
1. Introduction: The Strategic Frontier of Whole-Genome Duplication (WGD)
In the high-stakes landscape of botanical evolution and agricultural biotechnology, whole-genome duplication (WGD) represents a transformative phenomenon rather than a mere chromosomal aberration. As a Senior Cytogeneticist, I view WGD through the lens of the “C-value paradox” and the “two-round hypothesis,” acknowledging that while DNA content does not always correlate with organismal complexity, the ancient duplications at the base of vertebrate and teleost lineages underscore polyploidy as a recurring driver of genomic innovation. Polyploidy is a prime facilitator of rapid speciation because it establishes instantaneous reproductive isolation, allowing a newly formed lineage to bypass the genetic bottleneck of hybrid sterility and immediately begin the colonization of new, stressful niches.
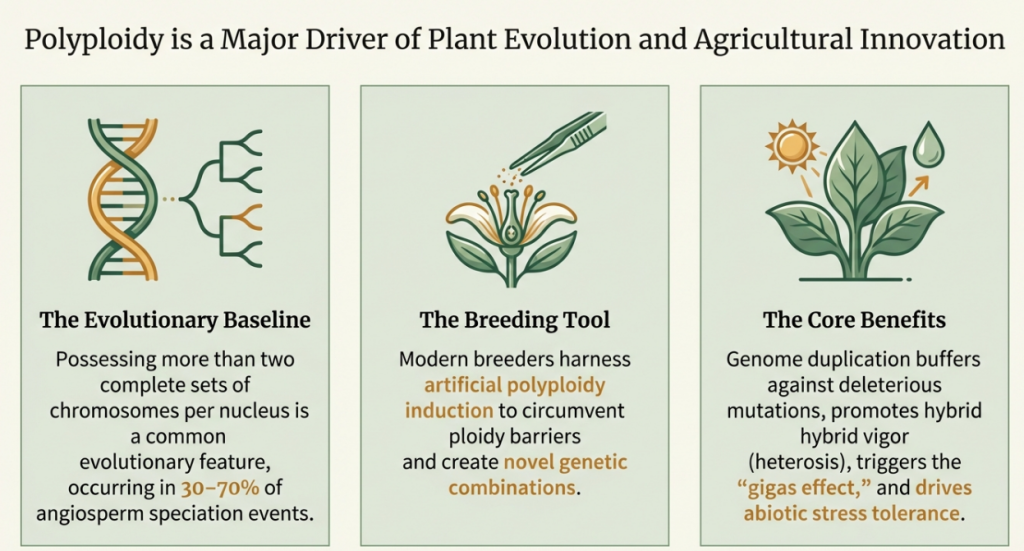
By definition, True Polyploidy is a genetic configuration where both somatic and germline cells contain more than two homologous sets of chromosomes. This genomic redundancy serves as a strategic facilitator for modern breeding, offering a buffer against environmental stochasticity and providing the raw material for biochemical novelty. This document analyzes the mechanistic pathways of induction, the fundamental “nucleotypic” shifts in cellular geometry, and the physiological manifestations that define the next generation of resilient, high-value cultivars.
Diploid vs. Polyploid: Physical Architecture
| Trait | Diploid (2n) | Polyploid (4n+) |
|---|---|---|
| Cell Volume | Standard | Significantly Increased (~2x) |
| Surface-Area-to-Volume Ratio | Higher | Lower (approx. 1.58x increase in SA vs 2x Vol) |
| Stomatal Profile | Denser, smaller pores | Lower pore space per unit area; larger pores |
| Leaf Thickness | Standard | Increased (Thicker mesophyll) |
| Growth Strategy | Rapid (r-selection) | Slower, sturdier (K-selection) |
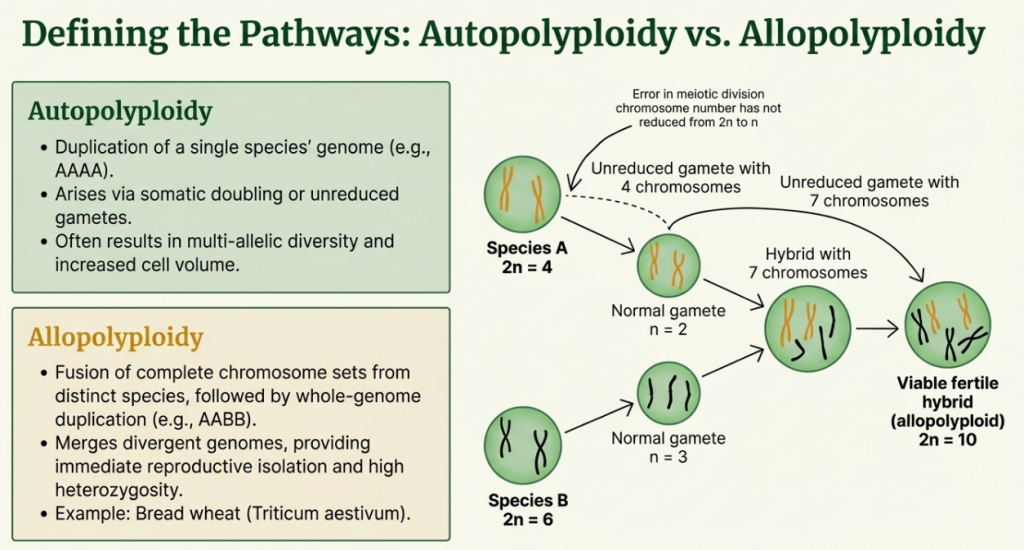
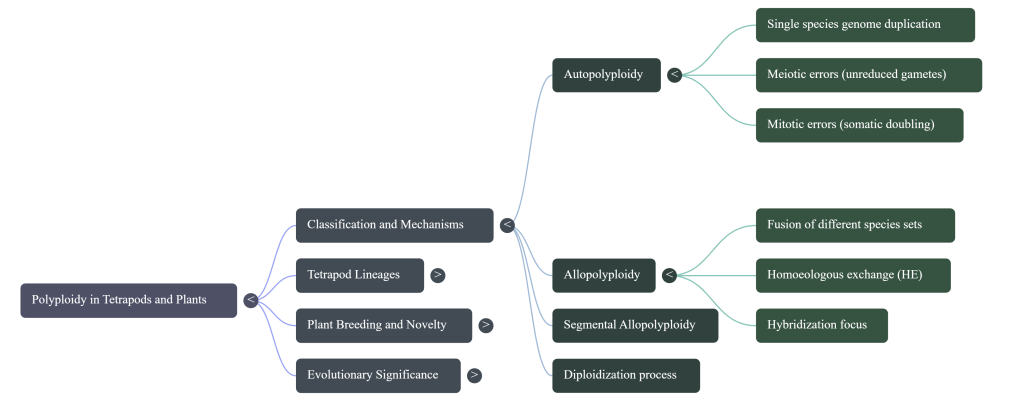
2. Mechanistic Pathways: Autopolyploidy vs. Allopolyploidy
The strategic utility of a polyploid lineage is dictated by its biological origin. Breeders must distinguish between the mere multiplication of a single genome and the sophisticated fusion of divergent ones to predict the stability of the resulting germplasm.
- Autopolyploidy: This pathway involves the duplication of the chromosome set of a single species, typically through mitotic errors (somatic doubling) or meiotic errors (gametic non-reduction). While autopolyploids benefit from increased biomass and cell size, they often struggle with inbreeding depression and reduced fertility due to the formation of multivalents (quartets or sextets) during meiosis, which leads to unequal chromosome segregation and aneuploidy.
- Allopolyploidy: Arising from the fusion of genomes from two different species followed by WGD, allopolyploidy is the bedrock of many successful crops. It stabilizes hybrid derivatives by allowing an escape from hybrid sterility. Crucially, allopolyploids exhibit Homoeologous Exchange (HE)—recombination between regions of high similarity in the parental genomes. This process fuels neo-functionalization and sub-functionalization, where duplicated genes evolve new roles or partition ancestral ones, significantly expanding the plant’s adaptive toolkit.
| Feature | Autopolyploidy | Allopolyploidy |
|---|---|---|
| Definition | Duplication of a single species’ genome. | Fusion and doubling of genomes from different species. |
| Genetic Homology | Complete homology; chromosomes form quartets/sextets. | Homoeologs from parental sets form pairs. |
| Meiotic Behavior | Formation of multivalents; higher risk of aneuploidy. | Formation of bivalents; superior genetic stability. |
| Evolutionary Outcome | Increased biomass; redundancy buffers environmental noise. | Functional novelty through Homoeologous Exchange (HE). |
| Mechanism of Origin | Mitotic/meiotic errors within a single species (somatic doubling/non-reduction). | Interspecific hybridization followed by WGD. |
| Selection Strategy | Shift toward K-selection (stress tolerance/longevity). | Exploitation of transgressive traits and hybrid vigor. |
In allopolyploids, Homoeologous Exchange (HE) is a critical driver of novelty. When parental genomes interact, HE promotes neo-functionalization and sub-functionalization, creating novel transcripts and biochemical pathways. This genetic reorganization sets the stage for the physical changes in cellular geometry that drive the polyploid phenotype.
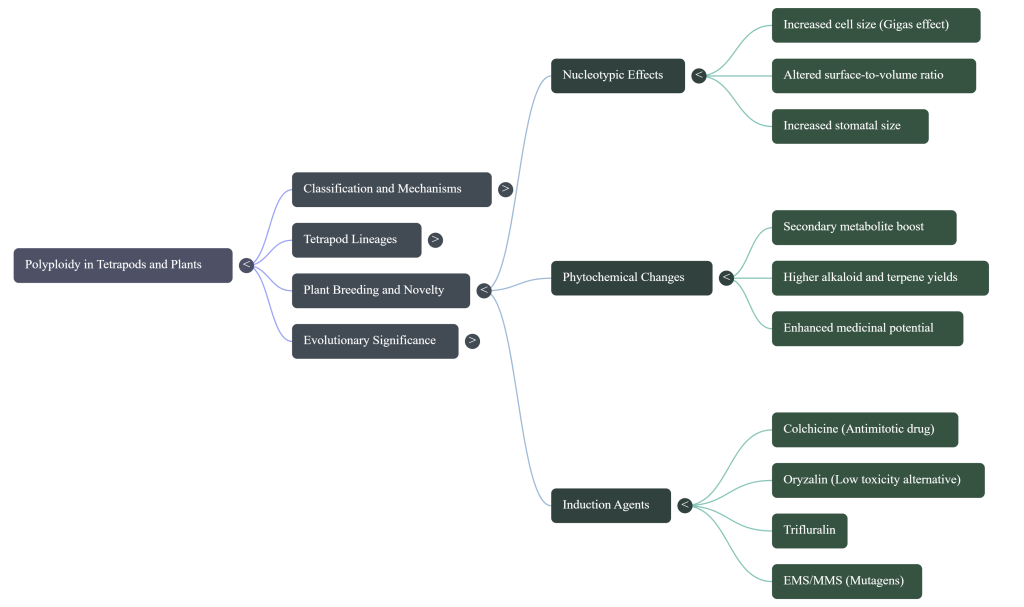
3. The Nucleotypic Effect: Cellular and Genomic Consequences
Polyploidy triggers a fundamental shift in cellular geometry and metabolic regulation, known as the nucleotypic effect. Chromosome doubling is not just a change in the DNA template; it is a physical restructuring of the cell’s operational parameters.
Surface-to-Volume Ratio and Metabolic Partitioning
Doubling the genome typically increases cell volume by approximately 2x, while the surface area only increases by ~1.58x. This shift alters the balance of enzymatic activity: glycosidases associated with the cell wall are linked to surface area, while prime metabolic enzymes associated with internal organelles are linked to volume. This geometric imbalance can fundamentally change metabolic rates and flux.
| Metric | Change in Tetraploids |
|---|---|
| Volume | Increases by approximately 2.0x |
| Surface Area | Increases by approximately 1.58x |
| Surface-to-Volume Ratio | ~21% Reduction |
Nucleocytoplasmic Transfer Constraints
In autotetraploids, nuclear pore density remains relatively constant, meaning there are fewer pores per unit of cell volume. This creates a bottleneck for nucleocytoplasmic transfer, specifically retarding the movement of messenger and ribosomal RNA from the nucleus to the cytoplasm. This limitation often manifests as a slower overall growth rate compared to diploid progenitors.
- The RNA Bottleneck: Nuclear pore density remains relatively constant (e.g., ~16-17 pores/µm² in Hordeum vulgare), meaning the rate of RNA transfer is limited by the surface area of the nuclear membrane.
- Cellular Compensation: To overcome this, polyploid cells develop complex nuclear lobes and cytoplasmic invaginations. This is not merely a survival fix; it is the essential physical infrastructure that permits higher metabolic output and the synthesis of complex compounds.
