1. Introduction
The Black Soldier Fly (Hermetia illucens) represents a critical linchpin in the global transition toward circular bioeconomics. As biological intermediaries, these larvae possess a unique metabolic capacity to transform low-value and even contaminated biological resources into high-value proteins and lipids suitable for agri- and aquafeeds. This technical guide evaluates the systemic role of Black Soldier Fly larvae (BSFL) in waste recovery, addressing recent safety breakthroughs in mycotoxin detoxification and the technical hurdles associated with processing complex animal-derived residues. By synthesizing data on bioconversion efficiency and toxicological safety, this document outlines the current landscape of Black Soldier Fly bioconversion as a mechanism for revalorizing waste at a systemic level, bridging the gap between waste management and sustainable food production.

Meet the Larvae: The Ultimate Recyclers
The Black Soldier Fly (Hermetia illucens) is an exceptionally hardy species characterized by a rapid life cycle and a voracious appetite. Their growth rate is staggering: upon hatching, “neonates” weigh a mere 20μg, yet they can increase their body weight by 8,000 times within the nursery phase alone. This biological vigor allows them to handle significant contaminant loads while producing high-quality biomass.

| Phase | Typical Individual Weight | Dietary & Environmental Focus |
|---|---|---|
| Neonate Phase (Day 0–7) | 20μg to 10mg | High-humidity “nursery” feed; reaches a 6mg–10mg milestone by Day 7. |
| Harvest Phase (Day 7–25) | 100mg to 160mg | High-protein/fat “finishing” feed; high tolerance for organic contaminants. |
While these larvae are resilient, the specific “fuel” they consume determines the nutrient yield and the ultimate success of the transformation.
Environmental Impact Pivot Points:
• Nutrient Upscaling: Converting low-value hatchery mortality into high-grade, endogenous protein biomass rather than traditional rendering, which frequently results in nutrient degradation.
• Odor and Emission Stabilization: Replacing the putrid, uncontrolled decomposition of animal matter with stable, mildly acidic biological processes through fermentation and larval digestion.
• Waste Volume Mitigation: Utilizing the larvae’s high substrate reduction capacity to decrease the physical and environmental footprint of hatchery waste.
• Integrated Biosecurity: Shifting from simple disposal toward a managed system that actively mitigates microbiological risks through controlled stabilization and rigorous post-harvest processing.
2. The Strategic Role of BSFL in Circular Bioeconomics
In the framework of circular bioeconomics, invertebrates serve as high-efficiency processors of organic waste. The objective is to transition from traditional agriculture toward integrated sustainable food systems where renewable biological resources are recycled rather than discarded. BSFL are uniquely suited for this intermediary role due to their robust physiological capacity to process diverse and putrescible waste streams.

Key Insight: Circular Bioeconomics Circular bioeconomics focuses on replacing linear “take-make-waste” models with closed-loop systems. By using BSFL to process “spoilt” matter, we transform environmental liabilities into life-sustaining feed ingredients. This strategy effectively recycles renewable biological resources, reducing the environmental footprint of traditional agri- and aquafeed production.
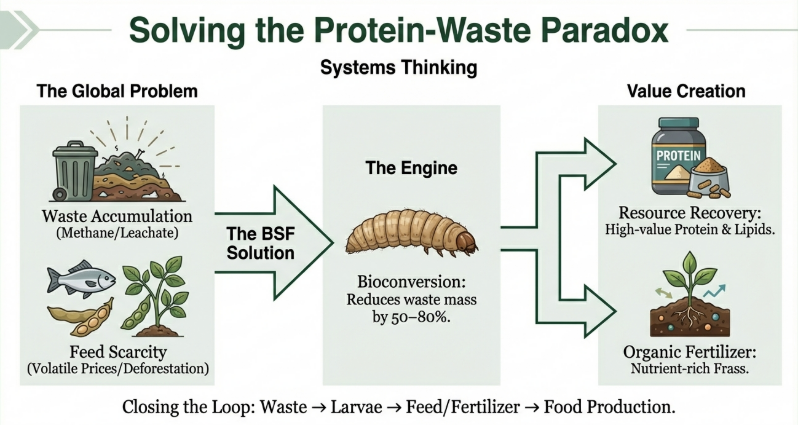
Based on technical assessments from recent bioconversion studies, several characteristics make BSFL the ideal candidate for industrial waste processing:
• Nutritional Versatility: Capable of subsisting on nearly any biological material, including rotting produce, manure, and sewage sludge.
• High Contaminant Tolerance: Demonstrates significant resilience to heavy metals and organic pollutants (pesticides, dioxins, and PCBs).
• Rearing Efficiency: Undemanding regarding rearing conditions with short life cycles, facilitating frequent, high-volume harvesting.
