1. Introduction: The Strategic Imperative of Distant Hybridization
Distant hybridization serves as a primary catalyst for genetic innovation by facilitating the exchange of genomic material across traditionally restrictive biological divides. By engaging in interspecific hybridization (between species within the same genus) or intergeneric hybridization (between different genera), breeders can effectively shatter species boundaries that limit the potential of domesticated crops. This process is essential for revitalizing narrow germplasm resources, allowing for the systematic expansion of genetic diversity and the introgression of novel physiological traits. Navigating these taxonomic gradients is not merely a classification exercise; it requires a surgical approach to the reproductive barriers that have served as evolutionary safeguards for millennia.
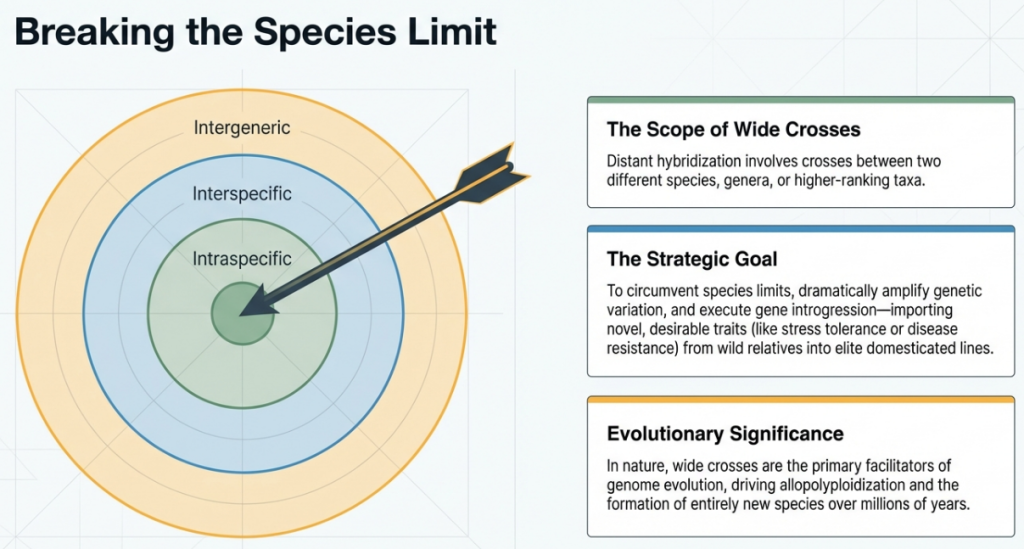
Defining the Field: Distant vs. Traditional Hybridization
In the discipline of plant genetics, hybridization serves as the primary engine for generating novel trait combinations. While traditional breeding focuses on crosses between closely related varieties, distant hybridization—also known as “wide crossing”—seeks to bypass natural reproductive boundaries by mating individuals that are taxonomically disparate. These crosses typically occur between different species within a genus (interspecific) or between different genera within a family (intergeneric).
The success of these efforts is governed by the Taxonomic Distance Rule: the further apart two parental lines are on the phylogenetic tree, the more robust the biological “safety locks” preventing a successful union.
| Type of Hybridization | Description | Botanical Example |
|---|---|---|
| Intraspecific (Traditional) | Breeding between varieties within the same species. | Solanum lycopersicum var. A × S. lycopersicum var. B |
| Interspecific (Intrageneric) | Crossing two different species within the same genus. | Solanum lycopersicum (Domesticated) × Solanum pennellii (Wild) |
| Intergeneric | Crossing species from two different genera within the same family. | Triticum aestivum (Wheat) × Secale cereale (Rye) |
Research focuses on four primary goals of wide hybridization:
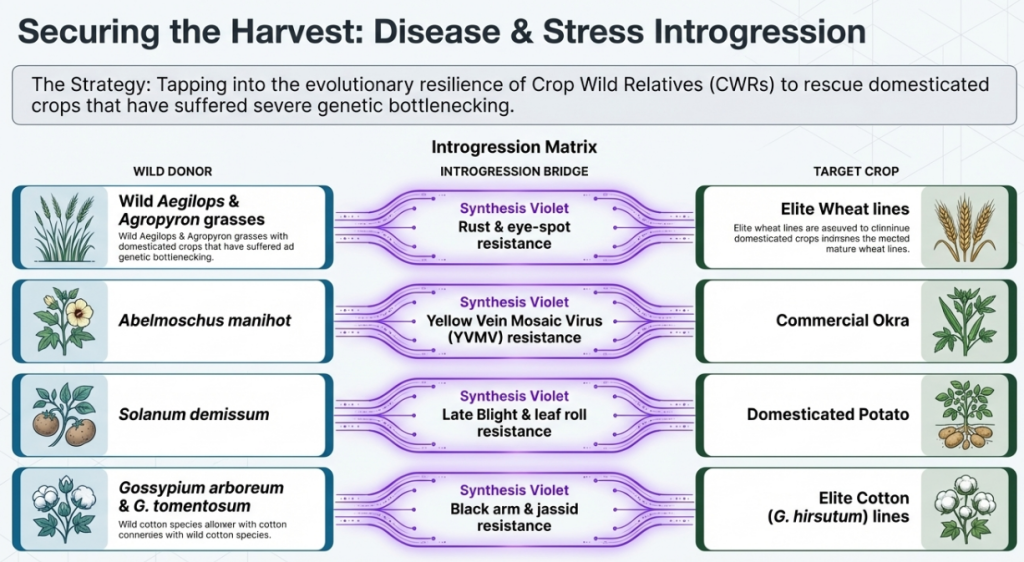
- Disease and Insect Resistance: Wild relatives are reservoirs for “R genes.” For instance, the Bs4 gene provides resistance against Xanthomonas campestris, while the Prf gene offers protection against P. syringae in tomatoes. Similar successes include transferring Jassid resistance from G. tomentosum to cultivated cotton.
- Quality and Nutritional Parameters: Hybridization can “upgrade” chemical profiles. Crosses have been used to enhance protein quality in rice and oats, and to significantly elevate vitamin content, such as increasing ascorbic acid levels in tomatoes through L. peruvianum germplasm.
- Yield Improvement: Beyond simple growth, wide crosses introduce novel genetic interactions that can boost productivity. In specific oat trials, the inclusion of Avena sterilis genetics resulted in a 25–30% yield increase over traditional varieties.
- Environmental Adaptation: To counter climate change, geneticists look to wild species for “extremophile” traits. This includes sourcing drought and salinity tolerance from Solanum pennellii or cold resistance from Lycopersicon hirsutum.
While these genetic matches offer immense rewards, nature has installed significant biological reproductive barriers to maintain species integrity. Navigating these obstacles requires a deep understanding of both pre- and post-fertilization mechanics.
2. Conceptual Framework: Categorizing Distant Hybridization
The efficacy of a breeding program is often dictated by the taxonomic distance between parental lines, as this distance influences both the mechanical strategy required for the cross and the biological viability of the resulting offspring. While intrageneric crosses are more common and often result in higher fertility, intergeneric crosses represent a more radical leap, often requiring advanced interventions to overcome inherent sterility through allopolyploidization.
Intrageneric vs. Intergeneric Hybridization: A Comparative Analysis
| Parameter | Intrageneric (Interspecific) Hybridization | Intergeneric Hybridization |
|---|---|---|
| Taxonomic Scope | Crossing distinct species within the same genus. | Crossing species belonging to different genera within the same family. |
| Potential for Fertility | Ranges from completely fertile to fully sterile depending on structural homology. | Offspring are almost universally sterile; requires induction of amphidiploidy for fertility. |
| Primary Purpose | Transferring specific traits (e.g., disease resistance) from wild relatives to cultivars. | Creation of entirely new crop species or major germplasm overhauls. |
| Historical Examples | Upland cotton derivatives (MCU-2, MCU-5); Prabhani Kranti (Okra). | Triticale (Wheat x Rye); Raphanobrassica (Radish x Cabbage). |
Understanding these categories is the first step toward addressing the sophisticated reproductive isolation mechanisms that modern breeders must navigate.
The Interspecific Masterpiece: Understanding the Brassica Triangle
The Brassica Triangle, first modeled by U. Nagaharu in 1934, serves as the definitive curriculum model for natural speciation through hybridization. It demonstrates how three ancestral diploid “cornerstone” species combined their genomes to form the tetraploid “derivative” crops that dominate modern agriculture.
- The Cornerstones (Diploids):
- B. nigra (Black Mustard, n=8,2n=16)
- B. oleracea (Cabbage/Broccoli, n=9,2n=18)
- B. campestris (Rapeseed/Turnip, n=10,2n=20)
- The Derivatives (Tetraploids):
- B. carinata (Ethiopian Mustard, 2n=34): A genomic combination of n=8 and n=9.
- B. juncea (Leaf Mustard, 2n=36): A genomic combination of n=8 and n=10.
- B. napus (Oilseed Rape, 2n=38): A genomic combination of n=9 and n=10.
So What? for the Learner: Interspecific hybrids are typically sterile because they lack Homology—the structural similarity between chromosomes required for pairing during meiosis. The Brassica Triangle is a masterpiece of evolution because it demonstrates how polyploidy (chromosome doubling) restores fertility. By providing each chromosome with a perfect homologous partner, the plant can successfully produce functional gametes.
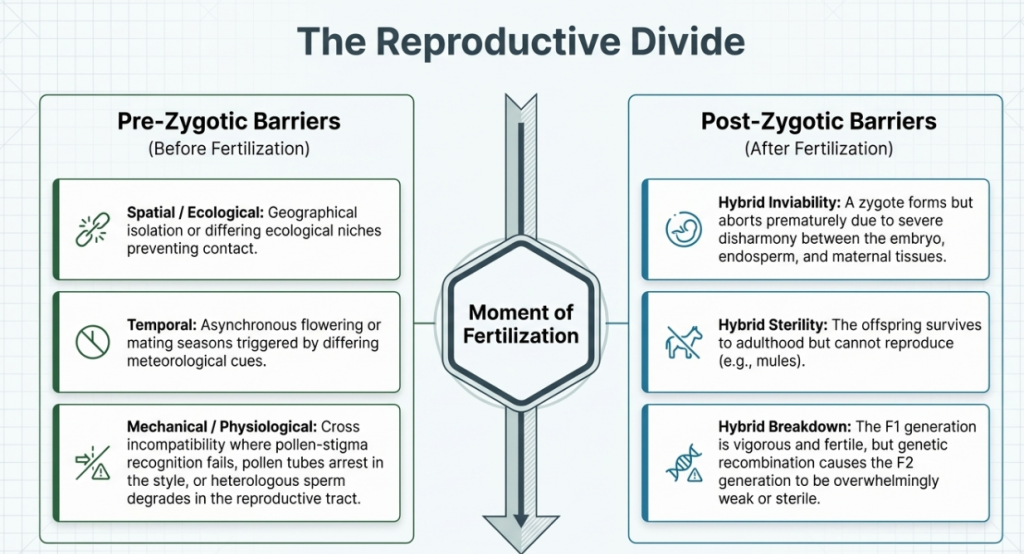
3. The Architecture of Reproductive Barriers: Pre- and Post-Fertilization
Reproductive isolation acts as a sophisticated evolutionary mechanism designed to maintain species integrity. For the modern geneticist, these barriers represent architectural challenges that must be deconstructed to allow for successful genetic recombination.
