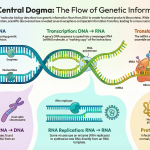
1.0 Introduction to Prokaryotic DNA Replication

DNA replication is one of the most fundamental biological processes, serving as the basis for reproduction, genetic continuity, and the survival of prokaryotic species. In organisms like bacteria, this process must be executed with extraordinary speed and precision to ensure that each daughter cell receives a complete and accurate copy of the genetic blueprint during binary fission.

The replication mechanism in prokaryotes, while complex, is defined by three core characteristics:
• Semi-Conservative: Each newly synthesized DNA molecule is a hybrid, composed of one original parental strand and one newly created daughter strand. This ensures that the genetic information is faithfully passed from one generation to the next.
• Bidirectional: Replication initiates at a single, specific point on the circular chromosome, known as the origin of replication, and proceeds in two opposite directions. Two replication forks move away from the origin, simultaneously copying the DNA until they meet at a termination site on the other side of the chromosome.
• Semi-Discontinuous: Due to the antiparallel nature of the DNA double helix and the directional constraints of the synthesizing enzymes, one of the new strands (the leading strand) is synthesized as a single, continuous piece. The other strand (the lagging strand) is synthesized in short, discontinuous segments that are later joined together.

The efficiency of this system is remarkable. In the well-studied bacterium E. coli, the entire genome of 4.6 million base pairs is replicated in approximately 42 minutes. This is accomplished at a blistering pace, with enzymes adding around 1,000 nucleotides per second to the growing DNA chains. This high-speed, high-fidelity process is orchestrated by a sophisticated toolkit of specialized enzymes and proteins, each performing a precise role in a tightly coordinated sequence.
2.0 The Enzymatic Toolkit: Key Proteins and Their Roles
A coordinated cohort of enzymes and proteins is required to carry out the intricate steps of DNA replication. These components assemble at the replication fork to form a dynamic molecular machine known as the replisome. This section provides a reference guide to the key players involved in prokaryotic DNA replication and their specific functions.
Core Components of the Prokaryotic Replication Machinery
| Enzyme/Protein | Primary Function(s) |
| DnaA | The initiator protein. Binds to the origin of replication (oriC) and triggers the initial unwinding of the DNA double helix. |
| Helicase (DnaB) | Unwinds the DNA double helix by breaking the hydrogen bonds between base pairs, creating two single-stranded templates. Moves along the lagging-strand template in the 5’→3′ direction. |
| Helicase Loader (DnaC) | Facilitates the loading of the DnaB helicase onto the single-stranded DNA at the origin of replication. |
| Primase (DnaG) | An RNA polymerase that synthesizes short RNA primers (approximately 5-12 nucleotides long) on the DNA template. These primers provide the necessary free 3′-OH group for DNA polymerase to begin synthesis. |
| Single-Strand Binding Proteins (SSB) | Bind to and stabilize the separated single strands of DNA, preventing them from reannealing (rewinding) or being degraded by nucleases. |
| Topoisomerase (specifically DNA Gyrase, a Type II Topoisomerase) | Relieves the torsional stress and prevents positive supercoiling ahead of the replication fork by introducing and resealing double-stranded breaks in the DNA. |
| DNA Polymerase III | The main replicative enzyme. Synthesizes new DNA strands by adding complementary nucleotides in the 5’→3′ direction. Possesses 3’→5′ exonuclease activity, which serves as a proofreading mechanism to remove mismatched nucleotides. |
| Sliding Clamp (β-clamp) | A ring-shaped protein that encircles the DNA and tethers DNA Polymerase III to the template, dramatically increasing its processivity (the number of nucleotides added before it detaches). Without this clamp, the polymerase would frequently dissociate from the template, making replication impractically slow and inefficient. |
| Clamp Loader (γ complex) | An ATP-dependent protein complex that loads the sliding clamp onto the DNA at the primer-template junction. |
| DNA Polymerase I | Removes the RNA primers from Okazaki fragments using its 5’→3′ exonuclease activity and fills the resulting gaps with DNA nucleotides. Also possesses 3’→5′ proofreading exonuclease activity. |
| DNA Ligase | Seals the “nicks” (breaks in the phosphodiester backbone) between adjacent Okazaki fragments on the lagging strand, creating a continuous DNA molecule. |
| Tus Protein | The Terminus Utilization Substance. Binds to termination (ter) sequences on the chromosome and acts as a unidirectional fork trap, halting helicase activity and stopping replication. |
| Topoisomerase IV | A Type II Topoisomerase that resolves the topological problem of catenated (interlinked) daughter chromosomes by decatenating them, ensuring they can be segregated into daughter cells. |
The precise interaction of these components in a carefully timed sequence allows for the complete and accurate duplication of the prokaryotic genome.
3.0 The Replication Process: A Three-Stage Breakdown
The complex process of DNA replication is most easily understood when divided into three distinct and sequential stages: initiation, elongation, and termination. Each stage involves a unique set of events as the components of the replisome assemble, operate, and disassemble in a precisely timed sequence.
3.1 Stage 1: Initiation – Launching Replication at oriC

