to possess the phenotypic characteristics of a hybrid.
• Mechanism: Through repeated screening and selection of F2 lines that closely resemble the F1 phenotype, breeders can eventually develop stable F5 or F6 lines where the heterotic vigor—including growth and defense traits—is effectively “fixed” into a homozygous state.
4. Synthetic Supergenes
A modern genetic engineering strategy proposes recruiting multiple heterosis-associated genes to create supergenes.
• Mechanism: Multiple heterotic alleles are fused across different loci into a single genetic cassette.
• Outcome: This architecture ensures that favorable allele combinations are inherited as a single unit, preventing them from being dissociated by recombination and thus preserving the heterotic defense effects across generations.
5. Epigenetic Maintenance
Superior hybrid performance is also governed by a heritable layer of epigenetic information, such as DNA methylation and histone modifications. These modifications can be stably transmitted during mitotic and meiotic cell divisions, meaning that some aspects of the hybrid’s unique regulatory state may persist, though they are still generally subject to the broader pressures of genetic segregation unless one of the fixation methods above is employed.
9. What is Single-Parent Expression and how does it affect yield?
Single-parent expression (SPE) is an extreme type of transcriptome-wide variation where a gene, although present in the genomes of both parents, is transcriptionally active in only one parent and the hybrid offspring. This phenomenon results in expression complementation, where the active allele from one parent compensates for the silent allele of the other in the hybrid.
Characteristics and Molecular Basis of SPE
• Prevalence: In maize hybrids, approximately 1,000 genes have been identified as active in the hybrid while remaining silent in one of the two parental inbred lines.
• Genetic Mechanism: While some SPE patterns are caused by genomic Presence-Absence Variation (PAV)—where a gene is physically missing from one parent—this only accounts for about 4% to 18% of such cases. The majority of these genes are present in both parents, suggesting that variations in regulatory sequences (such as SNPs or insertions/deletions) cause the gene to be silenced in one parent.
• Gene Types: SPE is particularly prevalent among evolutionarily younger, non-syntenic genes—those that emerged after the evolutionary separation of the maize and sorghum lineages. These genes are often involved in environmental adaptation.
10. How SPE Affects Yield and Performance
SPE influences hybrid vigor by increasing the total number of active genes and optimizing the hybrid’s response to its environment:
• Transcriptional Complementation: As a result of SPE, hybrids express an average of 600 additional genes compared to the average of their parental inbred lines. This extensive complementation aligns with the dominance model of heterosis, where favorable active alleles mask the effects of unfavorable silent ones.
• Correlation with Adult Traits: Research has demonstrated that the number of SPE genes expressed in seedling roots is significantly related to phenotypic heterosis for several critical adult traits, including ear size, plant height, tassel size, and leaf size.
• Environmental Adaptability: SPE patterns appear to be highly stable, even under stress conditions like water deficit. The activity of these additional genes is hypothesized to provide hybrids with a superior ability to adapt to fluctuating environments compared to their inbred parents.
• Coordination of Growth: Because these patterns are highly dynamic and tissue-specific (with roughly 80% tissue specificity), they allow the hybrid to fine-tune its genetic “inventory” at different developmental stages to maximize productivity.
11. Why do some crops benefit more from heterosis than others?
The degree of heterosis, or hybrid vigor, is not uniform across the plant kingdom; its magnitude varies significantly depending on a crop’s reproductive mode, genetic diversity, ploidy level, and specific genomic architecture.
1. Reproductive Mode (Pollination Type)
The most fundamental reason for varying heterosis levels is whether a species is naturally cross-pollinating (allogamous) or self-pollinating (autogamous).
• Outbreeders vs. Inbreeders: Species that naturally cross-pollinate, such as maize, rye, and alfalfa, typically display a much higher degree of heterosis than self-pollinating species like wheat, rice, and barley.
• Inbreeding Tolerance: Allogamous plants are highly sensitive to inbreeding depression—the loss of vigor during self-pollination—which makes the recovery of that vigor through hybridization much more dramatic. Conversely, autogamous species are more tolerant of inbreeding and therefore show a smaller heterotic response upon crossing.