I. Pre-fertilization Barriers (Cross Incompatibility)
The pre-fertilization stage represents the first critical hurdle, governed by the “pollen-pistil” interaction. For successful gamete fusion, the maternal tissue must recognize and support foreign pollen—a process that frequently fails in wide crosses due to complex biochemical and mechanical blockages.
A primary cause of failure is Cross Incompatibility, mediated by the abnormal accumulation of callose (β-1,3-glucan) on the stigma and within the style. Callose acts as a biological sealant, obstructing pollen tube elongation. Data from Camellia sinensis (tea plant) studies reveal a paradoxical kinetic profile: in Distant Hybridization (DH), pollen tubes germinate rapidly, with some seen penetrating the style interior within 3 hours and reaching the base of the style by 9 hours. In contrast, Intraspecific Hybridization (IH) tubes grow significantly slower, taking 24 hours to reach the base. However, this increased speed in DH does not correlate with success; instead, it is accompanied by severe, intermittent callose blockages throughout the style and ovary that prevent ovule union.
The cytological failure is tripartite:
Micropylar Guidance and Fusion Failure: The inability of the tube to navigate to the micropyle or the failure of gamete fusion once reached.
Pollen Germination Failure: The inability of foreign pollen to hydrate or germinate on the maternal stigma.
Callose Accumulation: A significant barrier involves the accumulation of callose (a β-1,3-glucan-linked polysaccharide). Excessive callose reactions in the style distort and entangle pollen tubes, creating intermittent blockages that prevent entry into the ovary and subsequent union with the ovule.
II. Post-fertilization Barriers
Even when fertilization is successful, post-zygotic hurdles can impede the development of a viable plantlet.
- Hybrid Inviability: The zygote may fail to develop or spontaneously abort due to disharmony between the cytoplasm and nuclear genes or hostile interactions between the endosperm and the embryo.
Major Causes of Hybrid Non-Viability:
- Genomic Maladaptation: Adverse interactions between the chromosomes of the two parental species hinder development.
- Nucleo-Cytoplasmic Disharmony: Hostile interactions between the maternal cytoplasm and the hybrid nuclear genes.
- Endosperm-Embryo Hostility: The most common cause of abortion; a lack of coordination between genotypes causes the endosperm to fail, effectively starving the developing zygote.
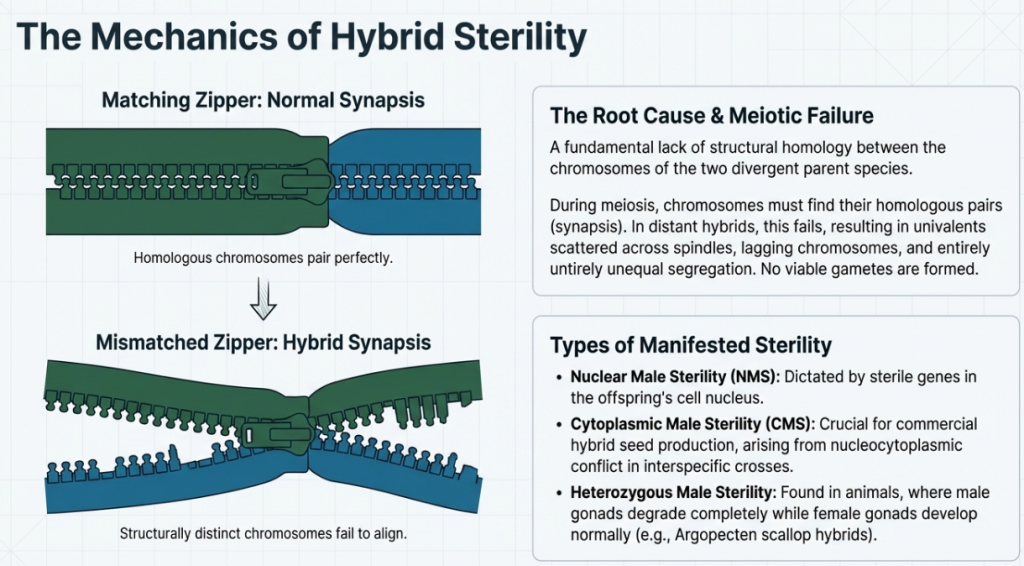
- Hybrid Sterility: Prevalent in intergeneric crosses, the lack of structural homology between parental chromosomes leads to meiotic irregularities, resulting in non-functional gametes.
- Hybrid Breakdown: A phenomenon where F1 hybrids appear vigorous, but the F2 generation exhibits weakness or sterility. This is frequently attributed to the disruption of advantageous gene combinations or cryptic structural differences in chromosomes that lead to deleterious deletions during recombination.
4. Strategic Methodologies: Overcoming Incompatibility and Hybrid Inviability
The evolution of plant breeding has shifted from passive selection to active technical manipulation, utilizing interventions to bypass natural reproductive barriers and ensure the survival of wide-cross progeny.

- Manipulation of Ploidy: Breeders utilize colchicine to induce allopolyploidization. By creating amphidiploids, each chromosome is provided with a homologous partner for meiosis, thereby restoring fertility in otherwise sterile hybrids.
- Bridge Crosses: When two species are directly incompatible, a “bridging species” compatible with both is utilized. For example, in Nicotiana breeding, N. repanda is incompatible with N. tabacum. However, by first crossing N. repanda with N. sylvestris, the resulting hybrid can then be successfully crossed with N. tabacum.
- Embryo Rescue Technique: Critical for instances of endosperm degradation, such as in Hordeum vulgare x Secale cereale crosses. The process follows a standardized sequence:
- Excision: The proembryo is surgically removed from the maternal tissue within 3–5 days post-pollination.
- Culturing: The proembryo is placed on a specialized agar medium containing essential growth regulators and nutrients.
- Collection: Once the embryo matures in vitro, the resulting plantlet is carefully collected.
- Transplantation: The lab-grown plantlet is acclimated and moved into soil to reach maturity.
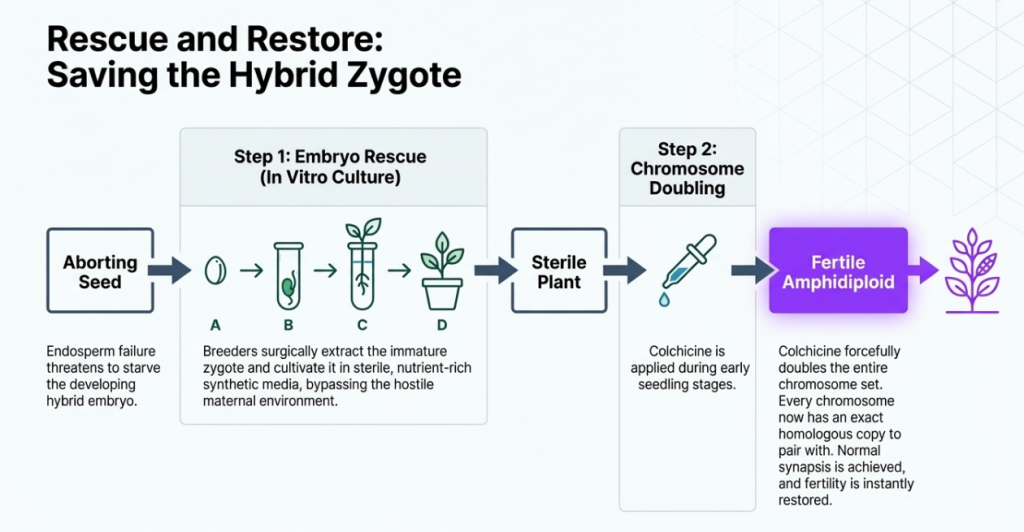
Where nature builds a wall, the geneticist builds a bridge. We utilize specific interventionist techniques to force these difficult unions.
| Technique | Primary Benefit |
|---|---|
| Bridge Cross | Utilizes a third, compatible “bridging species” to link two otherwise incompatible genomes. |
| Embryo Rescue | Circumvents embryo-endosperm disharmony by providing a controlled in vitro nutrient environment for the developing zygote. |
| Reciprocal Cross | Swapping the maternal and paternal roles to identify the most compatible cytoplasmic-nuclear interaction. |
| Colchicine Treatment | Induces polyploidy to double the chromosome set, restoring fertility by providing homologous partners for meiosis. |
| Pollen Mixture | Utilizes a blend of pollens to overpower adverse biochemical interactions among the pistil and foreign pollen grains. |
Intervention Matrix
| Biological Barrier | Remediation Technique |
|---|---|
| Style Length/Stigma Incompatibility | Style Decapitation or Pollen Mixtures |
| Cross Incompatibility (Bridge) | Bridge Crosses (e.g., using N. sylvestris to bridge N. repanda x N. tabacum) |
| Slow Pollen Tube Growth | Growth Regulators (IAA, NAA, Gibberellic Acid) |
| Endosperm Failure / Embryo Abortion | Embryo Rescue |
| F1 Hybrid Sterility | Colchicine Treatment (Amphidiploidy) |
| Failed Gamete Fusion | Protoplast Fusion |
5. The “So What?” of Genetic Transfer: Improving Yield, Quality, and Resistance
Wild germplasm acts as a “treasure reservoir,” harboring genes lost through the bottleneck effect of domestication. Distant hybridization allows high-value traits to be reintegrated into modern agricultural systems to ensure long-term food security in the face of climate instability.
Strategic Trait Acquisition via Wide Crosses
| Crop | Character Transferred | Source Species |
|---|---|---|
| Tomato (Solanum) | Late blight and leaf roll resistance | Solanum demissum |
| Cotton (Gossypium) | Jassid and Blackarm resistance | G. tomentosum / G. arboreum |
| Sugarcane (Saccharum) | Sereh disease and cold tolerance | Saccharum spontaneum |
| Okra (Abelmoschus) | Resistance to Yellow Mosaic Virus (YMV) | Abelmoschus manihot |
The strategic impact on fruit quality is profound. In tomatoes, manipulating the polygalacturonase (PG) enzyme—which degrades middle lamella pectin—is used to manage softening. Notably, mRNA for PG is not detectable in immature green fruits, making it a highly specific target for temporal regulation during ripening. In tea plants, distant hybrids exhibit significant chemical variation, providing a foundation for breeding specialized antioxidant-rich varieties.
6. Case Studies in Germplasm Innovation: Tomatoes and Tea
Solanum and Camellia serve as model organisms for understanding how distant genetic resources are utilized to meet specific industrial needs.
I. Solanum (Tomato) Breeding
The cultivated tomato (Solanum lycopersicum) is a classic example of “domestication syndrome,” where centuries of selection for fruit size have depleted genetic variation. To future-proof the crop, breeders look to wild relatives within the Solanum clade.
- The Problem: Domesticated varieties lack the genetic diversity to survive emerging pathogens and climate instability.
- The Distant Solution: By crossing S. lycopersicum with wild relatives like S. chilense (syn. Lycopersicon chilense) and S. habrochaites (syn. L. hirsutum), geneticists have harvested “super-traits”:
- Nutritional Fortification: High-impact traits from S. chilense have boosted β-carotene levels up to 885 mg/100 g and ascorbic acid up to 49.45 mg/100 g.