Gene Activity and Redundancy
The presence of multiple DNA templates provides a buffer against environmental “noise,” but the genomic response is rarely linear:
- Dosage Compensation: Not all genes increase activity by 2x; many undergo silencing to maintain metabolic homeostasis.
- Novel Heteromers: Polyploidy allows for the synthesis of “hybrid enzymes” or novel heteromers, expanding biochemical versatility and allowing the plant to maintain metabolic flux under extreme stress.
Within this framework, chromosome doubling serves as a mechanism for “punctuated evolution within a microevolutionary time scale,” producing “macromutants which may offer a population novel avenues of response to the exigencies of the environment.”
4. Physiological and Developmental Novelty in Polyploid Crops
The primary objective of polyploid breeding is often the manifestation of “Gigas” properties—the physical enlargement and enhancement of plant structures. These cellular transformations translate into significant commercial advantages.

Gas Exchange and Water-Use Efficiency
WGD consistently alters the stomatal apparatus. While stomatal density typically declines, the increase in stomatal size is significant. The resulting reduction in transpiration rates—improves water-use efficiency, providing a robust defense against drought.
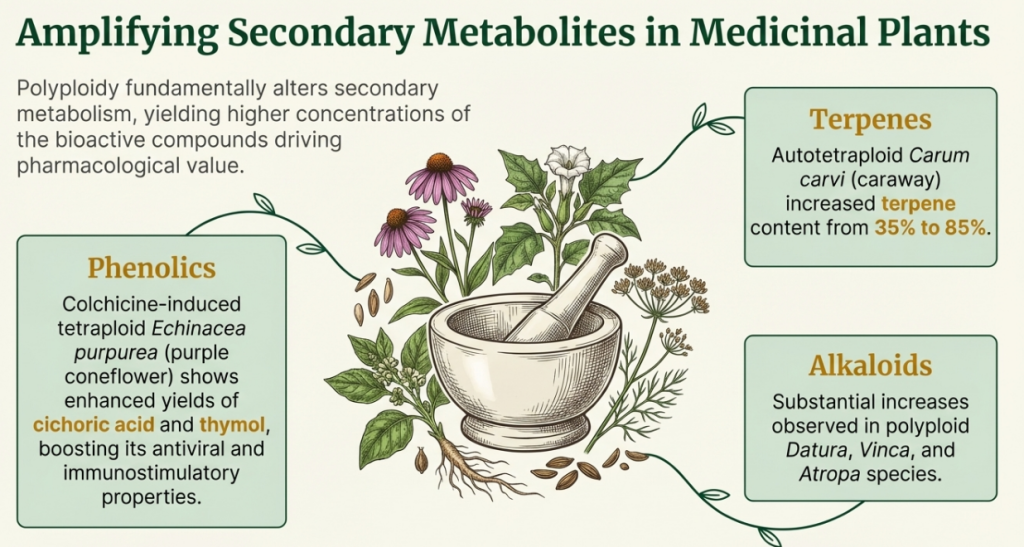
Secondary Metabolism: The “Why” of Yield Increases
Polyploidy sharply increases the concentration of alkaloids, terpenes, and flavonoids. These increases are driven by increased allelic diversity and altered gene expression patterns. For example, in Digitalis lanata, digoxin and digitoxin levels increase by 1.6x–1.7x. In Mentha spicata, hexaploid lines induced via oryzalin show higher carvone and limonene concentrations, unlocking new value for the pharmaceutical and cosmetic sectors.
The Biochemical Yield Advantage For medicinal and aromatic crops, WGD offers a significant strategic advantage in secondary metabolite yields:
- Alkaloid and Terpene Surge: Polyploid plants often exhibit a dramatic increase in secondary metabolites that deter herbivores. In many medicinal and wild species, alkaloid yields can surge by over 100% following genome duplication. Documented yield spikes in Datura, Vinca, and Atropa. Increases of 30% to 85% in Mentha arvensis and Carum carvi.
- Enhanced Flavonoids: Doubling the genome often leads to qualitative shifts in flavonoid profiles, providing better internal antioxidants and protection against ultraviolet radiation.
- Novelty: Expression of heat-stable flavonoid isomerases (e.g., Briza media) not found in diploid progenitors.
- Nutrient Density: 18-24% increases in calcium and magnesium accumulation in certain spearmint genotypes.
Developmental Shifts and K-Selection
Polyploidy shifts plants toward the K-selection end of the evolutionary spectrum. This is characterized by slower growth rates, delayed flowering, and a transition from annual to perennial habits. A classic example is Oryza punctata, where the tetraploid form survives for 2 to 9 years, whereas the diploid form rarely exceeds one year. This longevity is a strategic advantage for stabilizing yields in perennial systems.
Developmental Rhythms The high DNA content in polyploids is inversely correlated with developmental speed, leading to:
- Seed Dynamics: Seeds (e.g., Glycine max) can be up to 200% larger, though they germinate more slowly.
- Survival Dormancy: Increased seed dormancy in Oryza punctata acts as a critical survival mechanism in unpredictable environments.
- Flowering Schedules: Delays of several weeks (e.g., Ocimum kilimandscharicum) can reprogram the plant’s ecological relationship with pollinators and pests.
The Annual-to-Perennial Transition One of the most profound strategic shifts is the life-cycle extension. In Oryza punctata, diploid forms are strictly annuals, whereas their tetraploid counterparts undergo an annual-to-perennial transition, surviving for two to nine years. Similarly, autotetraploid Zea mays and Trifolium pratense exhibit increased longevity and winter hardiness, providing long-term stability for agricultural systems.
Transgressive Tolerance and Environmental Resilience
“Transgressive tolerance” allows polyploids to occupy habitats beyond the ecological limits of their diploid progenitors. This resilience is the cumulative result of nucleotypic and biochemical shifts.
- Drought and Water Status: Reduced stomatal density and increased leaf diffusive resistance lower transpiration rates. In Phlox drummondii, tetraploids achieve peak performance in significantly drier conditions than diploids.
- Nutrient Stress: Tetraploids exhibit superior Ion Uptake Efficiency. In Beta vulgaris and wheat, increased uptake of potassium and phosphorus allows for high biomass production on nutrient-deficient or calcareous soils.
- Cold and Frost Resilience: Temperature optima for photosynthesis shift; Dactylis glomerata tetraploids develop more rapidly during winter and maintain higher photosynthetic rates at low temperatures.
- Pest and Pathogen Defense: Enhanced resistance to fungi (e.g., leaf-rust in Glycine tabacina) and herbivores (e.g., aphids in Brassica campestris) is directly linked to higher concentrations of alkaloids and terpenes.
- Leaf-Rust Resistance: In Glycine tabacina, research shows a stark contrast in survival: 42% of polyploid individuals exhibit resistance to leaf-rust fungus, compared to a mere 14% of diploids.
- Sclerotinia Resistance: Polyploid red clover (Trifolium pratense) has demonstrated significantly higher resistance to clover rot and nematodes than its diploid progenitors.

4.1 The Metabolic Speed Limit: Why Polyploids Grow Slower
A central explanation for the slower growth of polyploid plants is the Nuclear Pore Limitation theory. All genetic instructions (RNA) must travel from the nucleus to the cytoplasm through nuclear pores. Because the number of pores per unit area of the nuclear membrane remains constant, the rate of RNA movement is strictly limited by the membrane’s surface area.
Furthermore, polyploidy lowers the ratio of the nuclear membrane area to chromatin volume. With more chromatin in contact with the nuclear membrane and more chromatin in condensed regions, the patterns of gene expression and regulation are fundamentally altered.
Primary Reasons for Retarded Growth
- Nucleocytoplasmic Transfer: Because the nuclear membrane area only increases by ~1.58x while volume may double, there are fewer “exit doors” (pores) per unit of cell volume, creating a traffic jam for genetic information.
- Enzyme Activity Shifts: Doubling the genes does not necessarily double the output. Major enzymes for photosynthesis, such as RuBisCO, often show significantly reduced activity (up to 10-30% less) in polyploids compared to diploids.
- Developmental Rates: There is a strong inverse correlation between DNA content and the speed of the life cycle. Polyploids experience slower cell division, leading to delayed flowering, slower seed germination, and longer generation times.
While this “slow-motion” biology may seem disadvantageous, it represents a strategic pivot from rapid growth to a model of increased durability and resource management.
4.2 The Quality Trade-Off: Durability and Defense
Evolutionarily, polyploidy shifts a plant along the r-K selection continuum. While diploid plants often favor “r-selection” (rapid reproduction), polyploids move toward “K-selection.” This represents a resource management strategy: slow-growing plants have modest requirements and are less likely to exhaust the resources of their immediate environment, allowing them to build reserves for favorable periods.
Diploid (Standard) vs. Polyploid (Enhanced) Traits
| Category | Diploid (Standard) | Polyploid (Enhanced) |
|---|---|---|
| Environmental Tolerance | Standard baseline | High resistance to cold, drought, and nutrient-poor soils. |
| Pest Resistance | More vulnerable | Increased resistance to pathogens (e.g., rust fungus) and herbivores. |
| Longevity | Often Annual | Frequent shift toward Perennial habits (longer lifespans). |
This increased resilience is largely driven by “Secondary Metabolites.” Polyploids often produce significantly higher quantities of defensive chemicals, such as alkaloids, terpenes, and essential oils. These compounds act as natural pesticides and environmental shields, providing the plant with unique tolerances that exceed the limits of its diploid progenitors. These internal chemical shifts are reflected in striking, visible physical changes.
4.3 The “Gigas” Effect: Visible Structural Changes
The term “Gigas” (derived from the Greek for “giant”) refers to the oversized, robust physical characteristics common in polyploid plants. However, these are not just “big” plants; they are re-architected organisms with altered anatomical proportions.
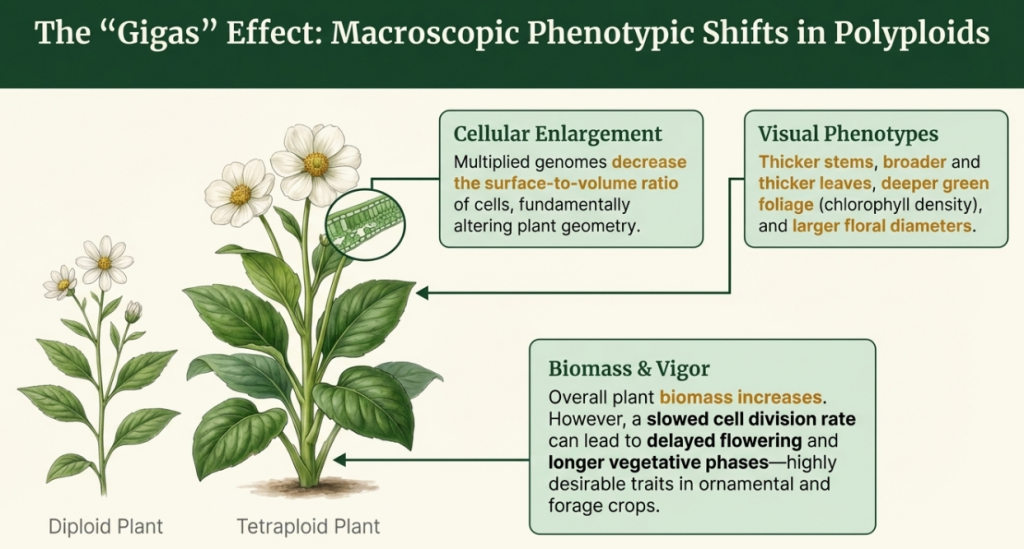
- Leaf Morphology: Polyploid leaves are typically thicker, wider, and a darker green. They often feature increased pubescence (hairs), which provides a layer of environmental protection.
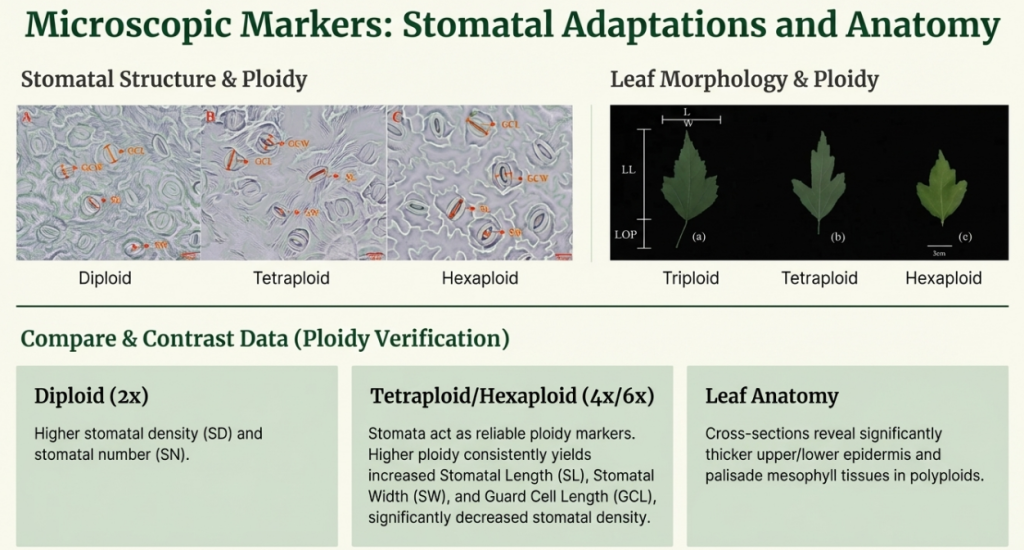
- Stomatal Changes: The “breathing pores” (stomata) become larger but less dense. Crucially, the increase in size usually does not counterbalance the decline in number, meaning the stomatal pore space per unit area is less at higher ploidal levels.