2. Genetic Distance and Divergence
The magnitude of heterosis is generally proportional to the genetic distance between the parental genotypes.
• Heterotic Groups: Crops where breeders have successfully established distinct heterotic groups (genetically divergent populations) benefit more because crossing individuals from different groups maximizes favorable allelic interactions.
• Intersubspecific and Wide Hybridization: Hybrids resulting from greater genetic disparity often show more spectacular vigor. For example, in rice, intersubspecific hybrids (e.g., indica × japonica) can display 8–15% more heterotic potential than intraspecific hybrids.
3. Ploidy Level and Progressive Heterosis
Polyploid plants (those with more than two sets of chromosomes) can exhibit a unique phenomenon known as progressive heterosis.
• Genome Diversity: In polyploids, the magnitude of heterosis often increases with the number of distinct genomes added.
• Double Crosses: For example, a “double-cross” tetraploid hybrid (ABCD) that combines four diverse parental genomes typically exhibits greater heterosis than a single-cross tetraploid (AABB).
4. Genomic and Transcriptomic Complementation
Some crops, particularly maize, have genomic structures that are uniquely suited to benefit from heterosis through complementation.
• Structural Variation: Maize is characterized by a high degree of non-colinearity, meaning many genes present in one inbred parent are entirely absent in the other. In the hybrid, these genes are complemented, leading to significantly increased performance.
• Single-Parent Expression (SPE): This complementation also occurs at the expression level. In maize hybrids, hundreds of genes are active that were silent in one of the parents, a mechanism that is significantly related to phenotypic heterosis for traits like plant height and ear size.
5. Trait Complexity and Environment
• Trait Specificity: The relative amount of heterosis increases with the complexity of the trait. For instance, complex quantitative traits like grain yield generally show much higher heterosis than simpler traits like fruit weight.
• Environmental Sensitivity: Heterosis is not a fixed constant but is dynamically influenced by the environment. Some crops benefit more in specific environments because their hybrids are better at buffering against environmental stress or adapting to fluctuating conditions compared to their inbred parents.
12. Can synthetic supergenes effectively fix yield and defense traits?
Synthetic supergenes represent a promising, though still emerging, strategy to effectively fix yield and defense traits that are typically limited to the F1 hybrid generation. This approach leverages modern genetic engineering to bypass the “hybrid breakdown” that occurs in subsequent generations due to genetic segregation.
Mechanism of Synthetic Supergenes
The primary goal of creating a synthetic supergene is to permanently capture heterosis (hybrid vigor) within homozygous inbred lines.
• Fusion of Alleles: Multiple heterotic alleles from different loci are fused linearly or in tandem into a single genetic cassette.
• Complete Linkage: By grouping these alleles together, they are inherited as a single unit. This prevents the favorable allele combinations from being dissociated by recombination during reproduction.
• Preservation of Heterotic Effects: This architecture is designed to maintain the beneficial interactions—including dominance, overdominance, and epistasis—that characterize superior hybrid performance.
Effectiveness for Yield and Defense
The sources provide both empirical evidence for yield and theoretical frameworks for defense:
• Yield Fixation: In maize, the fusion of a pair of heterotic alleles of the ZAR1 gene resulted in a consistent and stable yield advantage over the homozygous forms of either individual allele across diverse environments.
• Defense Fixation: While yield has been the primary focus, the strategy is being expanded to defense traits. Genes like CCA1, which coordinates both growth and disease resistance, are identified as prime candidates for recruitment into synthetic supergenes.
• Precise Engineering: Synthetic biology strategies can be used to engineer modules that fine-tune salicylic acid (SA) dynamics, potentially conferring broad-spectrum, durable disease resistance while maintaining high, stable yields.
Feasibility and Challenges
While the potential is transformational, especially for self-pollinating crops like wheat and barley, several challenges remain:
• Locus Identification: It is historically difficult to identify and validate a sufficient number of heterosis-associated loci that together contribute significantly to increased performance.
• Functional Viability: Researchers must determine if fusing these alleles and their regulatory elements can be done without negatively impacting their individual functions or the resulting heterotic effect.
• Model Testing: Current efforts are focused on using barley as a model crop due to its genetic relationship with wheat and its amenability to high-efficiency genetic transformation.