- Pathogen Defense: Successful transfer of R-genes, including Bs4 (resistance against Xanthomonas) and Prf (resistance against Pseudomonas syringae).
- Environmental Resilience: Integration of genes for cold and drought tolerance from wild species that evolved in the harsh Peruvian-Chilean deserts.
II. Camellia (Tea) Hybridization
A pivotal study in tea breeding compared Interspecific Distant Hybridization (DH)—using Camellia sinensis (JX) as the maternal parent and C. gymnogyna (CGC) as the paternal parent—against standard Intraspecific Hybridization (IH). This experiment highlights the “Life Cycle of a Barrier” that breeders must overcome.
The Post-Fertilization Crisis: While some fertilization occurred, a severe crisis followed. Between 5 and 10 months post-pollination, a massive fruit drop was observed. The resulting seeds exhibited a strikingly low germination rate of 11.8%, compared to 67.7% in the IH group.
The Early Sprint: Counter-intuitively, DH pollen tubes often start faster. DH tubes germinated as early as 3 hours post-pollination (compared to 6 hours for IH). DH tubes reached the style base in just 9 hours, while IH tubes required 24 hours to complete the same distance.
The Callose Blockade: Despite the early lead, DH tubes hit a pre-fertilization wall. Scientists observed an excessive accumulation of callose (a β-1,3-glucan-linked polysaccharide) at the style base. This acting blockade caused the tubes to twist, coil, and intermittently stall, significantly hindering ovary entry.
Successful distant hybridization creates “Super-Parenting” phenomena, where hybrids surpass their parents in chemical complexity or environmental resilience. In the tea study, the Shannon-Wiener diversity index for offspring chemical components was >2, indicating exceptionally rich genetic diversity.
| Crop | Parental Source | Hybrid Offspring Observation (The Payoff) |
|---|---|---|
| Tea | C. sinensis x C. gymnogyna | Caffeine: 21.31–58.21 mg/g; Amino acids >5%; Purple-green foliage. |
| Tomato | S. lycopersicum x L. minutum | Ascorbic Acid (Vitamin C) increased to 59 mg/100g. |
| Tomato | S. lycopersicum x L. chilense | β-carotene (Pro-vitamin A) reached 885 mg/100g. |
| Cotton | G. hirsutum x G. tomentosum | Successful transfer of Jassid resistance to cultivated varieties. |
| Triticale | Wheat x Rye | A man-made genus combining rye’s hardiness with wheat’s yield. |
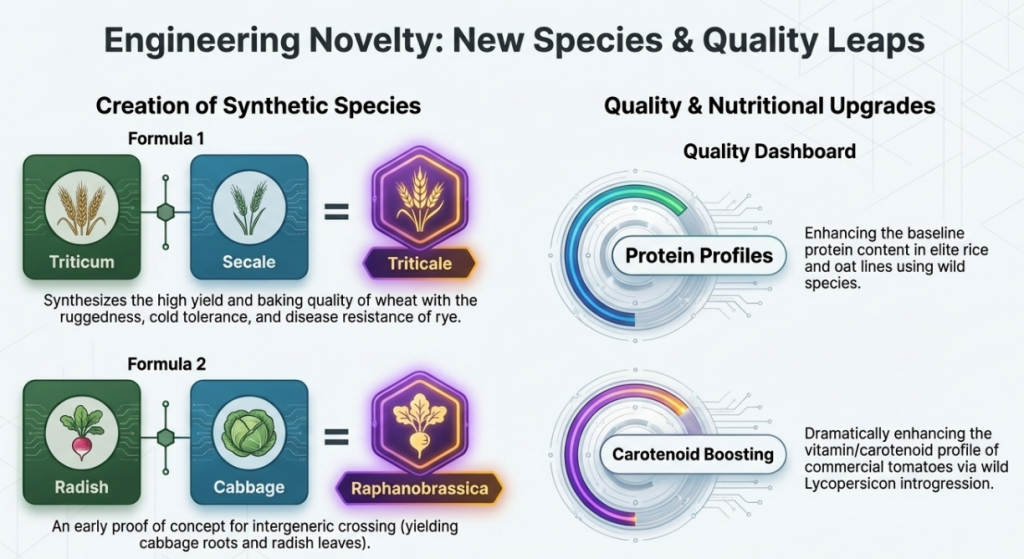
Commercial Value Drivers of Distant Hybridization
| Wild-Sourced Trait | Source Species Example | Impact on Crop Viability & Marketability |
|---|---|---|
| Jassid Resistance | G. tomentosum | Enhances cotton yield; reduces chemical pesticide dependency. |
| Blackarm Resistance | G. arboreum | Stabilizes cotton production in disease-prone environments. |
| Late Blight Resistance | Solanum demissum | Critical for potato security and post-harvest longevity. |
| YMV Resistance | Abelmoschus manihot | Prevents total crop loss in commercial Okra production. |
| Aphid Resistance | Fragaria chiloensis | Reduces vector-borne viral transmission in strawberry. |
| Sereh Disease Resistance | Saccharum spontaneum | Essential for sugarcane germplasm stability. |
| Nutritional Enhancement | L. pimpinellifolium | Increases lycopene/carotene for premium health markets. |
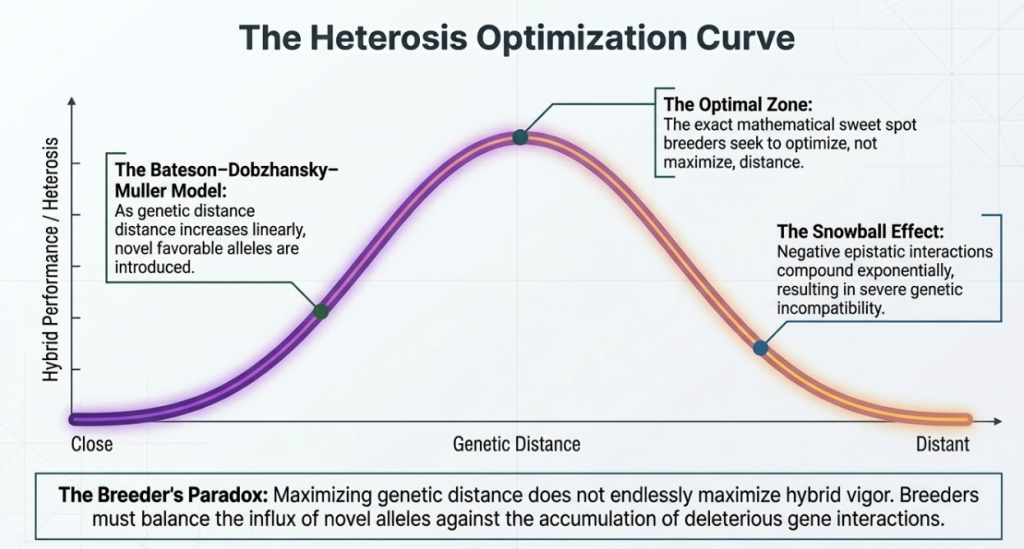
7. Maximization through Optimization: The Role of Genetic Distance
A critical challenge in wide breeding is identifying the “optimum genetic distance.” Heterosis (hybrid vigor) does not increase linearly; it follows a curve where vigor increases to a peak before plummeting due to genomic disharmony and hybrid breakdown.
Critical Takeaways for Heterosis Optimization:
- Linkage Disequilibrium (LD): Effective heterosis prediction using molecular markers requires tight linkage (high LD) between those markers and the actual heterotic quantitative trait loci (QTLs).
- Marker Heterozygosity: Within specific populations, marker heterozygosity serves as a primary indicator for potential hybrid performance, provided the markers are linked to heterotic QTLs.
- Genomic Harmony: Breeders must seek a balance where genetic diversity is maximized without triggering the incompatibility inherent in excessive taxonomic divergence.
8. Conclusion: The Strategic Outlook for Evolutionary Breeding
Distant hybridization remains an indispensable tool for shattering the genetic bottlenecks of modern agriculture. By leveraging wild germplasm and employing advanced interventions like embryo rescue and induced amphidiploidy, breeders can bridge the biological gaps in cultivated species. As global agriculture faces the dual pressures of climate resilience and food security, the strategic utilization of distant crosses is paramount for developing the next generation of robust, nutrient-dense crops.
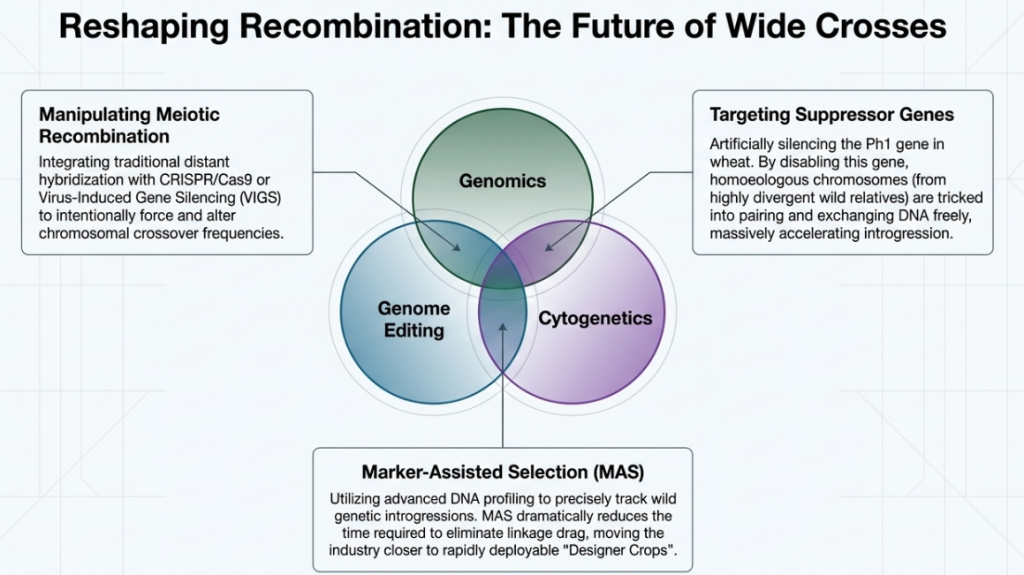
Strategic Roadmap for High-Value Hybridization:
- Integrated Observation: Programs must utilize real-time cytological monitoring of pollen tube kinetics to pinpoint the optimal therapeutic window for embryo rescue.
- Amphidiploid Stabilization: Deploy colchicine early in the F1 stage to bypass structural sterility and allow for immediate trait evaluation.
- Rigorous Backcrossing: Implement aggressive backcrossing to the cultivated parent to mitigate linkage drag, ensuring “R genes” are integrated without compromising habit or yield.
- Allelic Identification: Target specific genetic pathways, such as the TCS1 gene (specifically the TCS1e allele), as the most efficient path for breeding high-value traits like low-caffeine tea.
Image Summary

Questions/Answers
1. What are pre-fertilization and post-fertilization barriers in plant breeding?
In plant breeding, particularly during distant hybridization, reproductive barriers are categorized based on whether they occur before or after the formation of a zygote. These are known as pre-fertilization (pre-zygotic) and post-fertilization (post-zygotic) barriers.
Pre-fertilization Barriers
These mechanisms prevent the successful fusion of male and female gametes to form a zygote.