Replication in prokaryotes does not begin randomly but at a single, specific site on the circular chromosome known as the origin of replication (oriC). In E. coli, the oriC region is approximately 245 base pairs long and is characterized by its high concentration of adenine-thymine (AT) base pairs, which are held together by only two hydrogen bonds and are therefore easier to separate than guanine-cytosine pairs. The region is structurally composed of distinct binding sites: three tandem 13-base-pair (13-mer) repeats and four 9-base-pair (9-mer) repeats, also known as DnaA boxes.

The formation of the pre-priming complex proceeds through a series of ordered steps:
1. DnaA Binding: The process begins when multiple copies of the initiator protein DnaA, bound to ATP, recognize and bind to the 9-mer DnaA box sequences within oriC.
2. DNA Melting: This binding causes the DNA to wrap around the DnaA protein complex, inducing stress that leads to the melting or separation of the weaker, AT-rich 13-mer regions. This separation creates a small “replication bubble” of single-stranded DNA.
3. Helicase Loading: The DnaC protein, acting as a helicase loader, recruits two DnaB helicase enzymes and loads one onto each of the single DNA strands within the replication bubble.
4. Fork Establishment: The two DnaB helicases begin to move in opposite directions, powered by ATP hydrolysis, actively unwinding the DNA double helix. This action expands the replication bubble and establishes two bidirectional replication forks, which are the sites of active DNA synthesis.
With the replication forks established and the template DNA strands exposed, the machinery is now assembled and ready to begin the elongation stage.
3.2 Stage 2: Elongation – Synthesizing New DNA Strands
Elongation is the phase where the new DNA strands are synthesized. This stage highlights the elegant solution to a central biochemical problem: how to replicate two antiparallel template strands simultaneously when the primary replicative enzyme, DNA Polymerase III, can only synthesize DNA in one direction (5’→3′).

Before synthesis can begin, two key proteins prepare the template:
• Single-Strand Binding (SSB) Proteins: As the helicase unwinds the DNA, these proteins quickly coat the exposed single strands. This prevents the strands from snapping back together (reannealing) and protects them from enzymatic degradation.
• Topoisomerase (DNA Gyrase): Working ahead of the advancing replication fork, DNA gyrase relieves the torsional stress that builds up as the helix is unwound. It does this by cutting the DNA, allowing it to rotate, and then resealing the break, preventing the chromosome from becoming tangled in positive supercoils.

3.2.1 The Leading Strand: Continuous Synthesis
The leading strand is the new strand synthesized from the parental template that runs in the 3’→5′ direction. Synthesis on this strand is continuous and straightforward. A single RNA primer, synthesized by Primase at the origin, provides the necessary free 3′-OH group for DNA Polymerase III. Held securely to the template by the sliding clamp, the polymerase moves smoothly towards the advancing replication fork, continuously adding complementary DNA nucleotides as new template becomes available.

3.2.2 The Lagging Strand: Discontinuous Synthesis
The lagging strand is synthesized from the parental template that runs in the 5’→3′ direction. Because DNA Polymerase III must still synthesize in the 5’→3′ direction, it must move away from the replication fork’s overall movement. This geometric constraint means synthesis on this strand must be discontinuous, occurring in a series of short segments known as Okazaki fragments, which are typically 1,000–2,000 nucleotides long in prokaryotes. The synthesis of each fragment is a repetitive cycle in which Primase synthesizes a new RNA primer on the exposed template, DNA Polymerase III extends it until it reaches the previous fragment, and the polymerase then detaches to move to the next primer. To coordinate the synthesis of both strands, the lagging strand template loops around, allowing the DNA Polymerase III dimer to move along with the replication fork while synthesizing the lagging strand in the opposite direction.