- Floral and Seed Traits: These plants produce larger flowers and larger seeds. While the seeds may be fewer in number, they are often more vigorous and possess greater dormancy, which is an ecological advantage for long-term survival.
These physical markers—larger stomata with reduced pore space and thicker tissues—provide the mechanism for superior water retention and cold resistance, which is why humans intentionally induce these traits in agriculture and medicine.
5. Chemical Induction Strategies: Colchicine and Emerging Alternatives
Since natural WGD is a rare event, artificial chromosomal doubling is essential for bypassing evolutionary “slow-walks” in commercial breeding programs.
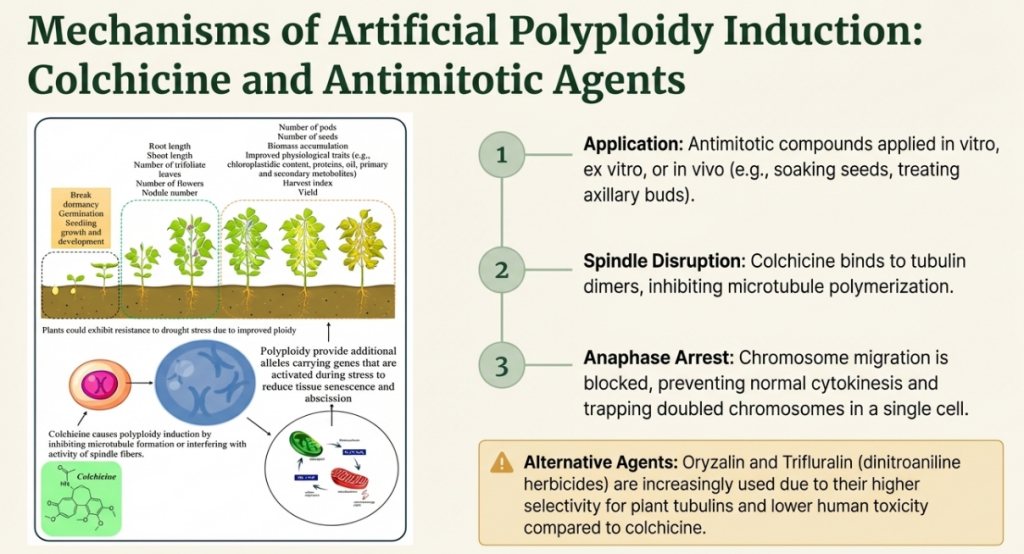
Colchicine: The Traditional Gold Standard
Derived from Colchicum autumnale, colchicine remains a cornerstone of cytogenetics. Its effectiveness is highly sensitive to concentration, exposure duration, and explant type. By binding to tubulin, it interferes with spindle fiber formation, preventing mitosis from reaching completion.
- Methods: In vitro (protocorms/calli), Ex vitro (shoot tips), and In vivo (epicotyl meristems).
Dinitroaniline Herbicides: The Oryzalin Advantage
Agents like Oryzalin and Trifluralin are increasingly preferred over colchicine. Their primary strategic advantage lies in their higher selectivity for plant tubulins and significantly lower human toxicity. In high-value species like Cannabis sativa, oryzalin has proven superior in inducing stable polyploidy with lower mortality.
Functional Genomics vs. Ploidy Induction
It is critical to distinguish WGD agents from alkylating agents like EMS (Ethyl Methanesulfonate) and MMS (Methyl Methanesulfonate). These do not induce WGD; rather, they cause point mutations used for Functional Genomics and Mutant Library Production to create minor trait variations.
5.1 Comparative Analysis of Antimitotic Agents: Colchicine vs. Dinitroanilines
Historically, colchicine has been the dominant agent for spindle fiber interference. However, a modern breeding strategy mandates a shift toward alternative agents like dinitroanilines (oryzalin, trifluralin) or amiprophos-methyl. These alternatives provide a higher affinity for plant-specific tubulins, allowing for lower effective concentrations and significantly higher plant survival rates.
Comparative Profile of Induction Agents
| Agent Name | Mechanism of Action | Relative Toxicity (Human/Plant) | Key Competitive Differentiator |
|---|---|---|---|
| Colchicine | Binds to tubulin; disrupts mitotic spindle formation. | High: Potent human toxin; often causes plant morphological defects. | The historical gold standard; remains effective across the broadest range of taxa. |
| Oryzalin / Trifluralin | Dinitroanilines; bind specifically to plant-specific tubulins. | Low: Higher operator safety; lower plant mortality rates. | High affinity for plant tubulins allows for efficacy in the µM range. |
| Amiprophos-methyl | Dinitroaniline herbicide; disrupts microtubule organization. | Low: Effective alternative to colchicine with reduced cytotoxicity. | Offers a viable substitute for dinitroaniline-resistant genotypes. |
| EMS / MMS | Alkylating agents; specifically alkylate guanine bases. | Moderate/High: Risk of meiotic aberrations and sterility. | Induces point mutations for genetic diversity, not whole-genome doubling. |
Strategists must strictly distinguish these from alkylating agents like Ethyl Methanesulfonate (EMS) and Methyl Methanesulfonate (MMS). These function via the alkylation of DNA bases—specifically guanine—to produce point mutations. They are tools for trait modification and increasing genetic variability, not for true ploidy manipulation.
5.2 Protocol Optimization: The Kinetics of Concentration and Exposure
The kinetic relationship between chemical concentration and exposure duration is the most critical variable in any induction program. Success is defined by identifying the specific threshold where chromosomal doubling is triggered while maintaining manageable mortality levels.
The Strategic Directive: Toxicity-Efficiency Trade-off
To optimize this window, the following directives must be mandated:
- Identify the LD50: Strategists must conduct range-finding studies to pinpoint the dose/time combination resulting in 50% mortality; induction is typically maximized at this threshold.
- Calibrate for Explant Sensitivity: Seeds, protocorms, and shoot tips possess distinct sensitivity profiles; protocols must be adjusted accordingly.
- Mandate Pre-culture Phases: For recalcitrant species like Populus hopeiensis, a 9-day pre-culture followed by a 4-day treatment at 100 µM colchicine is the mandated standard for maximizing octaploid induction.
System Selection Analysis
The application system must be matched to the species’ habit and scalability requirements:
- In Vitro: Provides maximum accuracy and control. Essential for species like Digitalis lanata to produce stable tetraploids.
- Ex Vitro: Optimized for scalability in field crops like Nigella sativa through seed soaking.
- In Vivo: The primary method for woody perennials. For Dendrobium crumenatum, an immersion protocol utilizing air bubbling is required to prevent oxygen depletion during the 0.05% colchicine treatment pulse.
Over-exposure must be rigorously avoided. Excessive durations lead to mixoploidy, stunted growth, and death, as cellular damage overwhelms the regenerative capacity of the apical meristem.
6. Case Studies in Medicinal and Commercial Crop Enhancement
The synthesis of the following data points serves as a proof-of-concept for the commercial viability of polyploid breeding:
- Cannabis sativa: Oryzalin-induced tetraploids show a 30% increase in floral terpenes and a 71.5% increase in leaf terpenes (notably α-bisabolol and α-humulene). While THCA remains stable, CBDA increases by 8.9%, shifting the cannabinoid profile toward pharmaceutical demand.
- Thymus vulgaris: Tetraploid lines exhibit a 2x increase in plant height and a surge in thymol content from 30.31% to 48.32%.
- Wild Blueberries (Vaccinium duclouxii) “Tetraploid blueberries showed notable anatomical improvements, including… increased vein thickness, upper epidermis, palisade tissue, and spongy mesophyll by 149.2%, 174.9%, and 162.4%, respectively. These results demonstrate the usefulness of trifluralin… in creating strong germplasm.”
- Spearmint (Mentha spicata) “The hexaploid spearmint plants grew faster, produced bushier shoots, and had thicker and larger leaves… with a stunning 48.9% rise [in] essential oil yields (carvone and limonene). Notably, Genotype P3 stood out among the others, reinforcing that WGD creates a reservoir for selection where specific genotypes can achieve elite industrial value.”
- Citrus limon: Polyploidy results in an 80% height increase and enhanced limonene production driven by the upregulation of LS, CHS, and PAL genes.
- Environmental Resilience:
- Paper Mulberry (Broussonetia papyrifera): Superior drought resistance due to tighter leaf morphologies and higher net photosynthetic rates.
- Caladium: Enhanced freezing tolerance via significantly higher antioxidant activity, specifically SOD and POD enzymes.
- Glycine tabacina: Tetraploids exhibit significantly higher resistance to leaf-rust fungus (Phakopsora pachyrhizi) compared to diploids.
- Drought Survival: Polyploid species like Arabidopsis thaliana and Caragana arborescens exhibit superior water-retaining capacity. Due to their specific stomatal architecture—specifically the lower pore space per unit area—they maintain lower transpiration rates. This allows them to sustain leaf water content in xeric (dry) conditions that would desicate a diploid ancestor.
- Cold and Frost Hardiness: Polyploidy offers two distinct advantages in cold climates. First is frost hardiness; species like Brassica campestris and Avena (oats) show increased resistance to freezing. Second is a shift in temperature optima; for instance, in Crimson Clover (Trifolium incarnatum), the temperature optimum for photosynthesis shifts from 15–20°C in diploids down to 10°C in tetraploids. This effectively “pre-adapts” the plant to thrive in cooler conditions rather than just surviving them.


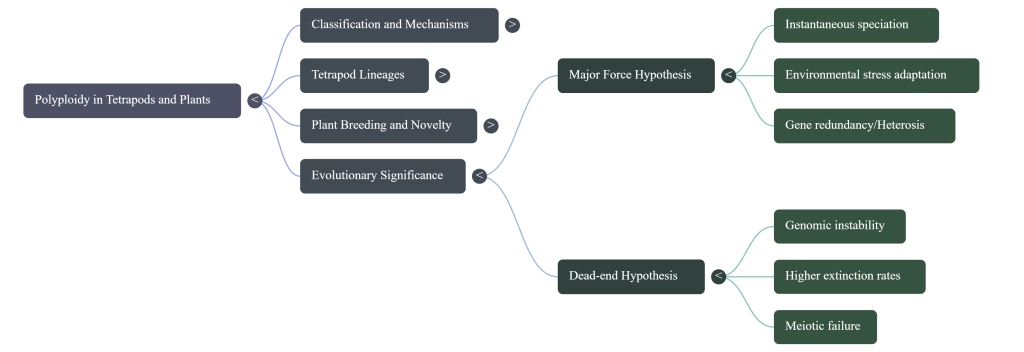
7. Conclusion: Polyploidy as an Evolutionary Advantage or Dead End?
The debate over whether polyploidy is a “dead end” or a “major evolutionary force” remains a central theme in modern cytogenetics. While doubling the genome can be maladaptive in stable environments—due to metabolic drag and initial fertility losses—its true value is revealed under stress. The genomic redundancy provided by WGD is the raw material for biochemical novelty and rapid adaptation. In the face of climate instability, targeted chemical induction using oryzalin or colchicine is not merely a laboratory curiosity; it is a high-yield strategy for engineering the agricultural and medicinal resilience required for the 21st century.
Induced macromutants offer a high-speed lane for agricultural speciation. By utilizing the genomic redundancy and nucleotypic physical changes of polyploidy, we can rapidly engineer crops designed for a volatile global climate.

Strategic Directives:
- Leverage Allopolyploid Pathways: Prioritize interspecific hybridization to exploit Homoeologous Exchange, creating biochemical profiles and “hybrid enzymes” that transcend parental limits.
- Deploy for Environmental Extremes: Target the deployment of polyploids specifically in saline or xeric environments (as demonstrated in the Lycopersicon studies of Tal 1977), where their superior ion uptake and water-use efficiency provide a decisive advantage.