• Economic Valorization: Facilitates the repurposing of materials previously deemed “lost,” such as fungal-infected fallen fruit or biological sludge from aquaculture and the paper industry.
The strategic importance lies in the larvae’s ability to recover nitrogen and energy from contaminated streams that were historically prohibited from the feed chain. This creates a viable economic incentive for waste recovery while significantly reducing the environmental burden of traditional disposal methods.

3. Mycotoxin Resilience and Detoxification: The Deoxynivalenol (DON) Analysis
A primary barrier to utilizing BSFL for grain recycling is the presence of mycotoxins—toxic secondary metabolites from fungi. Current legislative frameworks often apply the same maximum levels for mycotoxins to insects as they do to traditional livestock feed, effectively barring insects reared on “spoilt” grain. However, research into Deoxynivalenol (DON), the most prevalent grain mycotoxin in Europe, indicates that BSFL may provide a biological solution.
Analytical Extraction and Methodology
Assessing safety requires precise quantification in a complex larval matrix. As a researcher, it is critical to note that BSFL can contain up to 49% lipids, primarily lauric acid, which creates non-covalent hydrophobic interactions with analytes. Reliable detection requires a modified μQuEChERS approach (acidifying the solvent with 0.1% formic acid) and a crucial n-heptane defatting step to remove interfering lipids. This is followed by UPLC-HRMS (Ultra-Performance Liquid Chromatography High-Resolution Mass spectrometry) for unambiguous identification.
Findings on Active Detoxification
A study utilizing a diet contaminated with 29 mg/kg of DON showed that BSFL growth remained unaffected. However, detoxification is a temporal physiological process rather than an instantaneous state; DON concentrations in larvae actually doubled to 0.60 mg/kg by Day 11 of exposure before declining to base levels by Day 15 as metabolic adaptation occurred.
| Analyte | Feed Concentration (mg/kg) | BSFL Larvae (mg/kg, Day 15) | Frass/Excreta (mg/kg, Day 15) |
|---|---|---|---|
| DON (Deoxynivalenol) | 29.0 ± 1.4 | 0.27 ± 0.01 | 28.5 ± 1.05 |
| DON3G | 0.2 ± 0.1 | Below LOQ | 0.26 (Max Day 13) |
| 3ADON | 2.6 ± 0.4 | Below LOQ | 0.46 (Max Day 8) |
| 15ADON | 3.8 ± 0.5 | Below LOQ | 0.46 (Max Day 8) |
The larvae do not accumulate mycotoxins at levels exceeding regulated limits for feed ingredients. However, the frass (larval excreta) retains nearly all the initial contamination. Consequently, while the larvae are safe, the frass must be strictly excluded from agricultural applications to prevent toxicant re-entry into the environment.
4. Valorizing Hatchery Residues (HR)
4.1 Nutritional and Texture Optimization
Hatchery residues (HR)—comprising unmarketable chicks, non-viable eggs, and shells—represent a significant environmental burden. BSFL bioconversion is a superior alternative to landfilling, but it introduces complex nutritional and physical challenges.
While rich in protein and fat, HR is naturally deficient in carbohydrates, creating an imbalanced protein-to-carbohydrate (P:C) ratio that can limit larval performance.
• Unmarketable Chicks/Embryos: Provide high protein, but microbial/enzymatic hydrolysis of their connective tissue can cause substrate instability.
• Eggs (Liquid/Whole): Rich in lipids but contribute to a viscous, “sticky” texture that hinders larval movement.
• Eggshells: High in ash and calcium; excessive shell inclusion leads to calcium toxicity, which inhibits growth and increases mortality.
To balance this, carbohydrate supplements like potato residues (peelings and cuttings) are often added. However, educators must note two critical failure points for raw potato: its 75% water content can oversaturate the habitat, and its raw starch is largely indigestible to BSFL. To make these residues viable, they require heat or enzymatic (amylase) pre-treatment.
While nutritional balance is key, the physical habitat and chemical environment of the waste pile must be meticulously managed to prevent total colony collapse.
Nutritional Imbalance and Supplementation
HR is rich in protein and lipids but carbohydrate-deficient, leading to an imbalanced Protein-to-Carbohydrate (P:C) ratio that can hinder development. Experimental supplementation with 13% (dry basis) potato residues (peelings/cuttings) has been tested to balance this ratio, though raw starch exhibits limited digestibility in BSFL without heat treatment.
For optimal larval development, a balanced Protein-to-Carbohydrate (P:C) ratio is essential. While hatchery residues are rich in proteins and fats, they are inherently carbohydrate-deficient. Larval performance is maximized on diets approaching a 1:1 ratio of protein to non-fiber carbohydrates; pure HR substrates skew this balance, leading to developmental delays.