13. Why do cross-pollinating crops show higher heterosis than self-pollinating ones?
Cross-pollinating (allogamous) crops show significantly higher levels of heterosis than self-pollinating (autogamous) ones primarily because of their natural genetic structure, their heavy load of deleterious recessive alleles, and their differing sensitivity to inbreeding depression.
The sources highlight several key reasons for this disparity:
1. Sensitivity to Inbreeding Depression
The degree of heterosis is functionally the opposite of inbreeding depression—the loss of vigor that occurs during self-pollination.
• Outbreeders: Species like maize, rye, and alfalfa are naturally cross-pollinated and are highly sensitive to inbreeding depression. When these plants are forced to inbreed, their stature, vigor, and yield decline severely.
• Inbreeders: Species like wheat, barley, and oats have evolved reproductive mechanisms for self-pollination and are highly tolerant of inbreeding. Because they suffer minimal vigor loss during selfing, the “recovery” of vigor through hybridization is much less dramatic.
2. Presence of Deleterious Recessive Alleles
The “genetic load” of a species—the number of harmful recessive alleles hidden in its genome—is a major driver of heterosis under the dominance hypothesis.
• Masking in Cross-Pollinators: In cross-pollinating species, deleterious alleles are typically maintained in a heterozygous state, where they are masked by favorable dominant alleles. Because these populations tolerate the presence of these alleles, there are many opportunities for dominance complementation to occur when two genetically distinct parents are crossed, resulting in superior hybrid performance.
• Elimination in Self-Pollinators: Because self-pollinating plants naturally approach homozygosity over generations, they tend to eliminate (purge) deleterious recessive alleles over time. With fewer harmful alleles left to mask, the potential for heterosis through complementation is substantially reduced.
3. Differences in Heterozygote Advantage
The sources note that the genetic strategies for fitness differ between the two types of crops:
• Heterozygote Advantage: Outbreeding species tend to exhibit more heterozygote advantage (overdominance) and complex epistatic interactions that are only present in a heterozygous state.
• Homozygous Tolerance: Inbreeding species approach homozygosity naturally; their variability is distributed mostly between component lines rather than within the whole population. Consequently, they rely less on heterozygosity per se for their fitness.
4. Genetic Distance and Divergence
The magnitude of heterosis is generally proportional to the genetic distance between parental genotypes. Cross-pollinating species typically maintain higher levels of intraspecific genetic diversity and structural variation than self-pollinating species. When two divergent heterotic groups of an outcrossing species are crossed, the accumulation of favorable alleles and the masking of deleterious ones produce the “spectacular” vigor often seen in crops like maize.
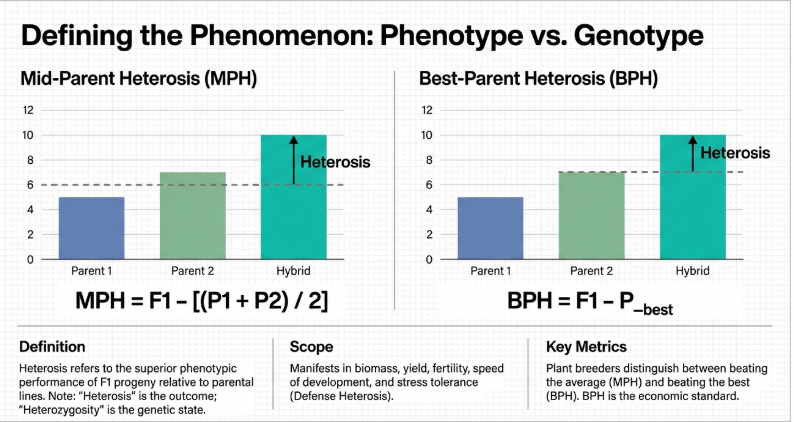
14. Can you explain the difference between midparent and best-parent heterosis?
The primary difference between midparent heterosis and best-parent heterosis (also referred to as high-parent heterosis or heterobeltiosis) lies in the reference point used to calculate the hybrid’s superior performance.