- Targeted Metabolite Amplification: Use WGD to intentionally trigger gene dosage effects in medicinal species, maximizing the synthesis of therapeutic alkaloids and terpenes.
Polyploidy is not an evolutionary accident to be corrected, but a cornerstone of genomic resilience that will secure our food and medicinal systems against the exigencies of the future.
Image Summary

Questions/Answers
1. How does chromosome doubling create evolutionary novelty in flowering plants?
Chromosome doubling, or polyploidy, is a major force in plant evolution that creates novelty by providing an immediate increase in genetic material and triggering profound phenotypic, physiological, and genomic changes. This process can occur through autopolyploidy (duplication within a single species) or allopolyploidy (hybridization followed by doubling).
The primary mechanisms through which chromosome doubling fosters evolutionary novelty include:
1. Genetic Redundancy and Functional Innovation
Chromosome doubling provides redundant gene copies, which releases them from existing functional constraints. While one copy maintains the original function, the extra copies are free to accumulate mutations, leading to:
- Neofunctionalization: The acquisition of entirely new gene functions.
- Subfunctionalization: The partitioning of roles between duplicated genes to optimize plant performance.
- Genome Buffering: Extra gene copies mask the effects of deleterious recessive mutations, providing mutational robustness.
2. Genomic and Epigenetic Restructuring
Polyploidization often causes a “genomic shock” that triggers rapid alterations beyond simple doubling. These include chromosomal rearrangements (deletions, translocations) and epigenetic modifications like changes in DNA methylation patterns. These changes can rewire metabolic pathways and protein networks, leading to the emergence of novel phenotypes not present in the ancestors.
3. Phenotypic Novelty (The “Gigas” Effect)
A direct and immediate consequence of chromosome doubling is an increase in cell size, which frequently results in larger plant organs. This can include:
- Enlarged organs: Thicker and larger leaves, sturdier stems, and bigger flowers, fruits, and seeds.
- Physiological shifts: Slowed growth rates and altered flowering times, which can change relationships with pollinators or predators.
- Metabolic diversity: Increased production and novelty in secondary metabolites, such as alkaloids and terpenes, which can enhance defense against herbivores or pathogens.
4. Adaptive Advantages and Niche Expansion
Chromosome doubling can propel populations into new ecological niches by increasing tolerance to environmental stressors.
- Stress resilience: Polyploids often exhibit enhanced resistance to drought, salt, heat, and cold.
- Niche differentiation: Polyploids are frequently found in more extreme or disturbed environments compared to their diploid progenitors, allowing them to colonize new geographic areas.
5. Reproductive Transformations
Polyploidy can trigger immediate shifts in reproductive strategies that facilitate speciation and persistence:
- Instant Speciation: Increased ploidy levels often create immediate reproductive barriers with parental lineages, leading to sympatric speciation in a single generation.
- Asexual Reproduction (Apomixis and Clonality): Polyploidy is strongly associated with apomixis (asexual seed production) and increased clonality (vegetative reproduction). In some cases, whole-genome duplication directly triggers novel modes of vegetative growth, such as root sprouting in Pilosella rhodopea, which helps the new polyploid persist despite initial fertility issues.
- Self-Compatibility: Doubling often breaks down self-incompatibility mechanisms, allowing for self-fertilization, which can be advantageous when mates are scarce.
2. What is the “Gigas” effect in chromosome-doubled plants?
The “Gigas” effect is a widely observed phenomenon in plants following chromosome doubling (polyploidy) where an increase in the number of gene copies leads to a significant enhancement of cell size, which in turn results in larger plant organs. The term was coined following the discovery of the Oenothera gigas mutant, the first recognized autotetraploid.
The primary characteristics and implications of the Gigas effect include:
1. Morphological Manifestations
The enlargement is typically seen across various vegetative and reproductive structures:
- Leaves: Polyploid leaves are frequently thicker, broader, and larger in area. They often exhibit a darker green color due to higher chlorophyll content.
- Flowers: Chromosome doubling often produces larger flower diameters, thicker and more “waxy” petals, and more vibrant colors, which are highly valued in ornamental breeding.
- Seeds and Fruits: The effect commonly results in larger, heavier fruits and bulkier seeds. Larger seeds often contain greater nutrient reserves, which can lead to more robust early seedling growth.
- Stems and Roots: Stems and petioles generally become thicker and sturdier. In some crops, this manifests as increased biomass in underground organs, such as larger tubers in potatoes or thicker roots in fodder crops.
2. Cellular and Nucleotypic Basis
The Gigas effect is a direct consequence of increased DNA content (a nucleotypic effect), which physically alters the size and geometry of the cell.
- Cell Enlargement: The volume of tetraploid cells is often roughly double that of their diploid progenitors. This enlargement is easily observed in stomatal guard cells, which are significantly longer and wider in polyploids.
- Chloroplast Density: Polyploid guard cells frequently contain a significantly higher number of chloroplasts compared to diploids.
- Pollen Size: Unreduced or doubled pollen grains typically have a much larger diameter (often 30–40% larger) than reduced grains.
3. Physiological and Developmental Trade-offs
While individual cells and organs are larger, the Gigas effect does not always result in a larger overall plant. It is often accompanied by several physiological shifts:
- Slowed Growth Rate: Polyploids often have longer cell cycles and slowed mitotic divisions, leading to an overall slower growth rate compared to diploids.
- Delayed Phenology: Polyploid plants frequently flower later and maintain a longer flowering duration.
- Altered Stomatal Density: While individual stomata are larger, their density (number per unit area) typically decreases, which can increase leaf diffusive resistance and lower transpiration rates.
- Reduced Fertility: Especially in newly formed autopolyploids, the Gigas effect is often balanced by reduced fertility due to meiotic irregularities, although this is sometimes exploited to produce seedless fruits (e.g., watermelons and bananas).
4. Agricultural and Evolutionary Importance
The Gigas effect is a primary target for plant breeders seeking to increase crop yield and biomass. Beyond size, it can improve the quality of harvested products, such as producing longer and tougher fibers in cotton or higher concentrations of secondary metabolites in medicinal plants. From an evolutionary perspective, these immediate phenotypic changes can “propel” a new polyploid into a different ecological niche, potentially providing greater tolerance to environmental stressors like drought or cold.
3. What causes the “genomic shock” that leads to new phenotypes?
“Genomic shock” refers to the period of profound genomic instability and restructuring that occurs immediately following a genome merger (hybridization) or whole-genome duplication (WGD). This phenomenon triggers a cascade of genetic and epigenetic changes that rewire a plant’s internal systems, ultimately leading to the emergence of novel phenotypes.
The causes and mechanisms of genomic shock include:
1. Activation of Transposable Elements
A central feature of genomic shock is the activation of transposable elements (TEs). These “jumping genes,” which are typically silenced in stable genomes, become mobile during the stress of genome merger. Their movement contributes to rapid genome restructuring and can disrupt or alter the regulation of neighboring genes, creating significant phenotypic variation.
2. Epigenetic Reprogramming
Polyploidization triggers massive epigenetic modifications, such as changes in DNA methylation, histone modification, and small RNA expression.
- Transcriptional Rewiring: These modifications regulate gene expression to minimize “conflicts” between merged genomes.
- Hypomethylation: WGD often induces DNA hypomethylation, which can activate stress-sensitive genes, allowing polyploids to respond more rapidly to environmental challenges than their diploid ancestors.
3. Structural Rearrangements and Diploidization
The initial “shock” often results in immediate structural changes to the DNA, including:
- Chromosomal Alterations: Deletions, duplications, inversions, and translocations of chromosome segments.
- Diploidization: To restore stability, the polyploid genome undergoes a long-term process called diploidization, which involves the progressive loss or silencing of redundant duplicated genes to return to a functionally “pseudodiploid” state.
4. Non-Additive Gene Expression
Genomic shock leads to gene expression patterns in the new polyploid that are not simply the sum of its parents.
- Expression-Level Dominance (ELD): The total expression of a gene aligns with only one of the parental species.
- Transgressive Expression: The polyploid expresses certain genes at levels significantly higher or lower than either parent, leading to “extreme” or entirely new traits.
- Homeolog Expression Bias: The relative contribution of the parental genome copies becomes unequal due to the modulation of regulatory elements.
5. Functional Innovation (Neo- and Subfunctionalization)
The redundancy provided by doubling the genome releases extra gene copies from their original functional constraints.
- Neofunctionalization: One gene copy accumulates mutations that allow it to acquire an entirely new function.
- Subfunctionalization: Duplicated genes partition original roles between themselves to optimize performance, such as developing tissue-specific or temporally distinct functions.
Phenotypic Outcomes of the Shock
These genomic disturbances result in “macromutants” that possess unique tolerances and developmental patterns. Observed novelties include:
- Enhanced Stress Resilience: Improved tolerance to drought, salt, heavy metals, and extreme temperatures through the rewiring of metabolic pathways.
- The “Gigas” Effect: Increased cell size leading to larger, thicker organs (leaves, flowers, and fruits).
- Metabolic Diversity: Changes in the production of secondary metabolites, such as alkaloids and essential oils, which can enhance defense against pests.
4. Can you explain the differences between neofunctionalization and subfunctionalization?
Neofunctionalization and subfunctionalization are two distinct evolutionary processes that occur following gene duplication, often as a result of polyploidy (whole-genome duplication). Both processes are driven by the genetic redundancy provided by having multiple copies of the same gene, which releases those copies from existing functional constraints.
The primary differences between them are as follows:
Neofunctionalization
- Definition: This occurs when one of the duplicated gene copies accumulates mutations that allow it to acquire an entirely new function.
- Process: While one copy of the gene maintains the original, essential function, the redundant copy is free to diverge and develop a novel role that was not present in the progenitor.
- Result: This leads to the creation of genetic novelty and can contribute to the development of new adaptations or metabolic pathways.
Subfunctionalization
- Definition: This occurs when the duplicated gene copies partition the original roles of the ancestral gene between themselves.
- Process: Instead of one copy gaining a new function, both copies specialize to perform a subset of the original gene’s duties more efficiently.
- Common Manifestations: This often results in duplicated genes developing tissue-specific or temporally distinct functions. For example, one copy might only be expressed in the leaves while the other is expressed in the roots.
- Result: This process optimizes plant performance and helps maintain the stability of the redundant genetic material over long evolutionary periods.
Summary of Key Differences
| Feature | Neofunctionalization | Subfunctionalization |
|---|---|---|
| Outcome | Acquisition of a new function. | Partitioning of existing roles. |
| Ancestral Roles | Maintained by one copy; the other diverges. | Divided between the two copies. |
| Specialization | Results in functional innovation. | Results in functional optimization. |
Both processes are central to the functional diversification and metabolic diversity observed in flowering plants following chromosome doubling. Together, they provide an expanded “genetic toolkit” that can enhance a plant’s resilience and its ability to adapt to challenging environments.
5. What is the difference between autopolyploidy and allopolyploidy?
The primary difference between autopolyploidy and allopolyploidy lies in their mode of origin: autopolyploidy involves chromosome doubling within a single species, while allopolyploidy results from the hybridization of two or more distinct species followed by genome duplication.
The following table and details summarize the key differences across genetic, cytological, and evolutionary dimensions:
Key Differences Between Autopolyploidy and Allopolyploidy
| Feature | Autopolyploidy | Allopolyploidy |
|---|---|---|
| Origin | Derived from the same individual or species. | Derived from interspecific hybridization between different species. |
| Chromosomes | Possesses multiple sets of homologous (identical) chromosomes. | Combines sets of non-homologous or homeologous (similar but distinct) chromosomes. |
| Meiosis | Multiple homologous chromosomes may form multivalents (groups of 3, 4, or more) during pairing. | Chromosomes from different parents often lack enough homology to pair, favoring bivalents (pairs). |
| Inheritance | Typically exhibits polysomic inheritance (random segregation of alleles). | Typically exhibits disomic inheritance (similar to diploids, where alleles segregate independently). |
| Genetic Diversity | Lower initial diversity; relies on redundant copies of the same genome. | High diversity due to heterosis (hybrid vigor) from combining divergent genomes. |
Detailed Comparison
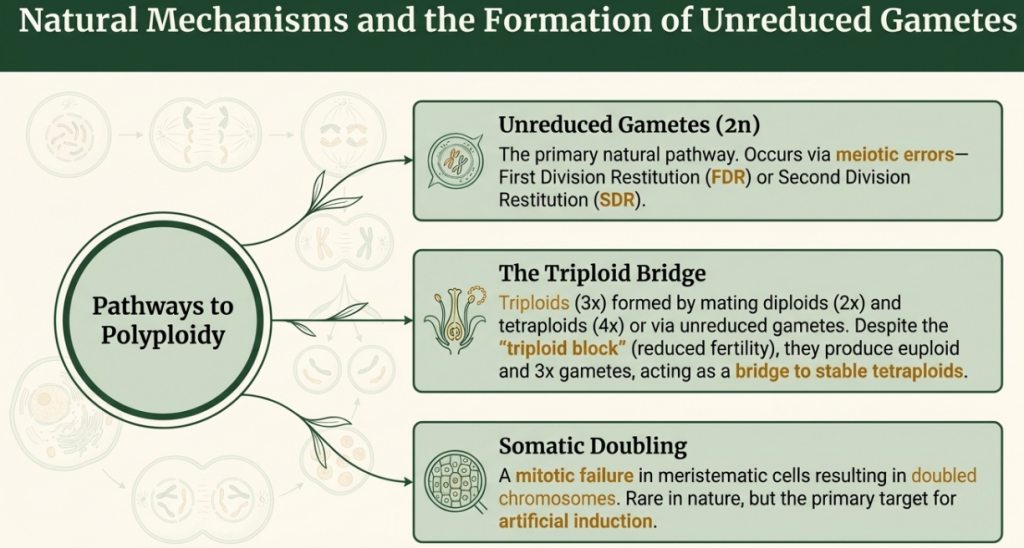
- Formation Mechanisms:
- Autopolyploidy generally occurs when an unreduced gamete (2n) joins a reduced one (n) or two unreduced gametes from the same species fuse. It can also arise through somatic doubling (mitotic errors) in meristematic tissues.