Supplementation with secondary co-products, such as potato residues (peelings and cuttings), has been evaluated to provide the necessary starch. However, as an engineer, one must account for the Solanine Risk: raw potato residues contain solanine, a glycoalkaloid that can exert lethal effects on insect populations and must be monitored during substrate formulation.
| Component (Dry Basis) | HR Nutrient Profile | HR + Potato Residue Profile |
|---|---|---|
| Dry Matter (DM) | 42% – 44% | 38% – 40% |
| Nitrogen | 3.8% | 4.1% |
| Starch/Carbohydrates | Deficient | Supplemented (13% inclusion) |
| Crude Lipids (Unfermented) | 12.6% | 14.2% |
| Crude Lipids (Fermented) | ~3.3% | ~3.7% |
| Ash (incl. Eggshells) | 48.2% | 42.3% |
Physical Impacts and Larval Mortality
1. Odour Control: Fermentation successfully shifts the substrate profile from putrid (decomposing carcasses) to mildly acidic and stable.
2. Substrate Texture and Asphyxiation: Fermented HR undergoes progressive liquefaction due to enzymatic hydrolysis. This leads to larval drowning; specifically, the sticky, liquefied substrate causes the obstruction of larval spiracles, leading to asphyxiation.
3. Calcium Toxicity: Fermentation solubilizes calcium carbonate from eggshells. Higher ash content in larvae from fermented groups suggests excessive calcium absorption, which may contribute to mortality.
| Metric | Control (Gainesville) | Unfermented HR | Fermented HR |
|---|---|---|---|
| Larval Yield (g) | 209.4 ± 18.1 | 65 ± 50.3 | 16.8 ± 9.0 |
| Bioconversion Rate (%) | 13.6% | 4.4% | 1.6% |
| Reduction Rate (%) | 50.8% | 19.9% | 12.3% |
| Survival Rate (%) | 97.8% | 76.1% | 13.4% |
4.2 The Invisible Hand: pH and Fermentation
The chemical environment, specifically pH, is a “make or break” survival factor. If the substrate becomes too acidic (pH 2–4), larval performance plummets. Fermentation is often used as a pre-treatment to stabilize waste and reduce the putrid odors of decomposing carcasses, dropping the initial pH to roughly 5.6.
While fermentation improves odor and reduces coliforms, it actually increases the concentration of Clostridium spp. (rising from 7 log cfu/g to 8 log cfu/g). Acidic environments do not inactivate these spores; instead, they can regerminate later, posing a significant safety risk.
The Optimal pH Journey:
• Starting Point: Fresh Hatchery Residue (~6.4 pH).
• Fermented State: Pre-treated stabilized waste (~5.6 pH).
• Harvest-Ready State: Active bioconversion by healthy larvae naturally raises the environment to an alkaline 8.0–9.0 pH.
Beyond chemical balance, the physical state of the habitat—specifically the structural integrity of the substrate—is the primary determinant of larval survival.
The Goal of Pre-treatment: Why Ferment the Waste?
We use fermentation as a passive stabilization technique to improve the microbiological profile and odor of the waste. Because HR is carbohydrate-deficient, we must supplement it with a sugar source—typically lactose from whey permeate—to fuel Lactic Acid Bacteria (LAB) activity. This process creates an acidic environment that suppresses specific spoilage organisms.
The following table contrasts the baseline challenges of raw waste with our fermentation objectives, grounded in specific experimental metrics:
| Feature | Raw Hatchery Waste Challenges | Fermentation Pre-treatment Goals | Data / Metrics (Source-Verified) |
|---|---|---|---|
| Microbiological | High initial coliform and E. coli loads. | Significant reduction of enteric pathogens. | Raw: 5 log cfu/g COL <br> Fermented: <2 log cfu/g COL |
| Odoric Profile | Putrid odors from decomposing carcasses. | Stable, mild, acidic, and “pleasant” odor. | Qualitative improvement in social acceptability. |
| pH Stability | Neutral pH favors rapid decomposition. | Rapid acidification to inhibit spoilage. | Raw: pH ~6.4 <br> Fermented: pH ~5.6 |
The data forces us to conclude that while fermentation “fixes” the immediate sensory and enteric pathogen issues, it creates a new, complex microbial landscape that we must navigate with caution.
Microbiological Shifts: Successes and Persistent Risks
Students must recognize that fermentation is a stabilization tool, not a sterilization method. The resulting shifts in microbial populations present a “Microbiological Paradox.”
• Reductions (Successes):
◦ Coliforms & E. coli: Fermentation effectively suppresses these to <2 log cfu/g.
◦ Salmonella spp.: While significantly reduced, students must note that Salmonella is not eliminated, with persistent loads of 3 log cfu/g often detected in fermented diets.
• Persistent Threats:
◦ The Clostridium Paradox: Fermentation fails to control spore-forming Clostridium spp. (measured at 8 log cfu/g in diets). Spores survive the acidic pH 5.6 and germinate as the larvae process the waste. Crucially, while unfermented diets reach an alkaline pH of 8–9, fermented diets peak and stabilize at pH 7.0, which is still sufficient for Clostridium to proliferate to 9 log cfu/g in the larvae.