Midparent Heterosis (MPH)
• Definition: MPH measures the performance of a hybrid (F1) relative to the average performance (mean) of its two parents.
• Biological Significance: It is often considered the simplest model for estimating heterosis and is used by scientists to detect non-additive gene action.
• Calculation:
◦ Absolute MPH: F1−Midparent Value (MPV), where MPV is (P1+P2)/2.
◦ Relative MPH: 100×(F1−MPV)/MPV.
• Inheritance Patterns: A hybrid exhibits “positive MPH” if its performance falls anywhere between the midparent value and the value of the best parent.
Best-Parent Heterosis (BPH)
• Definition: BPH measures the performance of a hybrid relative to the better-performing parent (the “high parent”) for a specific trait.
• Agronomic Significance: BPH is the most relevant measure for plant breeding and agriculture because it determines if a new hybrid actually surpasses the best currently available inbred lines.
• Calculation:
◦ Absolute BPH: F1−Pmax, where Pmax is the higher-performing parent.
◦ Relative BPH: 100×(F1−Pmax)/Pmax.
• Inheritance Patterns: For BPH to occur, the hybrid must strictly exceed the maximum value of either parent.
Key Comparisons
| Feature | Midparent Heterosis (MPH) | Best-Parent Heterosis (BPH) |
|---|---|---|
| Reference Point | Average of both parents. | The superior/better parent. |
| Utility | Used to study genetic interactions and “dosage” effects. | Used to evaluate commercial value and yield advantage. |
| Magnitude | Generally higher than BPH because the average is lower than the best parent. | More difficult to achieve; reflects “spectacular” vigor. |
| Prediction | Theoretically a quadratic function of parental genetic distance. | Harder to predict; requires specific favorable allele combinations. |
In practical terms, a hybrid can exhibit significant midparent heterosis without ever being “better” than its best parent, simply by performing better than the average of two divergent parents. However, for a hybrid to be agronomically successful, it typically must demonstrate best-parent heterosis to justify the higher cost of hybrid seed production.
15. How can we develop more vigorous and stable inbred lines?
Developing more vigorous and stable inbred lines involves a combination of traditional population improvement techniques, the purging of deleterious genetic loads, and the application of advanced molecular tools to “fix” beneficial traits that are typically found only in hybrids.
1. Purging Deleterious Alleles through Selection
The most fundamental method for improving inbred vigor is the systematic removal of deleterious recessive alleles (genetic load) through repeated cycles of self-pollination and stringent selection.
• Inbreeding and Purging: While self-pollination naturally causes inbreeding depression, it also exposes harmful recessive alleles that were previously masked in a heterozygous state. By selecting only the most vigorous individuals in each generation, breeders can purge these “detrimental” factors from the germplasm.
• Fixing Favorable Alleles: The dominance hypothesis predicts that if all deleterious alleles are eliminated and replaced by favorable dominant counterparts, it would be possible to develop an inbred line with performance equal to that of a hybrid.
2. Recurrent Selection for Population Improvement
Rather than focusing on individual lines in isolation, recurrent selection is used to improve the entire base population from which future inbreds are derived.
• Incremental Gene Frequency Shifts: This process involves repeated cycles of selection and recombination to induce small, repetitive increases in the frequency of favorable alleles within a population.
• Intra-Population Improvement: Methods such as S1 or S2 selection focus on the additive genetic effects within a group, ensuring that the mean performance of lines subsequently derived from these populations is significantly higher.
3. Breaking Repulsion-Phase Linkages
A significant bottleneck in inbred development is the presence of favorable alleles linked in repulsion phase with deleterious ones, particularly in “recombination deserts” like pericentromeric regions.
• Targeted Recombination: Inbred yields can be increased by establishing selection programs that specifically identify and recombine plants that have experienced rare recombination events in these regions.
• Fixed Heterosis: Breaking these linkages makes it possible to develop inbreds that contain favorable alleles at both linked loci, effectively “fixing” some of the yield advantage traditionally provided by the hybrid state.
4. Molecular and Genomic Strategies
Modern breeding leverages big data to move beyond the “black box” of traditional phenotypic selection.