- Spatial and Ecological Isolation: Species may be physically separated by geographic distance or occupy different ecological niches (e.g., spring versus winter wheat), preventing natural hybridization.
- Temporal and Seasonal Isolation: Parents often have non-overlapping reproductive seasons or asynchronous flowering times, which hinders gene exchange.
- Mechanical Isolation: Differences in flower morphology or specific pollinator requirements can prevent successful mating.
- Cross Incompatibility: This is a major physiological barrier where functional pollen fails to fertilize the egg cell. It manifests through:
- Germination Failure: Pollen grains may fail to germinate on the stigma due to recognition failure or lack of hydration.
- Inhibited Pollen Tube Growth: Pollen tubes may grow too slowly, swell at the apex, twist, or be unable to reach the ovary.
- Callose Deposition: Excessive callose accumulation in the style or ovary can physically block pollen tube elongation.
- Signaling and Recognition Failures: In families like Brassicaceae, complex peptide-receptor kinase systems (e.g., RALF peptides) act as a “lock-and-key” mechanism to reject foreign pollen.
Post-fertilization Barriers
These barriers occur after successful fertilization, preventing the resulting hybrid from developing into a stable, fertile individual.
- Hybrid Inviability and Embryo Abortion: The zygote may form but fail to develop into a normal embryo. Common causes include:
- Genotypic Disharmony: Unfavorable interactions between the parental genomes or between the nucleus and the foreign cytoplasm.
- Endosperm Failure: Disharmony between the embryo, endosperm, and maternal tissues often leads to nutritional deficiencies and subsequent embryo death.
- Hybrid Sterility: The hybrid offspring lives to maturity but is unable to produce viable gametes. This is primarily caused by a lack of chromosomal homology, which prevents normal pairing (synapsis) during meiosis.
- Hybrid Breakdown: While F1 hybrids may appear vigorous and fertile, their F2 or backcross progenies are often weak, sterile, or non-viable due to the segregation of incompatible gene combinations.
- Hybrid Necrosis: An extreme form of outbreeding depression where simple epistatic interactions trigger a genetic autoimmunity syndrome, leading to tissue necrosis and hybrid death.
Overcoming Barriers
Breeding programs utilize several advanced techniques to circumvent these barriers, including embryo rescue to salvage aborting embryos, chromosome doubling (using reagents like colchicine) to restore fertility in sterile hybrids, and bridge species to facilitate gene transfer between incompatible parents.
2. What are bridge species and how do they work?
Bridge species (also known as bridging species) are intermediate species used in plant breeding to facilitate gene transfer between two other species that are cross-incompatible or cannot produce fertile hybrids directly. This technique is a vital tool for broadening the genetic base of crops by accessing desirable traits in wild relatives—particularly those in the secondary and tertiary gene pools—that are otherwise unreachable through standard hybridization.
How Bridge Species Work
The method exploits the inter-relationships between species: while Species A (the cultivated crop) and Species C (the wild donor) may be incompatible, Species B (the bridge) is chosen because it can successfully hybridize with both. The mechanism typically follows these steps:
- Intermediate Hybridization: The donor species with the target trait is first crossed with the bridge species to produce a transitional intermediate hybrid.
- Restoring Fertility: If the intermediate hybrid is sterile, chromosome doubling (often via colchicine treatment) is used to create a fertile synthetic amphidiploid.
- Introgression into the Crop: This fertile intermediate is then crossed with the recipient crop species.
- Stabilization: The resulting plants undergo several generations of backcrossing to the cultivated parent, coupled with selection to stabilize the desired trait and eliminate undesirable “linkage drag” from the wild relative.
Key Examples in Crop Improvement
- Lentil: Lens ervoides has been successfully used as a bridge species to transfer resistance to ascochyta blight and anthracnose from L. lamottei and L. nigricans into the cultivated lentil, L. culinaris.
- Tobacco: To bypass the incompatibility between Nicotiana repanda and N. tabacum, researchers utilized N. sylvestris as a bridge parent.
- Vigna (Beans): Bruchid resistance was transferred from V. umbellata to the azuki bean (V. angularis) by utilizing V. nakashimae as a bridge.
- Wheat: Introgression of leaf rust resistance from the wild Mediterranean grass Aegilops umbellulata into common wheat was achieved through a bridge cross with T. dicoccoides.
- Brassica: Brassica rapa served as a genetic bridge to move Alternaria resistance from the wild species Diplotaxis erucoides into cultivated B. juncea.
- Barley: Hexaploid tritordeums (hybrids of Hordeum chilense and wheat) are used as bridge species to transfer stress tolerance and disease resistance from wild barley into durum wheat.
Advantages and Requirements
Bridge crossing provides a non-transgenic alternative for accessing “super genes” for biotic and abiotic stress resistance. However, the process is technically demanding and often requires supplementary interventions such as embryo rescue to salvage aborting intermediate hybrids, as well as skillful management of large segregating populations to identify successful recombinants.
3. What is embryo rescue and when is it used?
Embryo rescue (ER) is a vital in vitro plant breeding technique used to salvage immature embryos that would otherwise abort or fail to develop into viable seeds due to reproductive barriers. It involves the surgical excision of the developing embryo from the seed or ovule, followed by its cultivation in a sterile, nutrient-rich artificial medium that provides the necessary environment for growth.
When Embryo Rescue is Used
Embryo rescue is primarily employed to bypass post-fertilization (post-zygotic) barriers that prevent the formation of a stable hybrid. Specific applications include:
- Successful Distant Hybridization: In wide crosses (interspecific or intergeneric), the zygote may form, but the embryo often aborts due to genotypic disharmony, endosperm failure, or unfavorable interactions between the embryo and maternal tissues. ER acts as an artificial endosperm to nurture these embryos.
- Haploid and Doubled-Haploid Production: It is a critical step in producing homozygous lines rapidly. For instance, in “wide crosses” (e.g., wheat × maize) or irradiated pollen techniques, an embryo may begin to develop but will die without intervention because no functional endosperm exists to support it.
- Breaking Seed Dormancy and Conserving Rare Species: ER is used to germinate seeds from rare or endangered species that have poor natural viability or extreme dormancy that traditional methods cannot overcome.
- Shortening Breeding Cycles: It can be used for rapid generation advancement by germinating immature seeds earlier than they would mature naturally on the plant.
Key Examples in Crop Breeding
The sources highlight several successful implementations of embryo rescue across various crops:
- Legumes: Used to obtain hybrids between cultivated chickpea and wild species like Cicer bijugum, and in lentils to transfer disease resistance from Lens ervoides to cultivated varieties.
- Cucurbits: Widely used in melons, cucumbers, and pumpkins for interspecific hybridization to introgress traits like disease resistance and for haploid plant production.
- Cereals: Used in rice to produce hybrids with distantly related wild ancestors and in wheat to produce haploids through chromosome elimination after crossing with maize or barley.
- Ornamentals: Successfully used in lilies (Lilium) to create unique flower characteristics by overcoming incompatibility between different lily groups.
- Tea Plants: Employed to obtain seedlings from distant interspecific crosses where seed germination is otherwise zero.
Factors for Success and Constraints
- Timing: Excision should generally be performed as late as possible before the embryo begins to degrade, but early enough to prevent complete abortion. For cucurbits, this window is often 7 to 14 days post-pollination.
- Media and Hormones: Success depends on optimized nutrient formulations (often MS or E20/E21 media) and growth regulators like Auxin and Cytokinin.
- Constraints: The technique is technically demanding, requires specialized laboratory facilities, and is not always cost-effective. There is also a risk of inducing mutations during the in vitro phase.
4. How can callose accumulation be reduced to improve hybridization?
Reducing callose accumulation is a strategy to overcome pre-fertilization barriers, as excessive callose deposition in the stigma and style can physically block or inhibit pollen tube elongation, preventing it from reaching the ovary.
Drawing from the sources, the following methods are used to mitigate or bypass the effects of callose accumulation to improve hybridization success:
1. Phytohormone Treatments
Hormonal imbalances are often linked to the callose response in wide crosses. Applying exogenous hormones can restore a physiological environment that favors pollen tube growth:
- Gibberellic Acid (GA3): In intergeneric crosses of Lagerstroemia, where extensive callose deposition typically leads to fruit abscission, a foliar application of 250 mg·L⁻¹ GA3 has been shown to partially mitigate incompatibility and significantly improve fruit-set rates.
- Ethylene Inhibitors: Ethylene often promotes the programmed cell death (PCD) associated with callose deposition and pollen rejection. Treatments with the ethylene inhibitor Aminooxyacetic acid (AOA) have been used to allow pollen tubes to bypass rejection mechanisms and reach the ovary.
- Other Growth Regulators: Post-pollination sprays of 2,4-D, IAA, and NAA can help maintain developing seeds by facilitating the division of the hybrid zygote and endosperm, even when initial tissues are prone to inhibitory responses like callose build-up.
2. Peptide Signaling Manipulation
In specific families like Brassicaceae, a “lock-and-key” gating mechanism involving RALF peptides and receptor kinases (like FERONIA) controls pollen penetration.
- The accumulation of reactive oxygen species (ROS) in the stigma induces callose and prevents hydration.
- Treating the stigma with specific pollen-derived RALF peptides (e.g., pRALF26) can “unblock” this barrier, allowing the pollen tubes of distantly related species to germinate and penetrate the stigma despite natural reproductive isolation.
3. Physical Circumvention (Cut-Style Technique)
If callose deposition or coiling is localized to the stigma or the upper part of the style, breeders can use cut-style pollination.
- This involves removing the stigma and the section of the style where callose typically accumulates and applying pollen directly to the remaining style or the ovary.
- This physical bypass prevents the pollen tube from encountering the tissues where the inhibitory “foreign” recognition and callose deposition occur.
4. Selection of Compatible Genotypes
Research shows that some specific accessions within a species may lack the gene-controlled barriers that trigger callose deposition. For example, certain sorghum genotypes (homozygous for the iap allele) lack the standard inhibition response, allowing maize pollen tubes to grow through the ovary without being stopped by typical stylar barriers. Identifying and using such lines as maternal parents can eliminate the need for chemical or physical intervention.
5. Tell me more about the role of bridge species.
Bridge species (also known as bridging species) act as intermediate genetic links to facilitate gene transfer between two species that are cross-incompatible or cannot produce fertile hybrids through direct mating. This technique is essential for accessing “super genes”—desirable traits like disease and pest resistance—found in the secondary and tertiary gene pools of wild relatives that are otherwise unreachable for crop improvement.
Mechanism of Bridging
The process exploits the varying degrees of phylogenetic relationships between species. If Species A (the cultivated crop) cannot successfully hybridize with Species C (the wild donor), Species B is selected as a “bridge” because it is compatible with both. The typical workflow involves:
- Initial Hybridization: The wild donor species (C) is first crossed with the bridge species (B) to produce an intermediate hybrid.
- Restoring Fertility: Because these intermediate hybrids are often sterile, researchers frequently use chromosome doubling (often via colchicine treatment) to create a fertile synthetic amphidiploid.
- Final Transfer: This fertile intermediate is then crossed with the recipient crop species (A).
- Stabilization: The resulting progeny undergo several generations of backcrossing to the cultivated parent, coupled with rigorous selection to stabilize the desired trait and eliminate undesirable “linkage drag” from the wild relative.