- Allopolyploidy requires distant hybridization. An infertile hybrid is formed first, and its fertility is subsequently restored when its genome doubles, allowing each chromosome to find a matching pairing partner for meiosis.
- Cytological Behavior:
- In autopolyploids, the presence of four or more identical chromosome sets leads to complex meiotic configurations, such as quadrivalents, which can cause meiotic irregularities and unbalanced gametes, often resulting in reduced fertility.
- In allopolyploids, if the parental genomes are sufficiently different (“true” allopolyploidy), they undergo regular bivalent pairing, behaving like a functional diploid (pseudodiploid) during reproduction. If the parental genomes are closely related, they are termed segmental allopolyploids and may show intermediate pairing behavior.
- Evolutionary and Phenotypic Impact:
- Allopolyploids typically show more pronounced changes in gene expression than autopolyploids due to the “genomic shock” of merging two disparate systems. This often results in transgressive traits—phenotypes that exceed the limits of either parent species.
- Autopolyploids are frequently characterized by the “Gigas” effect, where increased DNA content leads to larger cell sizes and, consequently, thicker leaves, larger flowers, and bigger fruits.
- Scientific Definitions:
- Researchers sometimes distinguish these types based on Mode of Origin (MO) or Cytological Criteria (CC). A polyploid might originate as an allopolyploid (MO) but, over time, undergo “diploidization” to behave cytologically like an autopolyploid or vice versa, blurring the taxonomic boundaries.
6. Which plant traits are most affected by the Gigas effect?
The “Gigas” effect is a direct and widespread consequence of chromosome doubling, characterized by an increase in cell size due to higher DNA content, which often leads to the enlargement of various plant organs. While the effect is prominent, it does not always result in a larger overall plant, as it is frequently accompanied by a decrease in the rate of cell division.
The plant traits most affected by the Gigas effect include:
1. Morphological Traits (Organs)
The most visible impacts occur in the vegetative and reproductive structures of the plant:
- Leaves: These commonly become thicker, broader, and coarser. They often exhibit a darker or deeper green color.
- Flowers: The effect frequently produces larger floral structures, including increased bud size and flower diameter.
- Seeds and Fruits: Polyploidy often results in larger, heavier, and bulkier seeds and fruits. For example, in cumin, tetraploid seeds and embryos are significantly larger due to the Gigas effect, which enhances storage reserves. However, this often comes with a trade-off where the plant produces fewer total seeds.
- Stems and Roots: Stems and petioles typically become thicker and more robust. Underground organs like tubers and roots also show increased size and biomass.
2. Cellular and Anatomy Traits
The Gigas effect is rooted in physical alterations at the cellular level:
- Cell and Nuclear Volume: The volume of tetraploid cells is typically about twice that of their diploid progenitors, though the surface-to-volume ratio decreases.
- Stomata: Stomatal guard cells are significantly affected, typically becoming longer and wider (expanded). This increase in size is often accompanied by a decrease in stomatal density (number of stomata per unit area).
- Pollen: Unreduced or doubled pollen grains typically exhibit a 30–40% larger diameter than reduced grains.
- Internal Anatomy: Leaf anatomical structures, such as the epidermis and mesophyll layers, often become significantly thicker.
3. Physiological and Developmental Traits
The structural changes of the Gigas effect trigger several developmental shifts:
- Slowed Growth Rate: Polyploids often exhibit slower initial growth and longer cell cycles compared to diploids.
- Delayed Phenology: Plants affected by the Gigas effect tend to flower later in the season and maintain a prolonged flowering duration.
- Metabolic Activity: The increase in cell size and gene dosage can lead to heightened metabolic activities and increased production of secondary metabolites.
- Water and Gas Exchange: Because stomatal pores are larger but less dense, leaf diffusive resistance may increase, leading to lower transpiration and respiration rates in some polyploids.
7. Can you explain the trade-offs between organ size and growth?
The trade-off between organ size and growth in chromosome-doubled (polyploid) plants is primarily defined by the “Gigas” effect, where an increase in DNA content leads to larger individual cells and organs, but often at the expense of the speed of development and overall growth rate.
The specific trade-offs identified in the sources include:
1. Enlarged Organs vs. Slowed Cell Division
While polyploidy typically results in larger vegetative and reproductive structures—such as thicker leaves, larger flowers, and heavier seeds—this does not always result in a larger total plant.
- Reduced Division Rates: The enlargement of cell size is often counterbalanced by a decrease in the rate of cell division.
- Longer Cell Cycles: Polyploid cells generally have longer cell cycles and increased duration of both mitosis and meiosis, which retards the overall pace of metabolism and growth.
- Dwarfism: In some species, such as newly generated apple tetraploids or Populus hopeiensis, the increase in cell size is associated with a dwarf phenotype, characterized by shorter internodes and reduced plant height compared to their diploid ancestors.
2. Reproductive Quantity vs. Size
A common trade-off exists between the size of reproductive elements and their total number or fertility.
- Fewer but Bigger Seeds: Polyploids frequently produce larger, more nutrient-rich seeds, but they often generate a smaller total number of seeds per fruit or inflorescence.
- Delayed Flowering: The slower developmental rate often leads to delayed phenology, where polyploid plants flower later in the season and maintain a longer blooming period than diploids.
- Reduced Fertility: The meiotic irregularities common in newly formed polyploids often result in reduced pollen viability and lower seed set, which can limit initial demographic success despite the increased vigor of individual organs.
3. Hormonal and Metabolic Constraints
Physiological shifts triggered by the increased nuclear DNA content (nucleotypic effects) also create developmental trade-offs.
- Hormonal Deficiencies: Polyploids often exhibit lower levels of essential growth hormones like auxin (IAA) and brassinosteroids, which are linked to their slower growth and compact habits.
- Energy and Resource Allocation: The increased metabolic cost of replicating and maintaining a larger genome can drain energy that would otherwise be used for immediate competitive growth or photosynthesis.
- Stomatal Density: While polyploid stomata are typically larger (improving gas exchange potential), their density is often significantly reduced, creating a delicate balance between photosynthetic efficiency and water loss through transpiration.
4. Adaptation vs. Competitive Speed
Polyploids may lag in growth rate or reproductive success under stable, favorable conditions but often show a trade-off advantage by exhibiting superior resilience to environmental stressors. For instance, they may exhibit thicker leaves and sturdier structures that help them survive drought, salt, or extreme temperatures that would kill faster-growing diploid progenitors.
8. Compare the effectiveness of colchicine versus oryzalin in plant breeding.
Both colchicine and oryzalin are powerful antimitotic agents used in plant breeding to induce polyploidy, but they differ significantly in their efficiency, safety, and genomic impact. While colchicine is the most established and universal treatment, oryzalin has surfaced as a prominent alternative due to its higher affinity for plant tissues and lower toxicity.
The following sections compare the two agents across several key dimensions:
1. Mechanism of Action and Affinity
- Target Proteins: Both chemicals induce polyploidy by interfering with microtubule polymerization during mitosis, preventing chromosome migration during anaphase. However, they bind to different subunits: colchicine binds to β-tubulin (or α– and β-tubulin dimers), while oryzalin binds to α-tubulin.
- Binding Affinity: Colchicine has a relatively low affinity for plant tubulins, meaning it requires significantly higher concentrations to be effective. In contrast, oryzalin has a high specificity and affinity for plant tubulin, making it effective at much lower doses.
2. Efficiency and Dosage
- Effective Concentrations: Because of its low affinity, colchicine is typically applied at millimolar (mM) concentrations. Oryzalin achieves similar or superior results at micromolar (μM) concentrations.
- Induction Rates: In many studies, oryzalin has demonstrated higher induction efficiency. For example, in Caladium, oryzalin achieved a 46.67% induction rate compared to only 15% for colchicine. In anise hyssop, oryzalin resulted in 20% tetraploids, while colchicine produced 16%. However, some studies, such as those on blackberries, found no significant difference in the ultimate effects of the two agents.
3. Genomic Stability and Side Effects
- Abnormalities: Colchicine treatment is more frequently associated with genomic instability, including chromosome losses, rearrangements, and aneuploidy. Oryzalin is more selective for plant tubulin, typically resulting in more stable polyploidy with fewer abnormalities.
- Chimeras: The use of colchicine often leads to the formation of mixoploids or chimeras (plants with cells of different ploidy levels), which can be difficult to stabilize or use in further breeding.
- Cytotoxicity: Colchicine is highly cytotoxic and can significantly reduce plant survival and regeneration rates, especially at the higher concentrations required for induction. Oryzalin is generally less toxic to plants and has fewer adverse effects on growth.
4. Safety and Practicality
- Human Health: Colchicine is a strong alkaloid that is highly toxic to humans and must be handled with extreme care. Oryzalin, a dinitroaniline herbicide, is much safer for breeders to handle.
- Cost and Accessibility: Colchicine remains widely used because it is water-soluble, thermostable, and its protocols are well-established for a vast range of species. Oryzalin is sometimes preferred for in vitro systems where low toxicity is critical for regenerating fragile explants.
Comparative Summary Table
| Feature | Colchicine | Oryzalin |
|---|---|---|
| Typical Dose | Millimolar (mM) | Micromolar (μM) |
| Affinity | Low for plant tubulin | High for plant tubulin |
| Tubulin Target | β-tubulin | α-tubulin |
| Plant Toxicity | High (cytotoxic) | Low |
| Human Toxicity | High | Low |
| Genomic Stability | Prone to mutations/rearrangements | More stable induction |
| Common Result | Mixoploids/Chimeras | More solid polyploids |
9. What are the common application methods for colchicine?
Common application methods for colchicine are categorized into in vivo, ex vitro, and in vitro systems, with the choice of technique depending on the targeted plant tissue and the desired efficiency.
1. In Vivo and Ex Vitro Methods
These methods involve applying colchicine to seeds or established plants outside of sterile laboratory environments:
- Seed Soaking: One of the most common and simplest techniques, where seeds are immersed in a colchicine solution for durations ranging from a few hours to several days.
- Dropping Solution on Buds: A colchicine solution is applied as a single drop directly to the axillary buds or the apical meristem of shoot tips to target actively dividing cells. This process is often repeated at regular intervals (e.g., twice daily for several days).
- Spraying: The solution can be sprayed directly onto the leaves and buds of established plants.
- Submerging Seedlings or Plantlets: Young seedlings or plantlets can be completely submerged in a colchicine solution for 24 to 96 hours to ensure the chemical reaches all meristematic regions.
- Injection and Cotton Leaching: In some species, colchicine is injected into tissues (such as flower buds) or applied using cotton soaked in the solution and placed against the plant.
- Application with Carriers: Chemical solutions can be applied to buds using carriers like agar, lanolin paste, or cotton to increase contact time and chemical absorption.
2. In Vitro Methods
Laboratory-based in vitro techniques are generally preferred because they offer greater control, higher consistency, and easier penetration of the chemical into actively dividing cells.
- Immersion of Explants: Isolated tissues such as stem segments, nodes, or leaf explants are immersed in a sterile colchicine solution before being transferred to a growth medium.
- Inclusion in Culture Medium: Colchicine is added directly to a solid or liquid culture medium where the explants are grown, ensuring continuous exposure during the early stages of regeneration.
- Callus Treatment: Undifferentiated callus tissue is exposed to colchicine, which then regenerates into polyploid or heteroploid shoots.