• Genomic Selection (GS) and GWAS: Researchers use Genome-Wide Association Studies to identify key heterotic QTLs and alleles. Once these loci are identified, GS models can predict the performance of candidate lines, accelerating the accumulation of superior alleles.
• Molecular Design Breeding (MDB): This strategy involves selecting optimal haplotype combinations of genes known to interact favorably. For example, the “ZAR1” pairing in maize demonstrated that fusing two heterotic alleles can result in a stable yield advantage in a homozygous state.
• Gene Editing: Technologies like CRISPR/Cas9 can be used to precisely modify or knock out unfavorable alleles, or to create “synthetic variation” in key regulatory genes to fine-tune plant architecture and vigor.
5. Developing “Hybrid Mimics” and Synthetic Supergenes
To achieve the stability of an inbred with the vigor of a hybrid, breeders are exploring methods to capture heterotic effects permanently.
• Hybrid Mimics: These are pure-breeding, homozygous lines developed through repeated screening of F2 generations to find individuals that closely resemble the F1 phenotype.
• Synthetic Supergenes: This approach involves fusing multiple heterotic alleles from different loci into a single genetic unit. Grouping these alleles ensures they are inherited together without being dissociated by recombination, preserving beneficial dominance and epistatic interactions across generations.
• Synthetic Apomixis: Engineering plants to reproduce asexually through seeds allows a high-performing F1 genotype to be cloned and maintained indefinitely as a stable line.
16. Does genetic distance between parents always lead to higher yield?
While genetic distance is a primary driver of heterosis, it does not always lead to a higher yield, as the relationship is governed by an optimal range of divergence and is influenced by genomic stability.
The General Correlation and Its Limits
The magnitude of heterosis is generally proportional to the genetic distance (GD) between parental genotypes. However, while genetic diversity is a necessary prerequisite, it is not by itself sufficient to guarantee superior hybrid performance.
• The Optimum Distance Hypothesis: Under simple genetic models, heterosis is theoretically a quadratic function of parental GD. Research in crops like maize has identified an optimum level of parental genetic distance, beyond which the degree of heterosis and actual hybrid performance begin to decline.
• Declines at Extreme Disparity: When parents are extremely divergent, such as in some wide-hybridization or interspecific crosses, the resulting hybrids may face “genomic shock,” leading to genomic instability, poor seed setting, and infertility. For example, despite strong vegetative vigor, indica × japonica rice hybrids often suffer from low and unstable seed-set due to homologous chromosome pairing issues.
Inconsistencies in Prediction
The ability to use genetic distance to predict yield is highly dependent on the germplasm and the specific markers used.
• Lack of Correlation: Several empirical studies in Arabidopsis, maize, and rice have reported that genetic distance determined by molecular markers is not always significantly associated with the level of mid-parent heterosis.
• Marker Specificity: Genetic distance measurements alone are often insufficient for accurate prediction because they typically reflect overall genomic similarity rather than the specific allelic variation at quantitative trait loci (QTLs) that actually govern yield. Predictions become much more reliable only when a significant portion of the selected markers are specifically linked to these yield-influencing loci.
Complexity of Gene Action
The influence of distance on yield is further complicated by the type of gene action involved:
• Additive vs. Non-Additive Effects: While dominance (complementation) often follows a linear relationship with distance, epistatic interactions (interactions between non-allelic genes) can create non-linear responses.
• Purging of Deleterious Alleles: In elite breeding programs, many major-effect deleterious alleles have already been purged from the parents. Consequently, the remaining heterosis often results from a large number of minor-effect loci, making the yield response to increased genetic distance less dramatic and more difficult to predict than in unselected populations.
17. What role does the microbiome play in boosting hybrid vigor?
The microbiome, which consists of complex microbial communities in habitats like the rhizosphere, functions as a “second genome” or an extension of the plant’s own genomic features. Recent research reveals that hybrid vigor is not solely a result of plant genetics but is significantly “microbe-dependent” in certain systems.
1. Driving Biomass Heterosis
In crops such as maize, the manifestation of biomass heterosis is dependent on the composition of specific rhizosphere microbial communities. Experiments have demonstrated that in sterile soil conditions, hybrids may fail to perform better than their inbred parents; however, their superior biomass is restored once the plants are inoculated with beneficial bacteria.