Key Examples in Plant Breeding
The sources provide several successful implementations of bridge species across major crops:
- Lentils: Lens ervoides has been used as a bridge to transfer resistance to ascochyta blight and anthracnose from the incompatible L. lamottei and L. nigricans into cultivated L. culinaris.
- Tobacco: To bypass incompatibility between Nicotiana repanda and N. tabacum, researchers utilized N. sylvestris as a bridge parent.
- Wheat: To introgress leaf rust resistance from the Mediterranean wild grass Aegilops umbellulata into common wheat, a bridging cross was made with T. dicoccoides to produce an amphidiploid. Additionally, tritordeums (hybrids of wild barley Hordeum chilense and wheat) serve as bridges to move stress tolerance and disease resistance into durum wheat.
- Brassica (Mustard): Brassica rapa served as a genetic bridge to move Alternaria resistance from the wild Diplotaxis erucoides into cultivated B. juncea by first creating a synthetic intermediate species named “Erurapa”.
- Vigna (Beans): Bruchid resistance was transferred from the rice bean (V. umbellata) to the azuki bean (V. angularis) by utilizing V. nakashimae as a bridge species.
- Buckwheat: Fagopyrum homotropicum has been used to improve the success of interspecific hybridization between the two cultivated species F. tataricum and F. esculentum.
Requirements and Challenges
While bridge crossing is a powerful non-transgenic approach for diversifying the primary gene pool, it is a long and tedious process. It often requires supplementary interventions such as embryo rescue to salvage the immature intermediate hybrids that would otherwise abort. Furthermore, breeders must manage large segregating populations over 5–10 years to break the linkage between the gene of interest and undesirable wild traits like pod shattering or small seed size.
6. Which growth regulators are most effective for embryo rescue?
The effectiveness of growth regulators in embryo rescue is largely dependent on the plant species and the specific reproductive barrier being targeted, such as poor fruit retention or developmental arrest in the embryo. The most commonly used regulators include Auxins, Cytokinins, and Gibberellins, often applied singly or in specialized combinations.
1. Auxins (IAA, NAA, 2,4-D, and IBA)
Auxins are primarily used to accelerate pollen tube growth, facilitate the division of the hybrid zygote and endosperm, and prevent the premature shedding of pods or fruits.
- 2,4-D: This is noted as being particularly effective for promoting the retention of flowers and pods in Vigna species when applied at low concentrations (approximately 1.0 mg/L). It is also used to overcome pollen tube arrest in wheat × maize crosses.
- IAA (Indole-3-acetic acid): Often used at a concentration of 0.01 mg/L, IAA is a standard component in media for rescuing immature embryos in cucurbits. It is frequently paired with a cytokinin to support embryo development.
- NAA and IBA: These are used to increase the scope of distant hybridization by preventing initial pod abscission and maintaining developing seeds.
2. Cytokinins (Kinetin, Benzyladenine/BAP/6-BA, and Zeatin)
Cytokinins are essential for stimulating cell division within the developing embryo and are a core part of the artificial “endosperm” provided by culture media.
- Kinetin and BAP (Benzylaminopurine): These are typically applied at 0.1 mg/L in conjunction with IAA for cucurbit embryo rescue.
- 6-BA: In Lagerstroemia (crape myrtle) breeding, 30 mg/L 6-BA was found to help with fruit retention, though it was less effective than gibberellins.
- Zeatin: This has been successfully used alongside IAA in interspecific crosses of Cucurbita.
3. Gibberellins (GA/GA₃)
Gibberellic acid is critical for reducing fruit drop and maintaining the heterotrophic state of a newly formed embryo.
- GA₃: In Lagerstroemia intergeneric crosses, a 250 mg/L GA₃ solution was found to be the most effective treatment for reducing fruit abscission, achieving a fruit-setting rate of 78%. It has also been used to obtain viable hybrids in Lens species.
Effective Combinations and Additives
Optimal results are frequently achieved through hormone-aided pollination followed by specialized in vitro media.
- Auxin + Cytokinin: A common standard for immature embryos is a combination of 0.1 mg/L Kinetin/BAP and 0.01 mg/L IAA.
- Natural Additives: Coconut water (often at 5%) is frequently used as a supplement to provide a complex mix of natural growth factors that further promote embryo survival and germination.
- Sequential Media: For some species like C. melo, a sequential strategy—starting with a high-nutrient medium for germination followed by a half-strength medium for seedling development—is most effective.
7. How do breeders use chromosome doubling?
Breeders use chromosome doubling as a primary technique to overcome hybrid sterility, particularly in distant crosses where parental species have different genomes that cannot pair normally during meiosis. By doubling the chromosome count, each chromosome receives a corresponding homologous partner, allowing for proper synapsis and the production of viable gametes.
The most common method involves treating seeds during early germination or young seedlings with chemical reagents, most notably colchicine. Other chemicals like oryzalin, Amiprophos-methyl, and pronamid are also utilized.
Key Applications in Breeding
- Restoring Fertility in Sterile Hybrids: Interspecific and intergeneric F1 hybrids are often self-sterile because their chromosomes lack homology. Doubling these chromosomes creates a fertile amphidiploid (or allotetraploid).
- Synthesizing New Crop Species: Several modern crops were created this way. A classic example is Triticale, an amphidiploid developed by doubling the chromosomes of a sterile wheat × rye hybrid. Another is Raphanobrassica, a hybrid of radish and cabbage.
- Production of Doubled Haploids (DH): Breeders use chromosome doubling to rapidly produce homozygous lines in a single generation. Haploid plants are first induced through methods like anther culture or wide crosses (e.g., wheat × maize) and are then doubled to create true-breeding lines, saving 4–6 generations of traditional selfing.
- Bridge Species: When two species are directly incompatible, breeders may cross one with a bridge species, double the hybrid’s chromosomes to restore fertility, and then cross that fertile intermediate with the second parent.
- Ornamental Trait Enhancement: In ornamentals, induced polyploidy is used to create cultivars with larger flowers, thicker leaves, more intense green coloring, or more compact growth.
- Increasing Resistance: Doubling chromosomes can broaden the genetic base for stress tolerance. For instance, tritordeums (hybrids of wild barley and wheat) serve as bridges to move disease resistance and drought tolerance into wheat after chromosome doubling.
Factors for Success
The success of these techniques often depends on the cross direction and ploidy level. Generally, hybrids are more likely to survive and be effectively doubled when the female parent has a higher or equal chromosome number than the male parent. Additionally, polyploid species are often more tolerant of the genetic disturbances caused by foreign gene introgression than diploids.
8. Can you explain the differences between DH and IH?
In plant breeding, Distant Hybridization (DH) and Intraspecific Hybridization (IH) represent two different levels of genetic crossing, primarily distinguished by the taxonomic relationship between the parents and the severity of reproductive barriers.
Core Definitions
- Distant Hybridization (DH): Refers to crosses between different species (interspecific), genera (intergeneric), or even higher-ranking taxa. It is used to break species limits, increase genetic variation, and introduce “super genes” from wild relatives that are unavailable in the cultivated gene pool.
- Intraspecific Hybridization (IH): Also known as “close hybridization,” this occurs between varieties, subspecies, or genotypes of the same species. While it can lead to gene mutations, the resulting range of variation is generally more limited compared to DH.
Comparative Differences in Reproduction
Based on cytological observations (specifically in tea plants), the two methods exhibit significant physiological differences during the fertilization process:
| Feature | Intraspecific Hybridization (IH) | Distant Hybridization (DH) |
|---|---|---|
| Pollen Tube Growth | Typically slower initial emergence; in some species, it can take up to 24 hours to reach the base of the style. | Often shows faster initial germination and growth, sometimes reaching the style base as early as 9 hours post-pollination. |
| Callose Accumulation | Callose may accumulate at the style base, causing some twisting or coiling, but fertilization is usually completed. | Exhibits intensified callose reactions and intermittent blockages throughout the style, making it difficult for tubes to unite with ovules. |
| Fruit-Setting Rate | Generally higher; for example, approximately 83.5% in tea plant studies. | Significantly lower; approximately 79.1% in the same studies. |
| Fruit Development | Minimal fruit drop after the initial stages of development. | Experiences continuous fruit drop from the time of pollination until harvest. |
| Seed Germination | High germination rates (e.g., 67.7%). | Very low germination rates (e.g., 11.8%) due to pronounced post-fertilization disorders. |
Barriers and Outcomes
- Reproductive Barriers: IH is largely a straightforward procedure, whereas DH faces severe pre-zygotic (e.g., pollen-pistil incompatibility) and post-zygotic (e.g., hybrid sterility or embryo abortion) barriers.
- Genetic Diversity: DH results in widespread phenotypic variation and rich genetic diversity in offspring, often producing new traits not found in either parent. IH typically results in progeny that are more similar to the parental range.
- Utility: IH is the standard for routine variety development. DH is a specialized tool for germplasm innovation, such as transferring disease resistance or creating entirely new species like Triticale.
9. What are some practical achievements of wide hybridization in crops?
Practical achievements of wide hybridization, also known as distant hybridization, include the creation of entirely new crop species, the transfer of “super genes” for resistance, and significant improvements in yield, quality, and abiotic stress tolerance across diverse families.
Synthesis of New Crop Species
Wide hybridization has been used to break species boundaries to create novel crops that combine the strengths of different genera.
- Triticale: This is the most successful example of a synthetic crop, developed by crossing wheat (Triticum aestivum) and rye (Secale cereale) to integrate wheat’s yield and grain quality with rye’s hardiness and winter vigor.
- Raphanobrassica: Developed from a cross between radish (Raphanus sativus) and cabbage (Brassica oleracea), this intergeneric hybrid was a landmark in cytogenetic research, though the initial forms were agriculturally impractical because they inherited the leaves of the radish and the roots of the cabbage.
- Raparadish: A cross between radish and turnip (Brassica rapa) that is utilized practically as a fodder crop.
- Nicotiana digluta: A new species synthesized by crossing N. tabacum and N. glutinosa.
Biotic Stress Resistance (Diseases and Pests)
One of the most valuable applications of wide hybridization is the introgression of resistance genes from wild relatives into cultivated crops.
- Wheat: Resistance to leaf rust was famously transferred from Aegilops umbellulata into common wheat, resulting in the “Transfer” line. Other resistance genes for rust and powdery mildew have been introgressed from Agropyron and rye.
- Rice: The Grassy Stunt Virus was managed by introgressing resistance from the wild species Oryza nivara into elite varieties like IR24. Additionally, the Xa21 gene for bacterial blight resistance was successfully transferred from O. longistaminata.
- Okra: The variety Parbhani Kranti was developed through a cross with Abelmoschus manihot, providing complete resistance to the Yellow Vein Mosaic Virus (YMV).
- Cotton: Resistance to Jassids and Blackarm has been transferred from wild species like G. tomentosum and G. arboreum into upland cotton varieties.
- Pigeon Pea: Resistance to pod borers (Helicoverpa armigera) has been successfully introgressed from its wild progenitor, Cajanus scarabaeoides.
Yield and Quality Improvement
Wide hybridization has provided a mechanism for overcoming the yield plateaus of elite cultivars.
- Oats: Interspecific crosses between Avena sativa and its wild relative A. sterilis resulted in yield increases of 25% to 30%.