- Targeting Specialized Structures: The chemical can be applied to protocorms, somatic embryos, or microspores in sterile conditions to produce stable polyploid lines.
Factors Affecting Success
The effectiveness of any application method is highly dependent on optimizing the concentration of the colchicine solution and the duration of exposure. While higher doses or longer treatment periods often increase the rate of chromosome doubling, they also frequently lead to reduced plant survival and the formation of chimeras (mixoploids) due to the drug’s cytotoxic effects.
10. How do oryzalin and trifluralin compare to colchicine?
Oryzalin and trifluralin are dinitroaniline herbicides used as safer and often more effective alternatives to colchicine, a traditional antimitotic agent used for chromosome doubling in plants. While both types of chemicals disrupt cell division to induce polyploidy, they differ significantly in their binding targets, toxicity, and required concentrations.
1. Mechanism of Action and Binding Affinity
- Binding Targets: Colchicine binds to β-tubulin, inhibiting microtubule polymerization and preventing chromosome movement during anaphase. In contrast, dinitroanilines like oryzalin and trifluralin bind specifically to α-tubulin.
- Affinity: Colchicine has a low affinity for plant microtubules, which means it must be used at higher concentrations to be effective. Oryzalin and trifluralin have a high specificity and affinity for plant tubulins, allowing them to work effectively at much lower doses.
2. Safety and Toxicity
- Human Health: A major disadvantage of colchicine is its high toxicity to humans. Oryzalin and trifluralin are considered much safer alternatives for researchers and breeders.
- Phytotoxicity: Colchicine can cause genomic instability, leading to chromosomal losses, rearrangements, and gene mutations. Oryzalin is often reported to produce more stable polyploids with fewer abnormalities. However, colchicine remains popular because its protocols are well-established for a wide variety of species.
3. Efficacy and Required Concentrations
- Dosage: Because of their high affinity for plant proteins, oryzalin and trifluralin are effective at micromolar concentrations. Colchicine typically requires millimolar concentrations to achieve the same result.
- Plant-Specific Performance: The relative effectiveness of these agents often depends on the species being treated:
- Greater Efficacy: In Cucumis sativus (cucumber) and Caladium, oryzalin was found to be the safest and most effective agent, with lower toxicity and higher induction rates than colchicine.
- Lower Efficacy: Conversely, in studies of hemp, colchicine was found to be more effective at producing solid polyploids, whereas oryzalin (Surflan) produced only mixoploids.
Comparison Summary Table
| Feature | Colchicine | Oryzalin / Trifluralin |
|---|---|---|
| Chemical Class | Alkaloid (from autumn crocus) | Dinitroaniline herbicide |
| Binding Site | β-tubulin | α-tubulin |
| Typical Dose | Millimolar (High) | Micromolar (Low) |
| Toxicity | High (human/plant) | Low (safer alternative) |
| Genetic Impact | Can cause mutations/rearrangements | More stable induction |
11. How do oryzalin and trifluralin reduce human toxicity risks?
Oryzalin and trifluralin reduce human toxicity risks primarily because they are effective at significantly lower concentrations and possess a different chemical profile than the highly toxic alkaloid colchicine.
The specific ways these agents reduce risks include:
1. Significantly Lower Required Concentrations
Because dinitroaniline herbicides like oryzalin and trifluralin have a high specificity and affinity for plant tubulins, they can induce chromosome doubling at micromolar (μM) concentrations. In contrast, colchicine has a relatively low affinity for plant microtubules and must be applied at millimolar (mM) concentrations—which are often 1,000 times higher—to achieve similar results. This drastic reduction in the amount of chemical required naturally lowers the potential for hazardous exposure.
2. Reduced Cytotoxicity and Chemical Hazard
Colchicine is a strong alkaloid known to be highly toxic to humans and is often associated with severe side effects and genomic instability in plants. Oryzalin and trifluralin are considered safer alternatives for breeders to handle. Specifically:
- Selective Binding: These herbicides bind to α-tubulin, whereas colchicine binds to β-tubulin. Their high specificity for plant-specific proteins makes them less generally cytotoxic to humans than the more “universal” disruptive action of colchicine.
- Stability and Residue: While colchicine is water-soluble and thermostable, making it persistent, oryzalin is noted for having fewer adverse effects and being a “less toxic substance” overall.
3. Enhanced Laboratory Safety
Researchers often prefer oryzalin and trifluralin for in vitro systems because their lower toxicity profiles make them easier to manage in controlled environments without the extreme health risks posed by handling large quantities of colchicine solution.
In summary, by allowing breeders to use micromolar doses of a substance that is chemically less hazardous than a potent alkaloid, oryzalin and trifluralin provide a safer pathway for inducing polyploidy in crops.
12. How does polyploidy affect secondary metabolite production in medicinal plants?
Polyploidy significantly impacts the medicinal and economic value of plants by increasing the concentration of desired compounds and altering their overall phytochemical profiles. This increase in metabolic output is a primary goal in the artificial induction of polyploidy for the pharmaceutical industry.
Quantitative Increases in Metabolites
Chromosome doubling frequently leads to a substantial increase in the yield of secondary metabolites:
- Essential Oils and Terpenes: Tetraploid chamomile varieties like ‘Lutea’ and ‘Goral’ produce over 20% more chamazulene than their diploid counterparts. Similarly, oryzalin-induced thyme tetraploids exhibit significantly higher amounts of essential oil, thymol, and carvacrol.
- Specific Bioactive Compounds: In ajowan, tetraploidy can produce 2.5 times more thymol than diploid controls. Echinacea purpurea tetraploids show a 45% increase in chicoric acid and a 71% increase in chlorogenic acid.
- Pharmaceutical Glycosides: In Digitalis lanata, tetraploids produce significantly higher amounts of digoxin (1.61x) and digitoxin (1.73x) compared to diploids.
- Alkaloids and Scent: Polyploidy in drug plants like Datura, Atropa, and Cannabis often results in sharply increased quantities of alkaloids. In Hedychium, triploids have been found to release higher levels of floral scent compared to tetraploids.
Qualitative Changes and Novel Compounds
Beyond simple concentration increases, polyploidy can trigger the production of entirely new chemical components or remove existing ones through gene silencing or genomic restructuring:
- New Phytochemical Profiles: Colchicine-induced tetraploids of Citrus limon possess unique profiles characterized by the appearance of β-bisabolene and the deletion of some original components.
- Novel Components: Tetraploid Tetradenia riparia produces α-humulene and viridiflorol, substances that are completely absent in its diploid form.
- Diverse Flavonoids: In Elsholtzia splendens, tetraploids produce methoxylated flavonoids—such as wogonin and oroxylin A—which are not found in the diploid progenitors and possess unique anti-cancer properties.
Molecular and Physiological Mechanisms
The impact of polyploidy on secondary metabolism is driven by several internal factors:
- Gene Dosage and Enzyme Activity: Doubling the genome provides an immediate increase in gene dosage, allowing for higher levels of gene expression. Polyploids often exhibit superior enzymatic capacity and higher enzyme activity per milligram of protein compared to diploids.
- Functional Diversification: Genetic redundancy allows duplicated genes to undergo neofunctionalization (acquiring new functions) or subfunctionalization (partitioning existing roles), which lie at the heart of plant metabolic diversity.
- Nucleotypic Effects: The increase in DNA content leads to larger cell and nuclear volumes, which can alter biochemistry, anatomy, and metabolic regulation.
Interaction with Environmental Stress
Polyploidy often increases a plant’s tolerance to environmental stressors, which can, in turn, modulate metabolite production:
- Stress-Induced Synthesis: Under drought conditions, the triploid variety of Passiflora edulis exhibits heightened production of antioxidant enzymes.
- Stress Resilience Examples: In Lippia alba, triploid plants exhibited a 10-fold increase in linalool when subjected to water stress, highlighting a resilient metabolic response not seen in diploids or tetraploids.
13. What are the common trade-offs between organ size and growth?
The common trade-offs between organ size and growth in chromosome-doubled (polyploid) plants are primarily defined by the “Gigas” effect, where increased DNA content leads to larger individual cells and organs, but often at the expense of developmental speed, fertility, and organ quantity.
The specific trade-offs identified in the sources include:
1. Enlarged Organs vs. Slowed Cell Division
While polyploidy typically results in larger vegetative and reproductive structures—such as thicker leaves, larger flowers, and heavier seeds—this does not always translate to a larger total plant size.
- Reduced Division Rates: The enlargement of cell size is frequently counterbalanced by a decrease in the rate of cell division, which confines the plant’s overall growth.
- Longer Cell Cycles: Polyploid cells generally have longer cell cycles and increased duration for mitosis and meiosis, which retards the pace of metabolism and development.
- Dwarf Phenotypes: In many species, such as newly generated apple tetraploids or Populus hopeiensis, the increase in cell size and organ thickness is accompanied by dwarfism, characterized by shorter internodes, shorter shoots, and reduced height compared to diploid ancestors.
2. Organ Size vs. Quantity (Fecundity)
A consistent trade-off exists between the size of reproductive elements and their total number.
- Fewer but Larger Organs: Polyploids commonly produce enlarged reproductive structures (flowers, fruits, and seeds) that are less numerous than those of their diploid progenitors. For instance, tetraploid cumin and apples produce significantly larger seeds and fruits but a lower total number of seeds per plant or fruit.
- Floral Trade-offs: In eggplant, increased ploidy leads to longer and broader leaves but results in fewer total flowers and reduced general vigor.
3. Growth Speed vs. Environmental Resilience
Polyploids often lag in growth rate or reproductive success under stable, favorable conditions but exhibit a trade-off advantage in stress resilience.
- Adaptive Slower Growth: Inherently lower growth rates are adaptive during periods of stress, as these plants have more modest resource requirements and are less likely to exhaust their immediate environment.
- Superior Stress Tolerance: The physiological and anatomical changes associated with the Gigas effect—such as thicker leaf epidermis and larger roots—foster enhanced tolerance to drought, salt, and extreme temperatures.
4. Metabolic and Hormonal Costs
The trade-off between growth and size is also driven by internal resource allocation and chemical signaling.
- Hormonal Reductions: Dwarfism and slowed growth in polyploids are linked to decreased levels of endogenous growth regulators, specifically auxin (indoleacetic acid) and brassinosteroids.
- Genome Maintenance Costs: The metabolic cost of replicating and maintaining an enlarged genome can drain energy that would otherwise be used for immediate competitive growth.
- Secondary Metabolite Production: Polyploids often shift resources toward increased production of secondary metabolites (bioactive compounds), which can further reduce growth and biomass accumulation.
5. Delayed Phenology
The slower developmental rhythms of polyploids result in later flowering times and prolonged blooming durations. While this can be a commercial advantage for ornamental plants, it represents a trade-off in developmental speed compared to diploids.
14. How does genomic shock create new plant phenotypes?
Genomic shock refers to a period of rapid genomic instability and restructuring that occurs immediately following whole-genome duplication (WGD) or the merger of divergent genomes through hybridization. This “shock” triggers a cascade of genetic and epigenetic alterations that rewire the plant’s internal systems, ultimately leading to the emergence of novel phenotypes not present in the ancestors.
The mechanisms through which genomic shock creates new plant phenotypes include:
1. Activation of Transposable Elements
A primary feature of genomic shock is the activation of transposable elements (TEs). These “jumping genes,” which are typically silenced in stable genomes, can become mobile during the stress of a genome merger. Their movement contributes to significant genome restructuring and can disrupt or alter the regulation of neighboring genes, creating new phenotypic variation.
2. Epigenetic Reprogramming
Polyploidization triggers massive epigenetic modifications, such as changes in DNA methylation, histone modification, and small RNA expression.
- Transcriptional Rewiring: These modifications modulate gene expression to minimize “conflicts” between merged parental genomes.
- Hypomethylation: WGD often induces DNA hypomethylation, which can activate stress-sensitive genes. This allows polyploids to respond more rapidly to environmental challenges—such as salt or drought stress—than their diploid ancestors.
3. Structural and Numerical Chromosomal Changes
Genomic shock often results in immediate structural changes to the DNA and the formation of unbalanced chromosome sets:
- Rearrangements: Deletions, duplications, inversions, and translocations of chromosome segments can lead to structural genome changes that impact gene regulation and speciation.
- Aneuploidy and Pseudoeuploidy: The cytological instability of new polyploids often produces progeny with irregular chromosome numbers (aneuploids). These “chromosomal macromutants” can exhibit strikingly divergent phenotypes and increased population-level variability.
4. Non-Additive Gene Expression
The “shock” leads to gene expression patterns that are not simply the sum of the parent species:
- Gene Silencing: One set of duplicated genes may be silenced due to a lack of selective pressure or dosage compensation, resulting in novel functional outcomes.
- Transgressive Expression: Polyploids may express certain genes at levels significantly higher or lower than either parent, leading to “extreme” or entirely new traits.