◦ The Listeria Surprise: Counter-intuitively, research shows that Listeria monocytogenes levels can be significantly higher in plant-based control larvae (4.2 log cfu/g) than in HR-fed larvae. This suggests that certain animal-waste environments may actually be less hospitable to Listeria than standard grain-based diets.
The following matrix summarizes the specific microbiological hazards identified during the evaluation of starter larvae and hatchery residue (HR) diets.
Microbiological Hazard Matrix
| Pathogen | Initial Source (Diet/Starter Larvae) | Observed Persistence/Level |
|---|---|---|
| Salmonella spp. | Starter Larvae (6.0 log cfu/g) | Persistent in harvested larvae (up to 4.3 log cfu/g). |
| Listeria monocytogenes | Starter Larvae (5.9 log cfu/g) | Persistent in harvested larvae (up to 4.2 log cfu/g). |
| Clostridium spp. | Fermented Diet (8.1 log cfu/g) | Strategic Risk: Proliferated to 8.9 log cfu/g in larvae. |
| Coliforms (COL) | Unfermented HR Diet (5.0 log cfu/g) | High proliferation in larvae (6–8 log cfu/g). |
4.3 Texture and Moisture: The “Drowning” Risk
In high-protein animal waste like HR, a phenomenon called liquefaction occurs. This is caused by extensive microbial and enzymatic hydrolysis of the connective tissues in chick carcasses. As the substrate turns from a firm “pâté” into a viscous liquid, the larvae lose structural support and aeration.
Without a porous habitat, larvae become immobilized at the surface and die from asphyxiation or drowning. To combat this, structural co-products like sawdust or fibrous vegetable waste must be added.
Troubleshooting Guide for Substrate Management
| Observation | Resulting Risk | Solution |
|---|---|---|
| Liquefaction | Asphyxiation/Drowning; larvae cannot extract themselves. | Add fibrous “structural co-products” (e.g., sawdust) to improve aeration. |
| Excessive Shells | Calcium Toxicity; inhibited growth and high mortality. | Use separation technologies to remove shells before processing. |
| Water Saturation | Substrate oversaturation (common with raw potato). | Supplement with dry, absorbent materials like wheat bran; pre-treat potato to reduce moisture. |
While the larvae navigate the physical and chemical habitat, they simultaneously act as a biological filter, effectively managing several dangerous toxins.
The Physical Paradox: Substrate Liquefaction and the “Drowning” Hazard
While fermentation improves biological stability, it frequently causes the physical collapse of the substrate. This is the primary driver of the high mortality rates (often >90%) observed in fermented animal-waste trials.
The Divergent Physical Paths
• Unfermented HR: This substrate starts viscous but solidifies over time. This transition is beneficial, as it improves structural support and aeration for the larvae.
• Fermented HR (The 5-Day Shift):
◦ Initial Texture: Firm, creamy, “pâté-like” and navigable.
◦ Liquefaction: After approximately 5 days of bioconversion, the substrate liquefies. This is driven by enzymatic hydrolysis of connective tissues and protein denaturation caused by the initial acidification.
◦ The Calcium Factor: Fermentation solubilizes Calcium Carbonate (CaCO3) from eggshells. This leads to excessive calcium intake and potential toxicity, further weakening the larvae.
Impact on Larvae
The resulting viscous liquid traps the larvae. They exhibit “escape behavior,” attempting to climb container walls to avoid the sludge. The sticky substrate obstructs their spiracles (breathing holes), leading to asphyxiation and mass drowning. To mitigate this, a ratio of 50g of sawdust per 100g of diet must be added to maintain texture and porosity.
1. Fermentation is for Odor, Not Sterility: It suppresses coliforms but permits the germination of Clostridium spores once the pH stabilizes at 7.0.
2. Texture is a Survival Metric: Without structural additives (like sawdust), fermented high-protein waste will liquefy, leading to larval asphyxiation.
3. Post-Harvest Treatment is Mandatory: Because BSFL live in their own dejections (frass), even “clean-fed” larvae must undergo treatments like blanching to ensure they are microbiologically safe for the feed chain.
4.4 The Safety Filter: Handling Mycotoxins and Pathogens
A primary benefit of BSFL is their ability to consume “spoilt” materials without accumulating organic contaminants. In studies using a substantial load of 29 mg/kg of Deoxynivalenol (DON), larvae successfully metabolized the toxin, keeping their own biomass concentrations safely below regulatory limits. However, there is a sharp distinction between organic toxins and persistent pathogens.
Safety Checklist: Contaminant Management
• Naturally Managed by Larvae (Not Accumulated):
◦ DON (Deoxynivalenol): Larvae remain safe even at high (29 mg/kg) exposure levels.