- Sugarcane: Modern varieties were developed through “nobilization,” where high-yielding Saccharum officinarum was crossed with hardy, disease-resistant wild species like S. spontaneum and S. barberi.
- Rice (NERICA): “New Rice for Africa” was created by crossing the high-yielding Asian rice (O. sativa) with the hardy, weed-competitive African rice (O. glaberrima).
- Tomato: Wild species have been used to increase ascorbic acid (Vitamin C), carotene, and total soluble solids content in cultivated varieties.
Abiotic Stress Tolerance
Distant hybridization has enabled crops to adapt to harsher environments.
- Salinity Tolerance: A commercial variety of bread wheat with a 25% yield advantage in saline soils was developed using a gene from Triticum monococcum.
- Cold Tolerance: Winter hardiness has been introgressed into wheat from Agropyron and into grapes from the wild species Vitis amurensis.
- Submergence Tolerance: The Sub1 locus, conferring the ability to survive underwater for 14 days, was introgressed into major rice “megavarieties” from wild-derived breeding lines.
Practical Tools for Plant Breeding
- Cytoplasmic Male Sterility (CMS): Wide hybridization is a critical method for developing CMS systems (e.g., the Wild Abortive source in rice), which are essential for the commercial production of hybrid seeds in crops like rice, wheat, and pigeon pea.
- Haploid Production: Distant crosses (e.g., wheat × maize) are used to induce chromosome elimination, allowing for the rapid creation of doubled haploid lines and dramatically shortening the breeding cycle.
10. How do breeders use bridge species to overcome incompatibility?
Breeders use bridge species (or bridging species) as intermediate genetic links to facilitate the transfer of desirable traits between two species that are cross-incompatible or cannot produce fertile hybrids directly. This technique is essential for accessing “super genes”—such as those for disease, pest, and abiotic stress resistance—found in wild relatives that are otherwise unreachable through standard hybridization.
The Bridging Mechanism
The process exploits the varying degrees of phylogenetic relationships between species. If a cultivated crop (Species A) cannot hybridize with a wild donor (Species C), a third species (Species B) is selected as a “bridge” because it is compatible with both. The typical workflow involves:
- Intermediate Hybridization: The wild donor species is first crossed with the bridge species to produce a transitional intermediate hybrid.
- Restoring Fertility: Because these intermediate hybrids are often sterile due to a lack of chromosomal homology, researchers frequently use chromosome doubling (often via colchicine treatment) to create a fertile synthetic amphidiploid or allopolyploid.
- Final Transfer to the Crop: This fertile intermediate (now possessing a compatible genome) is crossed with the recipient cultivated crop.
- Stabilization and Selection: The resulting progeny undergo several generations of backcrossing to the cultivated parent, coupled with rigorous selection to stabilize the desired trait and eliminate undesirable “linkage drag” from the wild relative.
Supporting Technologies
Because the intermediate crosses themselves may still face reproductive barriers, breeders often use advanced interventions:
- Embryo Rescue: This is frequently required to salvage the immature intermediate hybrids that would otherwise abort before reaching maturity.
- Hormone Applications: Growth regulators like GA3 or 2,4-D may be used to promote fruit set and maintain the developing seeds of the bridge cross.
Key Examples in Plant Breeding
The sources document several successful implementations of bridge species:
- Lentil: Lens ervoides served as a bridge to transfer resistance to ascochyta blight and anthracnose from the incompatible L. lamottei and L. nigricans into cultivated L. culinaris.
- Tobacco: Nicotiana sylvestris was used as a bridge to bypass the incompatibility between N. repanda and N. tabacum.
- Vigna (Beans): Bruchid resistance was transferred from the rice bean (V. umbellata) to the azuki bean (V. angularis) by utilizing V. nakashimae as a bridge.
- Wheat: Leaf rust resistance from the wild Mediterranean grass Aegilops umbellulata was moved into common wheat by first making a bridging cross with Triticum dicoccoides to produce an amphidiploid.
- Brassica: Brassica rapa served as a genetic bridge to introgress Alternaria resistance from the wild Diplotaxis erucoides into cultivated B. juncea, a process that involved creating a synthetic intermediate species named “Erurapa”.
- Barley: Hexaploid tritordeums (hybrids of wild barley Hordeum chilense and wheat) are used as bridge species to transfer stress tolerance and disease resistance into durum wheat.
Challenges and Limitations
While powerful, the use of bridge species is a long and tedious process. It often requires 5 to 10 years of intensive breeding and backcrossing to samply enough recombination events to break the linkage between the targeted gene and undesirable wild traits. Furthermore, it demands significant technical expertise, specialized laboratory facilities, and large family sizes to identify successful recombinants.
11. What is the difference between alien addition and substitution?
In plant breeding and genetics, the difference between alien addition and substitution lies in whether the foreign genetic material is added to or replaces parts of the recipient species’ existing genome. Both are key methods of chromosome engineering used to import desirable traits from wild relatives.
Alien Addition
Alien addition involves adding one or more chromosomes (or fragments) from a foreign species to the complete, normal chromosome set of the recipient species.
- Mechanism: It creates a new genetic type by adding a pair (or two) of alien chromosomes to the host’s existing group.
- Result: The total number of chromosomes in the plant increases.
- Use: These “addition lines” are frequently used in crops like wheat to introduce “super genes” for disease resistance from wild relatives.
Alien Substitution
Alien substitution (or replacement) involves replacing one or more chromosomes or segments of the recipient species with corresponding genetic material from the alien species.
- Mechanism: Instead of just adding material, the alien chromosome or segment takes the place of a host chromosome or segment.
- Result: The total chromosome count typically remains the same as the host species, but the genetic composition is altered.
- Use: Substitution is often achieved through the development of Chromosome Segment Substitution Lines (CSSLs), which allow for more precise mapping and utilization of alien genes while potentially reducing linkage drag (the inclusion of unwanted wild traits).
Key Differences at a Glance
| Feature | Alien Addition | Alien Substitution |
|---|---|---|
| Action | Adds to the host genome. | Replaces parts of the host genome. |
| Chromosome Number | Increases the total count. | Typically remains the same. |
| Primary Goal | Creating a new type with extra genetic info. | Precise replacement of specific segments. |
Both techniques are instrumental in germplasm innovation, providing a basis for molecular marker development, gene cloning, and research into genome interactions.
12. Can you explain the difference between genotypic and cryptic sterility?
In plant breeding and distant hybridization, hybrid sterility—the inability of a hybrid to produce viable offspring—is often categorized by its underlying cause, such as genotypic (genic) sterility or cryptic structural hybridity.
Genotypic Sterility
Genotypic (often referred to in the sources as genic) sterility occurs when a hybrid is infertile despite having chromosomal pairings that appear entirely normal during meiosis.
- Cause: It is driven by specific sterile genes rather than large-scale structural mismatches between chromosomes.
- Nuclear Male Sterility (NMS): This form involves sterile genes located in the cell nucleus, which may be controlled by a single gene or complex multi-gene interactions.
- Cytoplasmic Male Sterility (CMS): This is controlled by genetic material within the cytoplasm, specifically the mitochondria, and is frequently used by breeders to facilitate hybrid seed production.
Cryptic Sterility (Cryptic Structural Hybridity)
Cryptic structural hybridity refers to sterility caused by small structural changes in the chromosomes that are not detectable during microscopic observations of meiosis.
- Cause: While the chromosomes may appear to pair correctly because the structural differences are so minute, these “cryptic” variations prevent the production of viable gametes.
- Detection: Because these changes are too small to notice during meiosis, the sterility may initially seem mysterious until analyzed further.
- Outcome: These minor structural differences can lead to deletions or duplications during recombination, resulting in the “hybrid breakdown” often seen in the F2 generation.
Key Differences and Solutions
- Mechanism: The primary distinction is that genotypic sterility is a functional failure of specific genes despite proper physical pairing, whereas cryptic sterility is a physical failure caused by undetected structural defects.
- Restoring Fertility: Cryptic structural hybridity can often be overcome through chromosome doubling (using reagents like colchicine). By doubling the chromosome set, each individual chromosome receives an identical homologous partner to pair with during meiosis, which ensures the production of functional, viable gametes. In contrast, genotypic sterility controlled by nuclear-cytoplasmic interactions may require specific restorer genes from a parent to regain fertility.
13. What are some practical achievements of wide hybridization in crops?
Practical achievements of wide (distant) hybridization in crops include the creation of entirely new species, the transfer of “super genes” for biotic and abiotic resistance, and significant improvements in yield and quality across diverse plant families.
1. Creation of New Crop Species
Wide hybridization has been used to combine the genomes of different genera to produce novel crops with combined advantages.
- Triticale: The most successful synthetic crop, developed by crossing wheat (Triticum aestivum) and rye (Secale cereale). It integrates the yield potential of wheat with the winter hardiness and vigor of rye.
- Raparadish: A hybrid of radish (Raphanus sativus) and turnip (Brassica rapa) used practically as a fodder crop.
- Nicotiana digluta: A new species synthesized by crossing N. tabacum and N. glutinosa.
- Tritordeums: Intergeneric amphiploids developed from wild barley (Hordeum chilense) and wheat, which exhibit high tolerance to drought and salt and resistance to diseases like rusts and powdery mildew.
2. Biotic Stress Resistance (Diseases and Pests)
Introgressing resistance genes from wild relatives has been essential for protecting crops from evolving pathogens and pests.
- Wheat: The variety “Transfer” was developed by moving leaf rust resistance from Aegilops umbellulata into common wheat. Other successes include stem rust resistance from Agropyron elongatum in the varieties “Eagle” and “Kite” and powdery mildew resistance from Dasyprum villosum (the Pm21 gene).
- Rice: The Grassy Stunt Virus was successfully managed by introgressing resistance from the wild species Oryza nivara into elite varieties like IR24. Additionally, the Xa21 gene for bacterial blight resistance was transferred from O. longistaminata and used in many commercial hybrids.
- Okra: The variety “Parbhani Kranti” was developed through a cross with Abelmoschus manihot, providing complete resistance to the Yellow Vein Mosaic Virus (YVMV).
- Pigeon Pea: Resistance to the Helicoverpa pod borer was introgressed from its wild progenitor, Cajanus scarabaeoides.
- Legumes: The mung bean variety “Pant Mung-4” was developed from a cross between mung bean and black gram, inheriting high resistance to foliar diseases.
3. Abiotic Stress Tolerance
Wide hybridization allows crops to adapt to harsher environmental conditions.
- Submergence Tolerance: The Sub1 locus, which allows rice to survive underwater for up to 14 days, was introgressed from the wild rice relative FR13A into major “megavarieties” like Swarna-Sub1.
- Salinity Tolerance: A 25% yield advantage in saline soils was achieved in bread wheat by introgressing a gene from the wild relative Triticum monococcum.
- Aluminum Toxicity: Tolerance to aluminum was moved from O. rufipogon into the cultivated rice variety IR64.
4. Yield and Quality Improvement
- Yield Increase: Crossing white oats (Avena sativa) with wild A. sterilis resulted in yield increases of 25% to 30%.
- Nutritional Quality: High grain protein content (the Gpc-B1 gene) was transferred from the wild tetraploid wheat Triticum dicoccoides to common wheat. In tomato, wide crosses with wild species have increased ascorbic acid (Vitamin C), carotene, and total soluble solids.