- Subgenome Dominance: In allopolyploids, one parental genome often becomes more transcriptionally active than the other, influencing trait inheritance and phenotypic stability.
5. Functional Innovation (Neo- and Subfunctionalization)
The redundancy provided by doubling the genome releases extra gene copies from their original functional constraints.
- Neofunctionalization: One duplicated gene copy is free to accumulate mutations and acquire an entirely new function.
- Subfunctionalization: Duplicated genes partition their original roles to optimize performance, such as developing tissue-specific or temporally distinct functions.
Phenotypic Outcomes of the Shock
These disturbances result in “macromutants” with unique tolerances and developmental patterns. Notable novelties include:
- Enhanced Stress Resilience: Improved tolerance to drought, salt, and extreme temperatures through the rewiring of metabolic pathways and antioxidant defense systems.
- The “Gigas” Effect: Increased cell size leading to larger, thicker organs, such as leaves, flowers, and fruits.
- Metabolic Diversity: Changes in the production of secondary metabolites (e.g., alkaloids, terpenes), which can enhance defense against pests or increase the medicinal value of the plant.
15. How does polyploidy impact plant resistance to pathogens?
Polyploidy impacts plant resistance to pathogens by providing an expanded genetic toolkit, altering secondary metabolism, and triggering structural changes, although the effects vary significantly depending on the type of pathogen and the plant species. While chromosome doubling often leads to enhanced biotic stress tolerance, it can also introduce meiotic instability or metabolic trade-offs that sometimes make polyploids more susceptible than their diploid progenitors.
1. Genetic Mechanisms of Resistance
The primary genetic advantage of polyploidy is gene redundancy, which facilitates enhanced defense in several ways:
- Gene Redundancy: Multiple copies of stress-related and resistance (R) genes provide a “backup system” to counteract pathogen damage.
- Integration of Ancestral Genomes: In allopolyploids, such as bread wheat, genomes from different ancestral species are merged, creating a rich reservoir of resistance genes against diseases like rust and powdery mildew.
- Constitutive Defense: Autotetraploid Arabidopsis has been shown to exhibit a higher baseline activation of defense genes, meaning its defense mechanisms are active even in the absence of a pathogen, whereas diploids primarily induce these genes only after exposure.
- Upregulated Signaling: In apple tetraploids, the expression of several disease-resistance genes (such as PR1 and WRKY29) is significantly enhanced following pathogen inoculation compared to diploids.
2. Secondary Metabolites and Anatomy
Polyploidy often induces physiological and structural changes that deter pathogens:
- Chemical Defense: Polyploids frequently produce higher concentrations of secondary metabolites, such as alkaloids and terpenes, which act as natural defenses against pathogens and herbivores.
- Structural Barriers: The “Gigas” effect often results in thicker leaves and epidermal layers, which can provide a more robust physical barrier against infection. For example, tetraploid Anthurium and certain apple clones show superior resistance to biotic stress partly due to these sturdier anatomical features.
3. Variations in Pathogen Response
The impact of polyploidy is not uniform across all types of pathogens:
- Hemibiotrophic Pathogens (e.g., Bacteria): Meta-analyses indicate that polyploids generally exhibit significantly better resistance than diploids against these pathogens.
- Biotrophic Pathogens (e.g., Fungi and Nematodes): Resistance in polyploids is less consistent for biotrophic pathogens. In some cases, polyploidy can actually decrease resistance because genome duplication may disrupt necessary signaling pathways or lead to the loss of duplicated R-genes during the process of diploidization.
- Autopolyploidy vs. Allopolyploidy: Some studies suggest that autopolyploids tend to show greater resistance than allopolyploids, though allopolyploidy is often used to transfer specific resistance traits between sexually incompatible species.
4. Documented Examples of Enhanced Resistance
- Apples: Autotetraploid clones have shown superior resistance to fire blight, apple scab, and leaf blight.
- Clover: Tetraploid red clover varieties often surpass diploids in overall disease resistance.
- Brassica: Triploid hybrids between B. napus and B. nigra exhibit inherited resistance to blackleg disease.
- Cotton: Allotetraploid cotton varieties utilize combined genomes to enhance tolerance to insect pests and fungal diseases like Fusarium wilt.
5. Trade-offs and Limitations
Despite these advantages, enhanced immunity often comes with fitness costs, such as reduced growth and yield, as resources are diverted from development to defense. Additionally, polyploidy can sometimes lead to heightened susceptibility; for example, some cultivated tetraploid potatoes are less resistant to fungal pathogens than their wild diploid relatives because the diploids exclusively possess certain pathogenesis-related proteins.
16. Can you explain the trade-offs between organ size and growth?
The trade-offs between organ size and growth in chromosome-doubled (polyploid) plants are primarily defined by the “Gigas” effect. While polyploidy consistently leads to larger individual cells and organs, it frequently results in a reduction in the overall rate of plant growth and development.
The key trade-offs identified in the sources include:
1. Enlarged Organs vs. Slowed Cell Division
Although polyploidy increases the volume of individual cells (often doubling it), this does not necessarily result in a larger total plant.
- Reduced Division Rates: The increase in cell size is often counterbalanced by a decrease in the rate of cell division, which can confine the plant’s total growth.
- Retarded Metabolism: Metabolism and growth are often retarded because larger polyploid cells have longer cell cycles and increased durations for mitosis and meiosis. This is partly due to altered geometric relationships between the nucleus and the cytoplasm, which can limit the rate of RNA transfer.
- Dwarf Phenotypes: In many species, such as apple, sweet cherry, and Populus hopeiensis, the increase in organ size is accompanied by dwarfism, characterized by shorter internodes and reduced plant height.
2. Organ Size vs. Reproductive Quantity (Fecundity)
A common trade-off exists between the size of reproductive structures and the total number produced.
- Fewer but Bigger Seeds: Polyploids often generate larger, heavier seeds with greater nutrient reserves, but they typically produce fewer total seeds per plant or fruit compared to diploids.
- Floral Trade-offs: Ornamental polyploids frequently produce larger, more vibrant flowers, but these are often fewer in number.
- Delayed Phenology: The slower growth rate often leads to delayed flowering and a prolonged flowering duration.
3. Growth Speed vs. Environmental Resilience
Polyploids may lag in growth rate or reproductive success under stable conditions but often exhibit a trade-off advantage in stress tolerance.
- Adaptive Slower Growth: Inherently low growth rates can be adaptive during stress, as slower-growing plants have more modest resource requirements and are less likely to exhaust their immediate environment.
- Structural Defenses: The anatomical changes of the Gigas effect—such as thicker leaf epidermis and mesophyll layers—provide enhanced tolerance to drought, salt, and extreme temperatures.
4. Metabolic and Hormonal Costs
- Hormonal Deficiencies: Dwarfism and slowed growth in polyploids are linked to reduced levels of endogenous growth regulators, specifically auxins and brassinosteroids.
- Genome Maintenance: The metabolic cost of replicating and maintaining a larger genome can drain energy that would otherwise be used for immediate competitive growth.
- Secondary Metabolites: Many polyploids shift resources toward increased production of secondary metabolites (e.g., alkaloids, terpenes), which can further reduce immediate vegetative development.
5. Photosynthetic Efficiency vs. Water Loss
Polyploidy creates a delicate balance regarding gas exchange. While individual stomata are larger in polyploids (potentially increasing photosynthetic rates), their density is often significantly reduced. This increased leaf diffusive resistance can lead to lower transpiration rates, which is beneficial for drought tolerance but may limit maximum growth capacity.
17. How do transposable elements contribute to genomic shock?
Transposable elements (TEs), often called “jumping genes,” are central players in the genomic shock that follows whole-genome duplication (WGD) or the merger of divergent genomes. Their contribution is primarily driven by epigenetic changes that release them from their typical state of suppression.
According to the sources, transposable elements contribute to genomic shock through the following mechanisms:
1. Activation via Epigenetic Reprogramming
In stable diploid genomes, TEs are typically silenced to prevent them from disrupting genetic stability. However, polyploidization triggers massive epigenetic modifications, such as changes in DNA methylation and histone modification. These changes can lead to the activation of transposable elements, which then become mobile within the newly merged or doubled genome.
2. Structural Genome Restructuring
Once activated, the movement of TEs contributes directly to genome restructuring. This includes:
- Chromosomal Rearrangements: TE activity can facilitate deletions, duplications, and translocations of DNA segments.
- Genome Downsizing: Over time, TEs can play a role in the loss or silencing of redundant genes (diploidization), helping to restore a more stable “pseudodiploid” state.
3. Regulation of Gene Expression
TEs frequently reside near or within gene regions. When genomic shock alters the methylation patterns of these elements, it has a “ripple effect” on neighboring genes:
- Transcriptional Rewiring: Changes in TE methylation can affect the transcription of adjacent stress-responsive genes, thereby altering the plant’s susceptibility or resistance to environmental challenges.
- Subgenome Dominance: In allopolyploids, variation in TE methylation patterns across subgenomes can lead to neighboring homeologs being expressed differently. Over long periods, this can even contribute to biased gene loss, where one parental genome is preferentially retained over the other.
4. Adaptive Feedback Mechanisms
The relationship between TEs and genomic shock is not purely destructive; it can be adaptive. Polyploidization often induces DNA hypomethylation, which can activate stress-sensitive genes (such as those for jasmonic acid biosynthesis) to form rapid responses to environmental threats. Interestingly, a feedback loop exists where the high expression of these stress genes can eventually trigger hypermethylation that suppresses the adjacent TEs again, potentially providing an evolutionary advantage by enhancing adaptation.
5. Exacerbation by Environmental Stress
The genomic instability caused by TEs is often intensified by external factors. Environmental stress (such as extreme temperatures or salinity) can further trigger TE activation and subsequent chromosomal rearrangements, which may either lead to detrimental mutations or provide the “macromutations” necessary for a polyploid to survive in a new ecological niche.
18. How do plants overcome the slowed growth caused by polyploidy?
Plants overcome or compensate for the slowed growth rates and reduced cell division cycles typically associated with polyploidy through several biological, evolutionary, and horticultural strategies. While chromosome doubling often leads to “dwarf” phenotypes with shorter internodes and slower development due to lower levels of growth hormones like auxin and brassinosteroids, several mechanisms allow these plants to persist and eventually thrive.
1. Horticultural Mitigation: Grafting
In agricultural contexts, breeders overcome the weak growth of newly generated polyploids by grafting them onto vigorous rootstocks. For example, own-rooted apple tetraploids often show poor growth, but when grafted onto M9 rootstocks, their growth parameters—such as shoot length and trunk cross-section area—improve significantly. This technique provides the polyploid “scion” with the robust resource-acquisition capabilities of a more established root system.
2. Shift to Vegetative Reproduction (Clonality)
Polyploidy frequently triggers a shift toward increased clonality or asexual reproduction, which compensates for initial fertility issues and slow sexual recruitment. In some species, such as Pilosella rhodopea, whole-genome duplication directly induces novel vegetative growth modes like root sprouting, a trait completely absent in its diploid ancestors. This enhanced vegetative vigor allows the new polyploid to establish and persist in its environment despite slow development.
3. Hybrid Vigor (Heterosis)
While autopolyploids (doubling within a single species) often suffer from slowed growth, allopolyploids (merging two different species’ genomes) can overcome these limitations through heterosis, or hybrid vigor. The combination of divergent parental genomes provides biochemical versatility and buffering, often resulting in allopolyploids that are more vigorous, have higher biomass, and exhibit faster growth rates than either parent.
4. Maturity-Related Vigor Recovery
The slowed growth of some polyploids appears to be a transient phase that lasts only until the plant reaches maturity. For instance, in certain apple cultivars like ‘Hanfu’, tetraploids initially grow more slowly than diploids but eventually show increased vigor and larger organs once they reach the reproductive phase, while the growth of their diploid counterparts begins to decrease.
5. Adaptive Slower Growth in Stressful Environments
In natural ecosystems, a slower growth rate can be a selective advantage rather than a hindrance to be overcome. Slow-growing polyploids have modest resource requirements, making them more tolerant of nutrient-deficient soils and less likely to exhaust their immediate environment. This shift toward a “K-selection” strategy (focusing on survival and defense rather than rapid expansion) helps polyploids succeed in harsh, stable, or disturbed habitats where fast-growing diploids might fail.
6. Long-term Genomic Evolution and DNA Loss
Over long evolutionary periods, polyploids undergo diploidization and genomic restructuring to regain stability and growth efficiency. Research has shown that established natural polyploids often have less DNA content and grow considerably faster than newly induced synthetic polyploids of the same species. This suggests that selection eventually favors genotypes that have “trimmed” redundant DNA or optimized gene expression to restore faster developmental rhythms.