◦ Pesticides & Organic Drugs: Generally metabolized or passed into frass.
◦ Coliforms: Initial loads are often reduced by larval activity.
• Persistent Risks (Requires Post-Harvest Heat Treatment):
◦ Clostridium Spores: Spores survive fermentation and larvae; require high-heat slaughter.
◦ Salmonella & Listeria: These can persist in the larval gut and require blanching to ensure safety.
This filtering ability creates a clear divergence between the two primary end products of the bioconversion cycle.
4.5 Harvest and Outputs: Biomass vs. Frass
The harvest produces two distinct materials. Aspiring producers must be aware of the “yield gap”: while BSFL are generally high in nutrients, those fed specifically on fermented HR typically show lower protein (26–30% DM) and significantly lower lipids (8–12% DM) compared to larvae fed on standard grain diets.
| Output | Larval Biomass (Nutrients/Safety) | Frass (Contaminant Load/Safety) |
|---|---|---|
| Characteristics | Concentrated protein and lipids; organic toxins remain low. | Contains feces, moulted cuticles, and unconsumed shells. |
| Safety & Use | Safe for agri/aquafeed ONLY after slaughter methods like blanching. | High Risk: Contains concentrated pathogens and toxins (like DON); contraindicated for feed. |

This underscores the larvae’s role as a biological “safety filter,” pushing contaminants into the residual waste (frass) while purifying the nutrients within their own bodies.

5. The Microbiological Safety Frontier: Pathogen Management
Microbiological safety is paramount when processing animal-based residues. Both plant-based and animal-based diets carry risks of Salmonella spp., Listeria monocytogenes, and Clostridium spp.

Effectiveness and Systemic Risks
• Initial Reduction: Fermentation reduces initial coliform and Salmonella loads compared to raw residues but fails to eliminate spore-forming Clostridium spp.
• Larval Reservoirs: Larvae and frass often exhibit higher microbial loads than the initial diet. A critical finding is that “starter larvae” (5-day-old neonates) can be a primary source of contamination, with loads of Salmonella and Listeria reaching 6 log cfu/g before inoculation.
• Post-Harvest Treatment: Because BSFL act as a reservoir for pathogens regardless of diet safety, post-harvest treatments like blanching or heat treatment are non-negotiable for regulatory compliance.
Regulatory Outlook
Current EU regulations strictly ban most animal-derived feeds for insects. However, data indicates that the microbiological risks of final products from poultry-based residues are not significantly higher than those from plant-based diets, provided proper sterilization is applied. This challenges the necessity of the current animal-feed ban for non-ruminant residues.

6. Conclusion and Strategic Takeaways
The Black Soldier Fly larva is a robust tool for the circular bioeconomy, capable of detoxifying mycotoxins and valorizing complex hatchery waste. Achieving industrial success requires customized analytical methods, such as UPLC-HRMS with defatting steps, and precise management of substrate texture to prevent spiracle obstruction and larval mortality.

Strategic Directives:
• Detoxification Adaptation: Recognition that mycotoxin detoxification is a metabolic process requires monitoring larvae across the full development cycle to ensure safety.
• Texture Management: HR bioconversion requires structural or absorbent additives (e.g., sawdust) to prevent substrate liquefaction.
• Frass Protocol: Because frass contains both residual mycotoxins and high pathogen loads, it must be treated as “spoiled meat” or “non-composted animal carcasses” rather than standard fertilizer until it undergoes further stabilization like composting.
Image Summary




Questions/Answers
1. How can BSF bioconversion transform organic waste into value?
Black soldier fly (BSF) bioconversion transforms low-value organic waste into value through a biological recycling process where larvae ingest decaying organic matter and convert it into high-quality nutrient streams and stabilized residues. This process creates value across multiple sectors, primarily by producing protein-rich biomass for animal feed, nutrient-dense organic fertilizers, and sustainable energy sources.
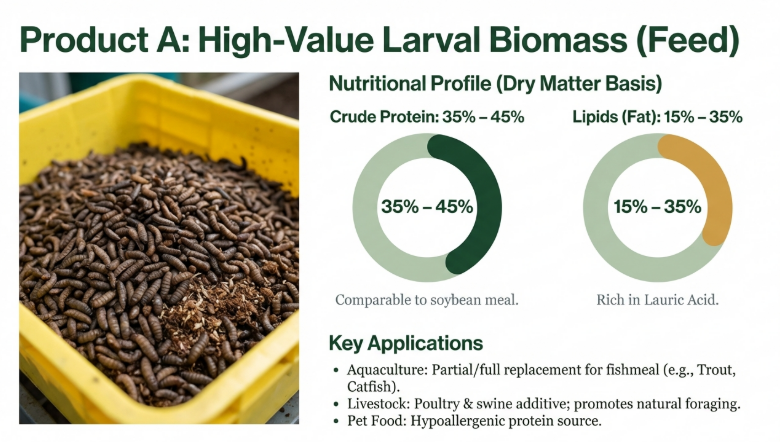
1. Production of High-Value Biomass (Larvae and Prepupae)
The primary value generated is the larval biomass itself, which typically contains 35–45% crude protein and 15–35% lipids on a dry matter basis.