2. Recruitment of Functional Microbes
Hybrids consistently differ from their parental lines in their microbiome composition, often recruiting microbial partners that provide specific advantages. This recruitment is an inherited trait regulated by heterosis:
• Nutrient and Growth Promotion: Hybrids more effectively recruit beneficial fungal symbionts and auxin-producing bacteria (such as Pseudomonas) that enhance growth.
• Pathogen Inhibition: Heterosis in root microbiota has been shown to inhibit the growth of soil-borne fungal pathogens, effectively boosting the hybrid’s natural defenses.
• Seed Vitality: In hybrid rice, endophytic microbiomes within the seeds have been found to improve germination rates compared to parental lines.
3. Heritability and Adaptation
The structure and function of the plant microbiome are considered heritable traits in cereal species, including maize, sorghum, and foxtail millet. This allows hybrids to maintain a consistent microbial “extension” that aids in environmental adaptation and yield stability across fluctuating soil conditions.
4. Future Ecological Strategies
Because the rhizosphere microbiota significantly influences the level of heterosis—and could even reverse it into a “hybrid disadvantage” if the community structure is poor—researchers are looking toward microbiome manipulation as an ecological solution. Screening for specific combinations of microbiota that enhance defense and growth offers a new way to predict and deploy heterosis at the agroecosystem level.
18. What are ‘hybrid mimics’ and how are they developed?
Hybrid mimics (also called hybrid simulation lines) are pure-breeding, homozygous lines that possess the superior phenotypic characteristics of an F1 hybrid. Unlike typical hybrids, which lose their vigor in the F2 and subsequent generations due to genetic segregation, hybrid mimics “fix” heterosis so that it is stably maintained over time.
Development Process
The development of hybrid mimics relies on a multi-generational screening and selection strategy:
1. Selection of a Heterotic F1: The process begins with a high-performing F1 hybrid that exhibits significant hybrid vigor.
2. Creation of Segregating Generations: The F1 is self-pollinated to produce a large, genetically diverse F2 population.
3. Repeated Screening and Selection: Breeders repeatedly screen the F2 and subsequent generations for individuals that most closely resemble the original F1 phenotype (e.g., in terms of vigor, yield, or plant architecture).
4. Fixation of the Phenotype: By continuing this stringent selection process through the F5 or F6 generations, researchers can obtain pure-breeding lines where the heterotic phenotype is fixed into a homozygous state.
Demonstrated Success
This strategy has been successfully used to maintain F1 heterosis in several plant species, including:
• Arabidopsis thaliana
• Tomato
• Pea
Developing hybrid mimics is considered a challenging but potentially transformative strategy for increasing global crop productivity, as it eliminates the need for farmers to purchase new F1 seeds every season.
19. How does sterile soil specifically impair hybrid performance?
Sterile soil specifically impairs hybrid performance by preventing the expression of microbe-dependent heterosis, a phenomenon in which the superior performance of a hybrid is tied to its interaction with soil-borne microorganisms.
Research indicates that in certain systems, such as maize, biomass heterosis is not purely a result of plant genetics but is dependent on the composition of specific microbial communities in the rhizosphere. Under sterile soil conditions, studies have shown that hybrids may perform worse than their inbred parents, effectively losing their expected vigor. Furthermore, the absence or alteration of the structure of the rhizosphere microbiota can significantly decrease the level of heterosis and may even reverse it into a “hybrid disadvantage”.
The impairment occurs because hybrids rely on the recruitment of specific beneficial bacteria to optimize traits such as nutrient uptake and biomass accumulation. This heterotic advantage is only restored after the plants are inoculated with these beneficial microbes, highlighting that the soil microbiome acts as a necessary extension of the hybrid’s genomic potential.
20. What are the biggest challenges in identifying heterosis-associated loci?
Identifying heterosis-associated loci is a significant challenge in modern genetics because hybrid vigor is a complex, polygenic phenomenon that involves intricate interactions between different genetic backgrounds and environmental factors.