19. How does polyploidy improve stress tolerance in plants?
Polyploidy improves stress tolerance in plants through a combination of morphological changes, biochemical enhancements, and genomic flexibility. By increasing the number of chromosome sets, plants acquire an expanded genetic toolkit and structural modifications that enable them to withstand various abiotic and biotic challenges.
1. Morphological and Structural Adaptations
A primary consequence of polyploidy is the “Gigas” effect, where an increase in DNA content leads to larger cells and, consequently, thicker and sturdier plant organs. These structural changes provide immediate physical advantages:
- Thicker Leaves and Epidermis: Polyploids often possess thicker leaf epidermis and mesophyll layers, which act as a more robust physical barrier against water loss and pathogen penetration.
- Stomatal Regulation: While polyploids typically have larger stomata, their stomatal density is often significantly reduced. This modification can improve water use efficiency by lowering transpiration rates while maintaining adequate gas exchange for photosynthesis.
- Robust Root Systems: Doubled genomes can result in enlarged root systems with thicker epidermis and more lateral roots, which enhance a plant’s ability to absorb water and nutrients from the soil, particularly under drought or salt stress.
2. Biochemical and Metabolic Resilience
Polyploidy triggers physiological and biochemical rewiring that protects cells from stress-induced damage:
- Enhanced Antioxidant Defense: Polyploids frequently exhibit higher levels of antioxidant enzymes (such as SOD, CAT, POD, and APX) and non-enzymatic antioxidants. These systems scavenge reactive oxygen species (ROS), reducing oxidative stress and preventing cell membrane damage.
- Osmotic Adjustment: Under drought or salt stress, polyploids accumulate higher concentrations of osmolytes, such as proline and soluble sugars. These compounds help maintain cell turgor and hydration during environmental extremes.
- Secondary Metabolite Production: Chromosome doubling often boosts the production of secondary metabolites (e.g., alkaloids, terpenes, and flavonoids), which serve as natural defenses against herbivores and pathogens.
- Hormonal Regulation: Polyploids can exhibit superior ABA (abscisic acid) signaling. Increased ABA production helps regulate stomatal closure and activates stress-responsive genes to mitigate water loss and enhance general adaptability.
3. Genomic and Genetic Mechanisms
The internal genetic environment of a polyploid provides unique mechanisms for adaptation:
- Gene Redundancy and Innovation: Extra gene copies provide a “backup system” that protects the plant from deleterious mutations. This redundancy also allows duplicated genes to undergo neofunctionalization (acquiring new functions) or subfunctionalization (partitioning roles), which can create novel pathways for stress response.
- Epigenetic Flexibility: Polyploidization causes massive epigenetic modifications, such as DNA hypomethylation, which can activate stress-sensitive genes that were previously silenced in diploids. This allows for a more rapid and potent response to environmental challenges.
- Transgressive Expression: Polyploids may express certain genes at levels significantly higher or lower than their progenitors, leading to “extreme” or entirely new phenotypes that offer a competitive edge in harsh environments.
4. Tolerance to Specific Stresses
Biotic (Pathogens): Many polyploids exhibit a higher baseline activation of defense genes, providing a constitutive immune response that is active even before a pathogen is detected.
Drought: Improved through a combination of thicker cuticles, enhanced ABA signaling, higher relative water content, and reduced electrolyte leakage.
Salinity: Enhanced by maintaining better K⁺/Na⁺ ratios in the cytosol and upregulating genes in the SOS (Salt Overly Sensitive) pathway, which helps manage ion toxicity.
Temperature: Polyploids often exhibit superior resistance to cold and freezing due to more rigid cell walls and more negative turgor loss points, while also maintaining higher photochemical integrity during heat stress.
References
Betto, A., Palumbo, F., Riommi, D., Vannozzi, A., & Barcaccia, G. (2025). Harnessing Genomics for Breeding Lantana camara L.: Genotyping and Ploidy Testing of Clonal Lines Through ddRADseq Applications. International Journal of Molecular Sciences, 26(10). https://doi.org/10.3390/ijms26104898
Cervantes-Díaz, C. I., Oyama, K., & Cuevas, E. (2024). Ecological and evolutionary implications of hybridization, polyploidy and apomixis in angiosperms. In Botanical Sciences (Vol. 102, Number 4, pp. 1043–1061). Sociedad Botanica de Mexico, A.C. https://doi.org/10.17129/botsci.3583
Chu, Y., & Lyrene, P. M. (2025). Artificial Induction of Polyploidy in Blueberry Breeding: A Review. In HortScience (Vol. 60, Number 1, pp. 100–110). American Society for Horticultural Science. https://doi.org/10.21273/HORTSCI18263-24
D’Amato, F. (1952). Polyploidy in the differentiation and function of tissues and cells in plants. Caryologia, 4(3), 311–358. https://doi.org/10.1080/00087114.1952.10797544
Gyani, P. C., Mahato, R., Yadav, P., Patidar, O. P., Kushwaha, N., Prajapat, R. K., Roy, A., Gayan, P., Maity, A., & Pakhira, R. (2025). Applications of Artificial Polyploid Induction for Genetic Improvement of Plants. Asian Journal of Current Research, 10(1), 114–130. https://doi.org/10.56557/ajocr/2025/v10i19097
Husband, B. C., Baldwin, S. J., & Suda, J. (2013). The incidence of polyploidy in natural plant populations: Major patterns and evolutionary processes. In Plant Genome Diversity (Vol. 2, pp. 255–276). Springer-Verlag Wien. https://doi.org/10.1007/978-3-7091-1160-4_16
Kolář, F., Čertner, M., Suda, J., Schönswetter, P., & Husband, B. C. (2017). Mixed-Ploidy Species: Progress and Opportunities in Polyploid Research. In Trends in Plant Science (Vol. 22, Number 12, pp. 1041–1055). Elsevier Ltd. https://doi.org/10.1016/j.tplants.2017.09.011
Kosser, S., Bakshi, P., Ji Bhat, D., Jasrotia, A., Angmo, T., & Nisa, Q. (2022). Ploidy manipulation in fruit crops: A Review. Indian Journal of Hill Farming, 35(2), 119–126. https://doi.org/10.56678/iahf-2022.35.02.17
Levin, D. A. (1983). POLYPLOIDY AND NOVELTY IN FLOWERING PLANTS. The American Naturalis, 122(1). http://www.journals.uchicago.edu/t-and-c
Mezzasalma, M., Brunelli, E., Odierna, G., & Guarino, F. M. (2023). Evolutionary and Genomic Diversity of True Polyploidy in Tetrapods. In Animals (Vol. 13, Number 6). MDPI. https://doi.org/10.3390/ani13061033
Mráz, P., & Šingliarová, B. (2025). Natural autopolyploids: Understanding their formation and establishment. American Journal of Botany, 112(5). https://doi.org/10.1002/ajb2.70047
Muhammed, P. K. M., Thomas, B., Seeja, G., Sindura, K. P., Ram, A. A., & Fahida, P. K. (2025). In vitro induction of heteroploidy in Anthurium andreanum Linden. In Vitro Cellular and Developmental Biology – Plant, 61(6), 1309–1315. https://doi.org/10.1007/s11627-025-10570-0
Ramakrishnan, R., Banadka, A., Dubey, S., Al-Khayri, J. M., & Nagella, P. (2025). Advances in triploid plant production: techniques, benefits, and applications. In Plant Cell, Tissue and Organ Culture (Vol. 160, Number 3). Springer Science and Business Media B.V. https://doi.org/10.1007/s11240-025-03011-8
Ramsey, J., & Schemske, D. W. (1998). PATHWAYS, MECHANISMS, AND RATES OF POLYPLOID FORMATION IN FLOWERING PLANTS. In Annu. Rev. Ecol. Syst (Vol. 29). www.annualreviews.org
Ramsey, J., & Schemske, D. W. (2002). Neopolyploidy in flowering plants. In Annual Review of Ecology and Systematics (Vol. 33, pp. 589–639). https://doi.org/10.1146/annurev.ecolsys.33.010802.150437
Rešetnik, I., Schönswetter, P., Temunović, M., Barfuss, M. H. J., & Frajman, B. (2023). Diploid chastity vs. polyploid promiscuity – Extensive gene flow among polyploid cytotypes blurs genetic, morphological and taxonomic boundaries among Dinaric taxa of Knautia (Caprifoliaceae). Perspectives in Plant Ecology, Evolution and Systematics, 59. https://doi.org/10.1016/j.ppees.2023.125730
Sabooni, N., Gharaghani, A., Jowkar, A., & Eshghi, S. (2022). Successful polyploidy induction and detection in blackberry species by using an in vitro protocol. Scientia Horticulturae, 295. https://doi.org/10.1016/j.scienta.2021.110850
Sanaei-Hoveida, Z., & Mortazavian, S. M. M. (2025). The role of polyploidy in improving seed traits, growth kinetics, and embryo development in Cuminum cyminum L. Seed Science Research. https://doi.org/10.1017/S0960258525100056
Šarhanová, P., Majeský, Ľ., & Sochor, M. (2024). A novel strategy to study apomixis, automixis, and autogamy in plants. Plant Reproduction, 37(3), 379–392. https://doi.org/10.1007/s00497-024-00499-6
Sattler, M. C., Carvalho, C. R., & Clarindo, W. R. (2016). The polyploidy and its key role in plant breeding. In Planta (Vol. 243, Number 2, pp. 281–296). Springer Verlag. https://doi.org/10.1007/s00425-015-2450-x
Shinde, A., Jadhav, R., Deshmukh, A., & Thakur, N. (2023). Polyploidy: An Evolutionary Plant Breeding Approach. In Elements of Plant Breeding (pp. 88–105).
Singh, B., Yun, S., Gil, Y., & Park, M. H. (2025). The Role of Colchicine in Plant Breeding. In International Journal of Molecular Sciences (Vol. 26, Number 14). Multidisciplinary Digital Publishing Institute (MDPI). https://doi.org/10.3390/ijms26146743
Sobhanian, H., Song, W.-Y., Soltis, P. S., Soltis, D. E., & Chen, S. (2026). Polyploidy and plant resilience to environmental stresses: Molecular mechanisms and future applications. Plant Communications, 101748. https://doi.org/10.1016/j.xplc.2026.101748
Sutherland, B. L., & Galloway, L. F. (2021). Variation in heteroploid reproduction and gene flow across a polyploid complex: One size does not fit all. Ecology and Evolution, 11(14), 9676–9688. https://doi.org/10.1002/ece3.7791
Thriveni, V., Paul, M., Voruganti, C., Kumar, V. V., Kumar, J. V., Reddy, B. S. K., & Kumar, M. (2024). Impact of Polyploidy on Crop Improvement and Plant Breeding Strategies: A Review. Journal of Advances in Biology & Biotechnology, 27(11), 714–725. https://doi.org/10.9734/jabb/2024/v27i111655
Ulyanov, A. V., Karlov, A. V., & Khatefov, E. B. (2022). The use of maize haploidy inducers as a tool in agricultural plant biotechnology. In Vavilovskii Zhurnal Genetiki i Selektsii (Vol. 26, Number 7, pp. 704–713). Institute of Cytology and Genetics of Siberian Branch of the Russian Academy of Sciences. https://doi.org/10.18699/VJGB-22-85
Walter, E., Biswal, A., Ozias-Akins, P., & Chu, Y. (2025). Leaf Organogenesis Improves Recovery of Solid Polyploid Shoots from Chimeric Southern Highbush Blueberry. BioTech, 14(2). https://doi.org/10.3390/biotech14020048
Wei, X., Zhou, Y., Abbas, F., Yan, F., Zou, X., Yu, Y., Gao, T., He, J., Wang, Q., Yu, R., & Fan, Y. (2023). Distant heteroploid hybridization improved Hedychium floral scent, floral color and morphologcal traits. Industrial Crops and Products, 194. https://doi.org/10.1016/j.indcrop.2023.116357
Wójcik, D., Marat, M., Marasek-Ciołakowska, A., Klamkowski, K., Buler, Z., Podwyszyńska, M., Tomczyk, P. P., Wójcik, K., Treder, W., & Filipczak, J. (2022). Apple Autotetraploids—Phenotypic Characterisation and Response to Drought Stress. Agronomy, 12(1). https://doi.org/10.3390/agronomy12010161
Yali, W. (2024). Polyploidy And Its Importance in Modern Plant Breeding Improvement. https://doi.org/10.31579/2835-7949/021
Zhang, J., Cheng, C., Xiao, F., Zhang, X., Zhang, C., Zhao, Y., Xu, J., Zhang, S., & Wang, X. (2024). Effects of ploidy level on leaf morphology, stomata, and anatomical structure of Hibiscus syriacus L. BMC Plant Biology, 24(1). https://doi.org/10.1186/s12870-024-05778-y