• Alternative Animal Feed: BSFL serve as a sustainable substitute for expensive and environmentally impactful protein sources like fishmeal and soybean meal. They have been successfully integrated into diets for aquaculture (tilapia, salmon, shrimp), poultry (laying hens, broilers), swine, and pets.
• Nutritional Quality: Larvae possess a balanced amino acid profile and are rich in lauric acid, a medium-chain fatty acid with antimicrobial and health-promoting properties for livestock.
• Economic Advantage: Utilizing waste as a free or low-cost feedstock reduces feed production costs, which can account for 60–70% of total expenses in poultry and swine farming.
2. Creation of Organic Fertilizers (Frass)
The residual material left after bioconversion, known as frass, consists of larval excreta, shed exoskeletons, and undigested substrate.
• Soil Amendment: Frass is a nutrient-rich byproduct containing nitrogen, phosphorus, and potassium (N-P-K) along with a high organic matter content (around 90% DM).
• Agronomic Performance: Field and pot trials have demonstrated that stabilized frass can match or exceed the performance of mineral fertilizers and traditional compost in promoting the growth of crops such as tomato, chili, maize, and ryegrass.
• Biological Benefits: Frass contains chitin, which can stimulate plant defense mechanisms and enhance resistance to pathogens.
3. Bioenergy and Bio-industrial Applications
BSF bioconversion facilitates the production of renewable energy as part of an integrated biorefinery model.
• Biodiesel: Larval lipids can be extracted and processed into high-quality biodiesel through transesterification, showing fuel properties comparable to rapeseed-based biodiesel.
• Biogas: BSFL can serve as a pretreatment for food waste, liquefying organic matter to increase its biomethane potential during subsequent anaerobic digestion.
• Chitin/Chitosan: Larval exoskeletons are a source of chitin, a biopolymer with versatile applications in medicine, wastewater treatment, and cosmetics.
4. Environmental and Sanitary Valorization
BSF bioconversion “upcycles” waste by providing critical ecosystem services that have intrinsic and economic value.
• Waste Mass Reduction: Larvae can reduce the dry matter of organic waste by 50–80%, drastically decreasing the volume of material destined for landfills.
• Emission Mitigation: The process enables up to a 90% reduction in greenhouse gas emissions compared to conventional waste management strategies. Aerobic larval activity suppresses methane generation.
• Pathogen and Odor Control: BSFL activity reduces populations of harmful bacteria such as E. coli and Salmonella. Additionally, it can reduce odorous compounds (e.g., phenols, indoles, fatty acids) in manure by over 87%.
• Pest Mitigation: The presence of BSFL naturally inhibits the breeding of nuisance house flies (Musca domestica).
5. Efficient Harvesting (Self-Harvesting)
The “self-harvesting” behavior of prepupae adds operational value; as they attempt to pupate, they instinctively migrate out of the moist substrate to a high, dry place. This natural habit simplifies collection and reduces the labor and energy required for processing.
2. What benefits does BSF meal provide for livestock health?
Black soldier fly (BSF) meal provides a wide array of health benefits for livestock, primarily driven by its high nutritional density and the presence of unique bioactive compounds.
1. Bioactive Compounds and Immune Support
BSF meal is rich in substances that actively promote immune health and disease resistance:
• Antimicrobial Peptides (AMPs): BSF larvae naturally secrete a diverse repertoire of AMPs, such as defensins and cecropins, which exhibit potent activity against various pathogenic bacteria, fungi, and viruses. These peptides can serve as a natural alternative to synthetic antibiotics, helping to mitigate the spread of multi-drug resistant pathogens.
• Lauric Acid: BSF lipids are exceptionally high in lauric acid (C12:0), a medium-chain fatty acid known for its strong antimicrobial, anti-inflammatory, and antioxidant properties. It is particularly effective at inhibiting Gram-positive pathogens and supports gastrointestinal health by acting as a prebiotic.
• Chitin: The larval exoskeleton contains chitin, which functions as an indigestible fiber that stimulates the immune system. Chitin and its derivatives (like chitosan) have been shown to increase the weight of immunological organs and enhance the phagocytic activity of macrophages, thereby improving innate immunity.