Once the entire chromosome has been copied through these parallel processes, the machinery proceeds to the final termination and clean-up stage.
3.3 Stage 3: Termination – Completing and Separating the Chromosomes
Termination of replication occurs when the two replication forks, moving in opposite directions around the circular chromosome, meet at a designated terminus region. This region is located roughly 180 degrees from the oriC.
The terminus region contains several termination (ter) sequences. The Tus protein binds tightly to these ter sites, creating a Tus-ter complex that functions as a unidirectional trap. This complex allows a replication fork to pass through from one direction but physically blocks the DnaB helicase when it approaches from the other, effectively arresting fork progression and ending elongation.
With synthesis complete, several final “clean-up” steps are required, particularly on the discontinuously synthesized lagging strand, to produce two intact DNA molecules.
1. Primer Removal: DNA Polymerase I leverages its 5’→3′ exonuclease activity to recognize and remove the RNA primers from the beginning of each Okazaki fragment.
2. Gap Filling: As it removes the RNA nucleotides, DNA Polymerase I simultaneously uses its polymerase activity to fill the resulting gaps with the correct DNA nucleotides, using the parental strand as a template.
3. Nick Sealing: Although the gaps are filled, a “nick” (a break in the sugar-phosphate backbone) remains between the 3′-OH of the newly synthesized DNA and the 5′-phosphate of the adjacent Okazaki fragment. The enzyme DNA Ligase catalyzes the formation of the final phosphodiester bond, sealing the nick and creating a continuous, unbroken lagging strand.

Finally, the cell must resolve a topological challenge. The two newly synthesized circular chromosomes are often interlinked, or catenated, like two rings of a chain. Topoisomerase IV resolves this by making a transient double-strand break in one chromosome, passing the other chromosome through the break, and then resealing it. This decatenation step results in two separate, identical DNA molecules, which can now be segregated into daughter cells during division.

4.0 Conclusion: A High-Fidelity Molecular Machine
Prokaryotic DNA replication is a masterful example of a highly coordinated and efficient molecular system. The precise orchestration of a diverse cast of enzymes—including helicase to unwind the DNA, primase to initiate synthesis, polymerases to copy the template, and ligase to seal the final product—ensures the rapid and complete duplication of the entire genome. This intricate process is not only fast but also remarkably accurate. The intrinsic proofreading capabilities (3’→5′ exonuclease activity) of DNA Polymerase I and III act as a molecular “delete” key, removing incorrectly paired nucleotides and maintaining the high fidelity necessary for genetic stability across generations. Ultimately, this elegant and robust process is the fundamental basis for prokaryotic reproduction, adaptation, and the continuity of life.


Image Summary





References
Hoelz, Derek J., Robert J. Hickey, and Linda H. Malkas. “DNA Replication: Prokaryotes and Yeast.” e LS (2001).
https://www.ncbi.nlm.nih.gov/books/NBK9940/#_ncbi_dlg_citbx_NBK9940
https://www.elearningcampus.io/blog/dna-replication-in-prokaryotes
https://geneticeducation.co.in/prokaryotic-dna-replication-initiation-elongation-and-termination/
https://www.theexpertta.com/book-files/OpenStaxBio2e/14.4%20DNA%20Replication%20in%20Prokaryotes.pdf
https://bio.libretexts.org/Bookshelves/Microbiology/Microbiology_(Boundless)/07%3A_Microbial_Genetics/7.01%3A_Genes/7.1B%3A_DNA_Replication_in_Prokaryotes
https://texasgateway.org/resource/144-dna-replication-prokaryotes
https://bio.libretexts.org/Courses/American_River_College/BIOL_400%3A_Principles_of_Biology_(Wolfe)/04%3A_Untitled_Chapter_4/15%3A_DNA_Structure_and_Function/15.04%3A_DNA_Replication_in_Prokaryotes
https://bio.libretexts.org/Workbench/Bio_11A_-_Introduction_to_Biology_I/17%3A_DNA_Replication/17.03%3A_DNA_Replication_in_Prokaryotes
https://bio.libretexts.org/Bookshelves/Microbiology/Microbiology_(Kaiser)/Unit_7%3A_Microbial_Genetics_and_Microbial_Metabolism/19%3A_Review_of_Molecular_Genetics/19.4%3A_DNA_Replication_in_Prokaryotic_Cells
https://en.wikipedia.org/wiki/Prokaryotic_DNA_replication
https://en.wikipedia.org/wiki/Rolling_circle_replication