The biggest challenges identified in the sources include:
1. Distinguishing Overdominance from Pseudo-overdominance
A major historical bottleneck is the inability to reliably distinguish true overdominance (where a single heterozygous locus is superior) from pseudo-overdominance (where two closely linked dominant alleles are in a repulsion-phase linkage). Without extensive recombinational separation or actual cloning of the Quantitative Trait Loci (QTLs), these two mechanisms appear identical in traditional genetic mapping studies.
2. Detection of Complex Epistatic Interactions
While epistasis (interactions between non-allelic genes) is increasingly recognized as a primary driver of heterosis, it is exceptionally difficult to quantify.
• Computational Burden: Testing all possible SNP-SNP interactions creates an “astronomical” computational load that often forces researchers to focus only on loci with significant additive effects, potentially missing key interacting pairs.
• Population Size: Traditional bi-parental populations often lack the statistical power to detect epistasis because interacting loci must be segregating simultaneously within that specific population to be detected.
• Genetic Repression: Some favorable alleles are epistatically repressed in the parental lines (making them invisible to GWAS in the parents) and are only “activated” in the hybrid when the paternal genome complements the maternal repressive allele.
3. Phenotypic Complexity and Low Heritability
Heterosis is often a “multiplicative” or “geometric” outcome of multiple, potentially unrelated quantitative traits (such as growth rate, flowering time, and seed number). Because it represents the ultimate output of the entire plant system, the heritability of heterosis is relatively low compared to more discrete phenotypes like fruit weight or sugar content, making it difficult to map with high resolution.
4. Subtle Effects of Minor Alleles
Variants influencing heterosis often have minor effects on morphology, which complicates their identification and isolation, especially in the relatively small populations used in most studies. Massive population sizes or advanced genomic selection (G2P) models are required to detect these subtle QTNs (quantitative trait nucleotides).
5. Identifying Causality in Transcriptome Data
While high-throughput sequencing can identify thousands of differentially expressed genes (DEGs) in hybrids, it is extremely difficult to separate the molecular causes of heterosis from its downstream effects. Many expression differences observed in hybrids may simply be a biological response to the hybrid’s increased growth vigor rather than the original genetic trigger for that growth.
6. Genomic and Population Specificity
Heterosis is highly trait-specific and population-specific, meaning a gene block that contributes to vigor in one hybrid combination may not have the same effect in another. This lack of consistency across different genetic backgrounds makes it difficult to identify a “consensus” set of heterosis genes. Furthermore, a significant loss of colinearity (where genes are physically missing in one parent) complicates the identification of corresponding loci across divergent parental lines.
References
Aguilar, F. S., Lamkey, K. R., & Edwards, J. W. (2026). Dual genetic mechanisms of heterosis: population structure and gene action. Frontiers in Plant Science, 16. https://doi.org/10.3389/fpls.2025.1715826
Barth, S., Busimi, A. K., Utz, H. F., & Melchinger, A. E. (2003). Heterosis for biomass yield and related traits in five hybrids of Arabidopsis thaliana L. Heynh. Heredity, 91(1), 36–42. https://doi.org/10.1038/sj.hdy.6800276
Birchler, J. A. (2015). Heterosis: The genetic basis of hybrid vigour. In Nature Plants (Vol. 1). Palgrave Macmillan Ltd. https://doi.org/10.1038/nplants.2015.20
Birchler, J. A., Yao, H., Chudalayandi, S., Vaiman, D., & Veitia, R. A. (2010). Heterosis. Plant Cell, 22(7), 2105–2112. https://doi.org/10.1105/tpc.110.076133