- Fiber and Starch: In cotton, wide hybridization has improved lint strength and staple length (e.g., the variety MCU-2). In potatoes, wild species have been used to increase starch content and frost resistance.
- Sugarcane Nobilization: Modern sugarcane varieties were developed by crossing high-yielding “noble” cane (S. officinarum) with hardy wild species like S. spontaneum and S. barberi to combine yield with disease resistance and cold tolerance.
5. Practical Tools for Breeding
- Male Sterility: Wide hybridization is a primary method for developing Cytoplasmic Male Sterility (CMS) systems, which are vital for commercial hybrid seed production in rice (e.g., the Wild Abortive source), wheat, and pigeon pea.
- Haploid Production: Distant crosses, such as wheat × maize or barley × H. bulbosum, are used to induce chromosome elimination, allowing for the rapid creation of doubled haploid lines that can shorten breeding cycles by up to five years.
14. What is the difference between genic and cytoplasmic male sterility?
The primary difference between genic (also known as nuclear) and cytoplasmic male sterility lies in the cellular location of the genetic material that causes the sterility and the patterns of inheritance.
Genic Male Sterility (GMS or Nuclear Male Sterility)
- Location: The sterile genes are located in the cell nuclei of the plant.
- Genetic Control: It can be controlled by a single gene or multi-gene interactions.
- Inheritance Patterns: It is most commonly controlled by recessive genes, although dominant forms can occur through mutations (e.g., Taigu genic male-sterile wheat).
- Characteristics: GMS can occur even when chromosomal pairings during meiosis appear entirely normal. Approximately 10% of known male sterile material in plants is nuclear-based.
Cytoplasmic Male Sterility (CMS)
- Location: The genes responsible for sterility are found in the cytoplasm, specifically within the mitochondria.
- Genetic Control: While the abortive gene is mitochondrial, cytoplasmic physiology is shaped by the interaction between nuclear genes and the coordinated cytoplasm.
- Inheritance Patterns: It is inherited maternally via the cytoplasm rather than through standard Mendelian patterns of the nucleus.
- Restoration of Fertility: CMS can be overcome by nuclear restorer genes found in the cytoplasm donor parent, which restore fertility to the offspring.
Comparison and Practical Application
- Origin: CMS is a frequent outcome of distant hybridization; at least 70% of CMS material results from interspecific or intergeneric crosses, compared to only 10% for nuclear male sterility.
- Hybrid Seed Production: CMS is a critical, cost-effective tool for the commercial production of hybrid seeds in crops such as rice, wheat, and pigeon pea. It simplifies seed production by eliminating the need for manual emasculation; breeders use “maintainer” lines to propagate the sterile line and “restorer” lines to produce fertile hybrid offspring for the farmer.
- Genotypic Stability: In distant hybridization, the cell nucleus of the cytoplasmic donor can restore fertility through nuclear replacement, indicating that the hybridization process itself does not typically cause cytoplasmic mutations but rather creates new, sometimes incompatible, nuclear-cytoplasmic combinations.
15. How does chromosome doubling specifically eliminate hybrid sterility?
Chromosome doubling specifically eliminates hybrid sterility by ensuring that every chromosome in the hybrid genome has a matching homologous partner to pair with during meiosis.
The Core Problem: Meiotic Failure
In distant hybridization (interspecific or intergeneric), the resulting F1 hybrid often possesses a single set of chromosomes from each parent species. Because these parental species are distantly related, their chromosomes frequently lack structural homology. This mismatch leads to several meiotic failures:
- Lack of Synapsis: The divergent chromosomes cannot pair up normally during prophase I.
- Abnormal Separation: Without proper pairing, chromosomes may scatter throughout the spindles, lag behind during anaphase, or separate unequally.
- Non-functional Gametes: These irregularities prevent the formation of normal, viable gametes, resulting in complete self-sterility.
The Solution: Restoring Homology via Doubling
Chromosome doubling—typically achieved by treating germinating seeds or seedlings with chemical reagents like colchicine—artificially replicates the entire chromosome set.
- Providing Partners: By doubling the chromosome number, the hybrid changes from having one set of divergent chromosomes to having two identical copies of every chromosome.
- Normal Synapsis: During meiosis, each individual chromosome now has an identical homologous partner for pairing and synapsis. Pairing occurs within each identical set rather than between the mismatched parental sets.
- Viable Gamete Production: With normal pairing restored, meiotic division can proceed accurately, leading to the production of functional, “energetic” gametes.
Result: The Amphidiploid
This process converts a sterile hybrid into a fertile amphidiploid (also known as an allotetraploid or allopolyploid). This technique is so effective that it can even overcome cryptic structural hybridity—sterility caused by structural changes that are too small to be detected under a microscope during meiosis. Famous practical examples of this success include the creation of Triticale (wheat × rye) and Raphanobrassica (radish × cabbage).
16. What are Chromosome Segment Substitution Lines (CSSLs) and their benefits?
Chromosome Segment Substitution Lines (CSSLs) are specialized genetic stocks in which specific, well-defined segments of a recipient species’ genome (typically an elite cultivar) are replaced by corresponding segments from a donor species (often a wild relative). They are produced using conventional breeding techniques—primarily repeated backcrossing—coupled with molecular marker technology to precisely track and select the substituted segments.
Benefits of CSSLs in Plant Breeding
CSSLs offer several powerful advantages for both basic genetic research and practical crop improvement:
- High-Resolution Mapping and Gene Cloning: CSSLs are essential tools for identifying and cloning Quantitative Trait Loci (QTLs). By isolating a single donor segment within a uniform elite background, researchers can more easily study the effects of specific genes without the interference of other donor genetic material. In rice, CSSLs have been successfully used to clone genes for key yield components, including grain size, weight, and the number of tillers.
- Functional Genomics: These lines provide a stable platform for studying the function of genes that govern complex agronomic traits. They allow researchers to observe how specific alien segments interact with the host genome and influence trait expression.
- Mitigation of Linkage Drag: One of the most significant challenges in distant hybridization is “linkage drag,” where undesirable wild traits are inherited alongside beneficial ones. CSSLs help minimize this by allowing breeders to select for the smallest possible substitution segments that still contain the target gene, thereby excluding unwanted linked genes.
- Broadening the Genetic Base: CSSLs serve as a bridge to introgress novel genetic variation from unadapted germplasm or wild relatives into elite breeding lines. This is vital for introducing “super genes” for biotic and abiotic stress resistance that may have been lost during crop domestication.
- Development of Molecular Tools: The creation of these lines facilitates the construction of highly saturated genetic maps and the development of new molecular markers. These tools can then be used in marker-assisted selection (MAS) to accelerate traditional breeding programs.
- Researching Genome Interactions: CSSLs provide a basis for studying homologous chromosome pairing, genome stability, and the expression of alien fragments within a new genetic environment.
17. How does protoplast fusion help when gametes fail to fuse?
Protoplast fusion, a form of somatic hybridization, is an in vitro technique used to bypass the natural barriers of sexual reproduction when gametes fail to fuse. This method allows breeders to combine genetic material from species or genera that are otherwise sexually incompatible.
How Protoplast Fusion Works
The process involves several key steps to achieve genetic recombination without the need for pollination or fertilization:
- Preparation: Somatic cells from parental plants are treated with lytic enzymes to remove their cell walls, creating naked cells known as protoplasts.
- Induced Fusion: Two or more genetically different protoplasts are brought into contact and induced to fuse using physical or chemical agents, such as polyethylene glycol (PEG) or electrofusion.
- Regeneration: The resulting hybrid protoplasts (heterokaryons) regenerate cell walls and are cultivated in a nutrient medium to differentiate into whole plants.
Types of Somatic Hybrids
Protoplast fusion provides flexibility in the amount of genetic material transferred:
- Symmetric Hybrids: These combine the complete nuclear and cytoplasmic information of both parents, often resulting in amphidiploid combinations.
- Asymmetric Hybrids: Only fragments of the nuclear genome from a donor species are introduced into the recipient’s genome, often achieved by irradiating the donor protoplasts to destroy their nuclei before fusion.
- Cybrids (Cytoplasmic Hybrids): These contain the nuclear genome of one parent and the cytoplasmic genome (mitochondria or chloroplasts) of the other, or a combination of both.
Benefits in Overcoming Incompatibility
- Bypassing Pre-fertilization Barriers: When gametes fail to fuse due to pollen tube arrest, lack of recognition, or morphological mismatches, protoplast fusion provides a parasexual alternative to merge genomes.
- Accessing Distant Gene Pools: It allows for the transfer of “super genes” for disease resistance, stress tolerance, or improved yield from wild relatives that cannot be crossed conventionally.
- Intergeneric Combinations: The technique can bridge extremely wide gaps, such as the successful creation of somatic hybrid plantlets from rice × barley or rice × barnyard grass.
Constraints and Limitations
While powerful, protoplast fusion is technically demanding and carries certain risks:
- Genomic Instability: Somatic hybrids are frequently aneuploid (gaining or losing individual chromosomes) or may undergo unexpected polyploidization during tissue culture.
- Sterility: Hybrids produced through this method, particularly asymmetric ones, can be sterile, requiring repeated backcrossing to the cultivated parent to restore fertility and eliminate undesirable wild traits.
- Specialized Requirements: Successful implementation requires highly skilled personnel, well-equipped tissue culture laboratories, and efficient regeneration protocols, which are not yet available for all crop species.
18. What is the difference between NMS and CMS?
The primary difference between Nuclear Male Sterility (NMS) and Cytoplasmic Male Sterility (CMS) lies in the cellular location of the sterile genes and their inheritance patterns.
Nuclear Male Sterility (NMS)
- Genetic Location: NMS refers to the presence of sterile genes within the cell nuclei of the plant.
- Genetic Control: It can be governed by a single gene or through multi-gene interactions.
- Common Traits: Most nuclear male sterility is controlled by recessive genes, although single gene-controlled dominant forms can occur through mutations, such as in Taigu genic male-sterile wheat.
- Frequency: Approximately 10% of male sterile material identified in plants is classified as nuclear.
Cytoplasmic Male Sterility (CMS)
- Genetic Location: CMS is primarily controlled by genetic material found in the cytoplasm, specifically the mitochondria.
- Genetic Control: While the abortive gene is mitochondrial, the physiological and biochemical expressions are shaped by nuclear gene regulation and interactions.
- Origin and Restoration: In distant hybridization, CMS often results from the interaction between a new nuclear genotype and a coordinated foreign cytoplasm. Fertility can be restored through “nuclear replacement” using restorer genes from the cytoplasmic donor parent.
- Frequency: At least 70% of male sterile material resulting from interspecific or intergeneric hybridization is cytoplasmic.
Practical Breeding Applications
CMS is considered a cost-effective tool for the commercial production of hybrid seeds in crops like rice, wheat, and pigeon pea. It simplifies the seed production process by eliminating the need for manual emasculation, allowing breeders to use “maintainer” lines to propagate the sterile line and “restorer” lines to produce fertile hybrid offspring for farmers. While NMS is also used, it is less common in commercial large-scale hybrid systems compared to the widely adopted CMS systems like the “Wild Abortive” (WA) source in rice.
19. Describe the main differences between interspecific and intergeneric hybridization.
Interspecific and intergeneric hybridization are the two primary types of distant (wide) hybridization, distinguished by the taxonomic distance between the parents and the severity of the resulting reproductive barriers.