2. Gut Health and Microbiota Modulation
BSF meal acts as a remodeling factor for the gastrointestinal tract across various species:
• Prebiotic Effects: Chitin serves as a substrate for beneficial gut bacteria, promoting the production of short-chain fatty acids (SCFAs) like butyrate. These SCFAs provide energy for intestinal epithelial cells, promote mucosal growth, and help inhibit acid-intolerant pathogens like Salmonella and E. coli.
• Pathogen Suppression: The active compounds in BSF meal help reduce the overall pathogenic load in the gut. For example, in pigs, it has been shown to reduce diarrhea and intestinal inflammation by decreasing the abundance of opportunistic pathogens like Fusobacterium.
3. Species-Specific Health Benefits
• Poultry: BSF meal improves the immune response of laying hens and broilers, evidenced by improved blood biochemical profiles (e.g., higher globulin levels). It also has a positive impact on animal welfare; for example, providing live larvae as an enrichment can reduce aggressive pecking and feather damage in layers by satisfying their natural foraging instincts.
• Swine: In pigs, BSF meal supports gastrointestinal function during the critical post-weaning phase, reducing food neophobia and improving overall health status.
• Aquaculture: For various fish species, BSF meal inclusion enhances antioxidative capacity and non-specific immunity. It has been specifically noted to prevent intestinal enteritis induced by soybean meal in rainbow trout.
4. Nutritional Density and Bioavailability
BSF meal is a high-quality protein source (typically 30–50% crude protein) with a balanced essential amino acid profile similar to fishmeal. It also contains highly bioavailable minerals, including calcium, phosphorus, zinc, and iron, which support enzymatic functions, metabolic processes, and skeletal development. This superior nutrient bioavailability can reduce the need for supplemental inorganic minerals in livestock rations.
3. How do environmental factors influence the BSF life cycle?
Environmental factors such as temperature, humidity, light, and substrate conditions are critical determinants of the Black Soldier Fly (BSF) life cycle, influencing everything from the duration of developmental stages to survival rates and adult reproductive success. Because BSF are native to tropical and warm-temperate regions, their biological processes are highly optimized for these environments.
Temperature
Temperature is the most pivotal factor affecting the metabolic rates and development times of BSF.
• Optimal Range: The most efficient development for all life stages occurs at approximately 27°C to 30°C. At 27°C, larvae tend to be roughly 10% longer and 5% heavier than those reared at 30°C.
• Cold Exposure: BSF are sensitive to cold and cannot persist in regions where temperatures consistently drop below 5°C. Low temperatures slow metabolism, extending the larval stage—typically two weeks under ideal conditions—to as long as four months.
• Heat Stress: High temperatures (30°C to 36°C) accelerate metabolism, resulting in smaller adults with shorter lifespans. While temperatures up to 50°C can be tolerated briefly, survival rates decline sharply above 36°C, and 47°C is considered lethal.
Humidity and Moisture
Humidity and substrate moisture significantly impact survival and the physical ease of processing the larvae.
• Relative Humidity (RH): An RH of 60% to 80% is optimal for BSF development. Higher humidity levels enhance egg hatching success and adult emergence rates. Conversely, low humidity (around 25% RH) causes high egg mortality due to desiccation through the egg membrane.
• Substrate Moisture: Larvae require moist environments but are sensitive to extremes. The optimal moisture content for food waste substrates is 70% to 85%.
◦ Excessive Moisture: Substrates above 85% moisture can create anaerobic conditions, which may immobilize larvae, lead to foul odors, and cause mass fatalities.
◦ Insufficient Moisture: If the substrate is too dry, it hampers the breakdown of organic matter and can cause larvae to migrate prematurely in search of a more suitable environment.
Light Source
Light is primarily essential for the adult stage, as it triggers the behaviors necessary for reproduction.
• Mating: Adult BSF require direct light to initiate mating, which typically occurs on the wing. Approximately 85% of mating activity happens in natural sunlight at an intensity of about 110 μmol m⁻²s⁻¹.
• Artificial Lighting: For indoor rearing, artificial light sources must provide a wavelength between 450 and 700 nm to stimulate mating behaviors; rare-earth lamps (350–450 nm) are generally ineffective.
Substrate pH and Nutritional Quality
The chemical and nutritional makeup of the larval feed dictates their final size and body composition.
• pH Levels: BSFL are highly resilient and can survive in substrates ranging from pH 2 to 10, though they grow significantly better in slightly acidic to alkaline environments (pH 6 to 10). Larvae are capable of regulating the pH of their medium to more neutral or alkaline levels as they feed.
• Dietary Composition: Substrates rich in proteins and soluble carbohydrates support faster growth and higher bioconversion rates. Nutrient-deficient or high-fiber diets (such as those rich in lignin) can significantly delay maturation into prepupae by up to two months.
• Larval Density: High stocking densities (e.g., above 5 larvae/cm²) can increase the substrate temperature through metabolic heat, potentially causing heat stress, but may also facilitate the breakdown of complex fibers through “social digestion”.
