1. For liquid cultures, spin down 1-3 mL of culture at 5,000 rpm for 3 min. and remove the media.
2. Add 570 µL of sterile Tris EDTA buffer to the pelleted cells. If agar media are used, choose one colony from the petri dish (using a sterile toothpick) and place the toothpick with the colony into a sterile Eppendorf tube containing the 570 µL of TE buffer.
3. Resuspend the pellet by repeated pipetting or by gently vortexing the solution so that the cells become resuspended.
4. Add 30 µL of 10% SDS and 3 µL of a 20 mg/mL solution of Proteinase K. Mix and incubate for 1 hour at 37°C.
5. After incubation, add 100 µL of 5 M NaCl and mix.
6. Afterwards, add 80 µL of a CTAB/NaCl solution (0.7 M NaCl, 10% CTAB).
7 Incubate this solution at 65°C for 10 min.
8. After incubation, add an equal volume of chloroform: isoamyl alcohol (24:1) and mix.
9. Centrifuge at 12,000 rpm for 5 min and transfer supernatant to a new tube. Be careful not to transfer the interface.
10. Add another equal volume of CI and mix well.
11. Centrifuge at 12,000 rpm for 5 min and transfer the supernatant to a new tube.
12. Add an equal volume of isopropanol and mix gently until the DNA precipitates.
13. Centrifuge at 12,000 rpm for 5 min. and remove isopropanol with the help of a pipette.
14. Add 1 mL of 70% ethanol to the pellet to wash the salt away from the DNA.
15. Centrifuge at 12,000 rpm for 2 min., and discard the ethanol.
16. Dry the tube on the bench top at room temperature.
17. Resuspend the pellet in 50-100 µL of TE buffer and keep it at 4°C until further downstream processes.
Note: The DNA thus extracted can be easily used for PCR.
Composition:
Tris EDTA:
10 mM Tris-HCL
1 mM disodium EDTA
pH 8

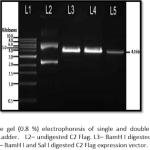
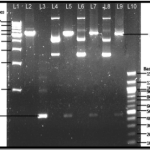



Pingback: Underlying Principle of Different Solutions Used for Isolation of Plasmid from Bacteria by Alkaline Lysis Method - Aneknowledge.com