Parents Involved and Taxonomic Distance
- Interspecific Hybridization: This involves crossing two different species that belong to the same genus. It is often referred to as intrageneric hybridization.
- Intergeneric Hybridization: This involves crossing two different genera within the same family. The parents are taxonomically more distantly related than those in interspecific crosses.
Fertility and Seed Setting
- Fertility of Offspring: Interspecific hybrids vary in their fertility, ranging from fully fertile to completely sterile depending on the degree of chromosomal homology between the parents. In contrast, F1 hybrids between two different genera are always sterile. Fertility in these sterile hybrids must usually be restored through chromosome doubling using reagents like colchicine.
- Seed Setting: The rate of seed setting is generally higher in interspecific crosses compared to intergeneric crosses, where seed set is typically very low or impossible without advanced intervention.
Practical Utility and Breeding Outcomes
- Use in Crop Improvement: Interspecific hybridization is widely used and highly successful in breeding programs for transferring specific “super genes” (e.g., disease and pest resistance) from wild relatives into cultivated crops. Intergeneric hybridization is infrequently employed because of the extreme biological obstacles involved.
- Evolution of New Species: Interspecific crosses often lead to introgression, where specific genes are infiltrated into the genome of another species. Intergeneric hybridization is more often associated with the synthesis of entirely new crop species that do not exist in nature, such as Triticale (wheat × rye) and Raphanobrassica (radish × cabbage).
- Success in Propagation Types: Interspecific hybridization is particularly effective in vegetatively propagated species like sugarcane and potato. Intergeneric hybridization is most commonly successful in asexually propagated species, particularly ornamentals like orchids and lilies, where novelty is highly valued.
Summary of Key Differences
| Feature | Interspecific Hybridization | Intergeneric Hybridization |
|---|---|---|
| Taxonomic Level | Between species of the same genus. | Between different genera of the same family. |
| F1 Fertility | Varies from fully fertile to sterile. | Always sterile naturally. |
| Seed Setting | Relatively high. | Very low or zero. |
| Primary Goal | Trait introgression from wild relatives. | Germplasm innovation and new species creation. |
| Examples | G. hirsutum × G. barbadense (Cotton). | Triticum × Secale (Triticale). |
20. What is the difference between alien addition and substitution?
In plant genetics and chromosome engineering, the difference between alien addition and substitution depends on whether the foreign genetic material is added to the host’s genome or replaces a portion of it. Both techniques are used to introduce “super genes” or desirable traits from wild relatives into cultivated crops.
Alien Addition
Alien addition involves adding one or more chromosomes (or fragments) from a donor species to the complete, normal chromosome set of the recipient species.
- Mechanism: It creates a new genetic type by incorporating extra genetic material without removing any existing chromosomes.
- Chromosome Number: This process increases the total number of chromosomes in the plant. For example, in rice (normally 2n = 24), the creation of “monosomic alien addition lines” results in a chromosome complement of 2n = 25.
- Examples: This technique has been used in wheat to introduce disease resistance from wild relatives and in Brassica carinata to produce “disomic addition lines” (the normal set plus two extra chromosomes).
Alien Substitution
Alien substitution involves replacing one or more host chromosomes or specific segments with corresponding genetic material from the alien donor species.
- Mechanism: Rather than just adding new material, the foreign segment or chromosome takes the place of a host counterpart.
- Chromosome Number: The total chromosome count typically remains the same as the host species, but the genetic composition is modified.
- CSSLs: A common application is the development of Chromosome Segment Substitution Lines (CSSLs). These are specialized genetic stocks where specific, defined segments of an elite cultivar are replaced by segments from a wild relative, allowing for high-resolution mapping and precise gene cloning.
Summary of Differences
| Feature | Alien Addition | Alien Substitution |
|---|---|---|
| Action | Adds to the host genome. | Replaces a portion of the host genome. |
| Chromosome Count | Increases the total number. | Typically remains the same. |
| Primary Use | Creating new types with extra info. | Mapping, gene cloning, and functional genomics. |
These methods serve as a foundation for constructing genetic maps, identifying molecular markers, and researching genome interactions.
References
Ameer Sohail, S. N., & Singh Sran, R. (2021). Distant Hybridization and Its Limitations. INTERNATIONAL JOURNAL OF INNOVATIVE RESEARCH IN TECHNOLOGY, 8(4).
Avinashe, H., Dubey, N., & Professor, A. (2018). Issue 11 www.jetir.org (ISSN-2349-5162). In JETIRDR06028 Journal of Emerging Technologies and Innovative Research (Vol. 5). www.jetir.org
Biswal, M., Soni, A., Payasi, D. K., Krishi, J. N., & Vidyalaya, V. (n.d.). Distant Hybridization in Crop Improvement Soumya Patel. In Strategy & Application of Plant Breeding. Retrieved https://www.kdpublications.in
Brar, D. S., Singh, K., & Khush, G. S. (2017). Frontiers in Rice Breeding. In The Future Rice Strategy for India (pp. 137–160). Elsevier. https://doi.org/10.1016/B978-0-12-805374-4.00006-3
Chen, J., Luo, M., Li, S., Tao, M., Ye, X., Duan, W., Zhang, C., Qin, Q., Xiao, J., & Liu, S. (2018). A comparative study of distant hybridization in plants and animals. In Science China Life Sciences (Vol. 61, Number 3, pp. 285–309). Science in China Press. https://doi.org/10.1007/s11427-017-9094-2
Debnath, P., Chakma, K., Bhuiyan, M. S. U., Thapa, R., Pan, R., & Akhter, D. (2024). A novel multi trait genotype ideotype distance index (MGIDI) for genotype selection in plant breeding: Application, prospects, and limitations. In Crop Design (Vol. 3, Number 4). Elsevier B.V. https://doi.org/10.1016/j.cropd.2024.100074
Kashyap, A., Garg, P., Tanwar, K., Sharma, J., Gupta, N. C., Ha, P. T. T., Bhattacharya, R. C., Mason, A. S., & Rao, M. (2022). Strategies for utilization of crop wild relatives in plant breeding programs. In Theoretical and Applied Genetics (Vol. 135, Number 12, pp. 4151–4167). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00122-022-04220-x
Kumar, J., & Pratap, A. (2014). Alien gene transfer: Challenges and opportunities. In Alien Gene Transfer in Crop Plants, Volume 1: Innovations, Methods and Risk Assessment (pp. 289–307). Springer New York. https://doi.org/10.1007/978-1-4614-8585-8_12
Li, W., Xu, Y., Yan, J., Guo, Y., Cai, M., & Zhang, Q. (2025). The cross compatibility and endogenous hormone variation of distant hybridization between lagerstroemia indica and lythrum salicaria. Scientia Horticulturae, 350. https://doi.org/10.1016/j.scienta.2025.114347
Mo, X., Wang, Y., Huang, Y., Zeng, Z., & Yan, C. (2025). Cytological Observation of Distant Hybridization Barrier and Preliminary Investigation of Hybrid Offspring in Tea Plants. Plants, 14(13). https://doi.org/10.3390/plants14132061
Mwangangi, I. M., Kiilu Muli, J., & Neondo, J. O. (2019). Plant Hybridization as an Alternative Technique in Plant Breeding Improvement. Asian Journal of Research in Crop Science, 1–11. https://doi.org/10.9734/ajrcs/2019/v4i130059
Rabbani, M. T., & Nayak, G. (2023). Recruiting Distant Hybridization for Reshaping Meiotic Recombination. In Plant Breeding and Biotechnology (Vol. 11, Number 3, pp. 168–184). Korean Society of Breeding Science. https://doi.org/10.9787/PBB.2023.11.3.168
Reddy, C. S., Ramireddy, S., & Reddy, U. K. (2024). Widening Genetic Diversity Using Embryo Rescue in Cucurbit Crops: A Review. In Plants (Vol. 13, Number 10). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/plants13101320
Saeed, A., & Fatima, N. (2021). Wild Germplasm: Shaping Future Tomato Breeding. In Wild Germplasm for Genetic Improvement in Crop Plants (pp. 201–214). Elsevier. https://doi.org/10.1016/B978-0-12-822137-2.00011-4
Sargar, P., Deskhmuch, S., Naik, G., Baraskar, S., & Mehtre, S. (2023). Distant Hybridization. In Elements of Plant Breeding (Vol. 1, 5).
Shinozaki, K., & Yamaguchi-Shinozaki, K. (2022). Functional genomics in plant abiotic stress responses and tolerance: From gene discovery to complex regulatory networks and their application in breeding. Proceedings of the Japan Academy Series B: Physical and Biological Sciences, 98(8), 470–492. https://doi.org/10.2183/pjab.98.024
Singh, D. P., Singh, A. K., & Singh, A. (2021). Wide hybridization. In Plant Breeding and Cultivar Development (pp. 159–178). Elsevier. https://doi.org/10.1016/b978-0-12-817563-7.00028-3
Singh, H. P., Raigar, O. P., & Chahota, R. K. (2022). Estimation of genetic diversity and its exploitation in plant breeding. In Botanical Review (Vol. 88, Number 3, pp. 413–435). Springer. https://doi.org/10.1007/s12229-021-09274-y
Van Huylenbroeck, J., & Bhattarai, K. (2022). Ornamental plant breeding: entering a new era? Ornamental Horticulture, 28(3), 297–305. https://doi.org/10.1590/2447-536X.v28i3.2516
Visarada, K. B. R. S., & Venkateswaran, K. (2018). Wide hybridization. In Breeding Sorghum for Diverse End Uses (pp. 131–139). Elsevier. https://doi.org/10.1016/B978-0-08-101879-8.00008-5
Wu, Z. Y., Milne, R. I., Liu, J., Nathan, R., Corlett, R. T., & Li, D. Z. (2023). The establishment of plants following long-distance dispersal. In Trends in Ecology and Evolution (Vol. 38, Number 3, pp. 289–300). Elsevier Ltd. https://doi.org/10.1016/j.tree.2022.11.003
Würschum, T., Zhu, X., Zhao, Y., Jiang, Y., Reif, J. C., & Maurer, H. P. (2023). Maximization through optimization? On the relationship between hybrid performance and parental genetic distance. In Theoretical and Applied Genetics (Vol. 136, Number 9). Springer Science and Business Media Deutschland GmbH. https://doi.org/10.1007/s00122-023-04436-5
Yaşar, S., Gehrke, F., Capdeville, N., & Puchta, H. (2026). Recent progress in plant genome engineering: from large insertions to chromosome number changes. In Current Opinion in Biotechnology (Vol. 97). Elsevier Ltd. https://doi.org/10.1016/j.copbio.2025.103426
Zakharova, E. V., Ulianov, A. I., Golivanov, Y. Y., Molchanova, T. P., Orlova, Y. V., & Muratova, O. A. (2025). Pollen–Pistil Interaction During Distant Hybridization in Plants. In Agronomy (Vol. 15, Number 7). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/agronomy15071732
Zhang, X., Ren, G., Li, K., Zhou, G., & Zhou, S. (2012). Genomic variation of new cultivars selected from distant hybridization in Lilium. Plant Breeding, 131(1), 227–230. https://doi.org/10.1111/j.1439-0523.2011.01906.x