Comings, D. E., & MacMurray, J. P. (2000). Molecular heterosis: A review. Molecular Genetics and Metabolism, 71(1–2), 19–31. https://doi.org/10.1006/mgme.2000.3015
Fiévet, J. B., Nidelet, T., Dillmann, C., & de Vienne, D. (2018). Heterosis is a systemic property emerging from non-linear genotype-phenotype relationships: Evidence from in vitro genetics and computer simulations. Frontiers in Genetics, 9(MAY). https://doi.org/10.3389/fgene.2018.00159
Fu, D., Xiao, M., Hayward, A., Fu, Y., Liu, G., Jiang, G., & Zhang, H. (2014). Utilization of crop heterosis: A review. In Euphytica (Vol. 197, Number 2, pp. 161–173). Kluwer Academic Publishers. https://doi.org/10.1007/s10681-014-1103-7
Fujimoto, R., Uezono, K., Ishikura, W., Osabe, K., Peacock, W. J., & Dennis, E. S. (2018). Recent research on the mechanism of heterosis is important for crop and vegetable breeding systems. Breeding Science, 68(2), 145–158. https://doi.org/10.1270/jsbbs.17155
Hochholdinger, F., & Baldauf, J. A. (2018). Heterosis in plants. In Current Biology (Vol. 28, Number 18, pp. R1089–R1092). Cell Press. https://doi.org/10.1016/j.cub.2018.06.041
Hochholdinger, F., & Hoecker, N. (2007). Towards the molecular basis of heterosis. In Trends in Plant Science (Vol. 12, Number 9, pp. 427–432). https://doi.org/10.1016/j.tplants.2007.08.005
Hochholdinger, F., & Yu, P. (2025). Molecular concepts to explain heterosis in crops. In Trends in Plant Science (Vol. 30, Number 1, pp. 95–104). Elsevier Ltd. https://doi.org/10.1016/j.tplants.2024.07.018
Jain, H. K., Kharkwal, M. C., Pandey-, M., & Jun Xu, W. (2004). Plant Breeding-Mendelian to MolecularApproaches Classical and Molecular Concepts of Heterosis.
Li, Z., Zhao, Y., & Luo, K. (2024). Molecular Mechanisms of Heterosis and Its Applications in Tree Breeding: Progress and Perspectives. In International Journal of Molecular Sciences (Vol. 25, Number 22). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/ijms252212344
Lippman, Z. B., & Zamir, D. (2007). Heterosis: revisiting the magic. In Trends in Genetics (Vol. 23, Number 2, pp. 60–66). https://doi.org/10.1016/j.tig.2006.12.006
Paril, J., Reif, J., Fournier-Level, A., & Pourkheirandish, M. (2024). Heterosis in crop improvement. Plant Journal, 117(1), 23–32. https://doi.org/10.1111/tpj.16488
Ren, H., & Sundaresan, V. (2026). Strategies and challenges for synthetic apomixis. In Current Opinion in Biotechnology (Vol. 97). Elsevier Ltd. https://doi.org/10.1016/j.copbio.2025.103433
Schnable, P. S., & Springer, N. M. (2013). Progress toward understanding heterosis in crop plants. In Annual Review of Plant Biology (Vol. 64, pp. 71–88). https://doi.org/10.1146/annurev-arplant-042110-103827
Sprague, G. F. (1983). Chapter 2 Heterosis in Maize: Theory and Practice. In Monographs on Theoretical and Applied Genetics (Vol. 6).
William, R. L., & Pollak, E. (1985). Theory of Heterosis. Journal of Dairy Science, 68(9), 2411–2417. https://doi.org/10.3168/jds.S0022-0302(85)81117-6
Xiao, Y., Jiang, S., Cheng, Q., Wang, X., Yan, J., Zhang, R., Qiao, F., Ma, C., Luo, J., Li, W., Liu, H., Yang, W., Song, W., Meng, Y., Warburton, M. L., Zhao, J., Wang, X., & Yan, J. (2021). The genetic mechanism of heterosis utilization in maize improvement. Genome Biology, 22(1). https://doi.org/10.1186/s13059-021-02370-7
Xu, K., Li, X., & Li, F. (2026). Defense heterosis as a novel plant protection strategy: From theory to breeding practice. New Plant Protection. https://doi.org/10.1002/npp2.70035
Yu, D., Gu, X., Zhang, S., Dong, S., Miao, H., Gebretsadik, K., & Bo, K. (2021). Molecular basis of heterosis and related breeding strategies reveal its importance in vegetable breeding. In Horticulture Research (Vol. 8, Number 1). Springer Nature. https://doi.org/10.1038/s41438-021-00552-9







Pingback: Decoding Hybrid Success: A Student’s Guide to GCA and SCA in Plant Breeding - Aneknowledge.com