4. How does BSF frass compare to chemical fertilizers in crops?
Black soldier fly (BSF) frass compares favorably to chemical fertilizers, often matching or even exceeding their performance in terms of crop yield while providing significant additional benefits to soil health, biodiversity, and pest resistance.
1. Nutrient Profile and Release Kinetics
While chemical fertilizers provide nutrients in a rapidly available mineral form, BSF frass typically functions as a slow-release organic fertilizer.
• Balanced Nutrients: Frass possesses a balanced N:P₂O₅:K₂O ratio of approximately 1:0.9:1.1, making it an effective compound fertilizer.
• Nitrogen Dynamics: A significant portion of nitrogen in frass is organically bound, releasing gradually as it mineralizes. This reduced synchrony during early growth can be a disadvantage, but it supports sustained nutrient availability during peak demand periods and reduces leaching risks compared to chemical alternatives.
• Immediate Availability: Despite its slow-release nature, some nitrogen in frass is present as plant-available ammonium, which can match the early-season growth stimulus of mineral fertilizers.
2. Crop Growth and Yield Performance
Multiple studies demonstrate that BSF frass can achieve yield parity with chemical fertilizers.
• Edamame: In production trials, edamame yields in frass-treated plots (~84.27 g/plant) were not significantly different from those receiving commercial NPK fertilizer (~85.05 g/plant).
• Maize: Field studies showed that BSF frass provided a higher nitrogen fertilizer equivalence than urea, resulting in greater nitrogen uptake and grain yield.
• Solanaceous Crops: In greenhouse trials, frass-amended pots supported tomato, brinjal (eggplant), and chili yields that matched mineral NPK programs.
3. Soil Health and Ecosystem Services
Unlike chemical fertilizers, which can lead to soil degradation and microbial decline over time, frass actively improves the soil ecosystem.
• Organic Matter: Frass is composed of roughly 90% organic matter, which enhances soil structure, porosity, and water retention.
• Microbial Stimulation: Frass application increases soil microbial biomass, activity, and diversity, including beneficial groups like mycorrhizal fungi and nitrogen-fixing bacteria.
• Biodiversity: Frass significantly increases the abundance of soil and foliar arthropods, particularly beneficial predators like ground beetles and spiders, which helps maintain a balanced ecosystem that chemical inputs often disrupt.
4. Pest Suppression and Systemic Resistance
One of the most distinct advantages of frass over chemical fertilizer is its role in biologically mediated pest regulation.
• Chitin Priming: Frass contains chitin from larval sheddings, which triggers plant defense pathways (such as jasmonic acid signaling), priming systemic resistance against herbivores and pathogens.
• Reduced Damage: Trials on edamame showed that frass-treated plants maintained significantly lower pest damage (~5-6%) compared to NPK-treated plants (~15-20%).
5. Environmental and Economic Considerations
• Sustainability: BSF bioconversion is a climate-smart alternative, producing significantly fewer greenhouse gas emissions (especially methane) than conventional composting or landfilling.
• Circular Economy: Using frass recycles low-value organic waste into a high-value agricultural input, reducing the reliance on expensive and energy-intensive synthetic fertilizers.
• Stabilization Requirement: It is important to note that fresh frass can be phytotoxic due to high salinity or ammonium levels. Proper stabilization through composting or curing is necessary to reach a germination index above 80% to ensure safety for sensitive crops.
5. Can BSF larvae lipids be used for biodiesel production?
Yes, Black Soldier Fly (BSF) larvae lipids are a highly viable and efficient feedstock for biodiesel production. Research demonstrates that BSF larvae (BSFL) can accumulate significant amounts of lipids—typically between 20% and 40% of their dry weight—making them comparable to traditional oilseed feedstocks like rapeseed and soybean.
Production and Efficiency
• Transesterification Process: Larval fat is isolated and then converted into biodiesel through a process called transesterification, where fatty acids are reacted to form methyl esters.
• High Yield: Studies have reported that the conversion rate from extracted larval oil to standard-quality biodiesel is approximately 93% by mass.
• Ideal Lipid Profile: BSFL lipids are particularly rich in medium-chain saturated fatty acids, such as lauric acid, and relatively low in polyunsaturated fatty acids. While high saturated fat content is less ideal for human health, it is considered ideal for biodiesel production because it enhances the fuel’s stability.
Comparative Advantages
• Sustainable Feedstock: Unlike traditional biodiesel crops, BSFL do not compete with human food production for agricultural land or water resources. They can be grown on non-arable land and have a significantly higher per-acre productivity.
• Rapid Growth: Larvae reach harvestable stages in just 14 to 28 days, a much shorter cycle than dedicated energy crops like maize or rapeseed.
• Waste Valorization: BSFL produce high-quality lipids while simultaneously reducing the volume of organic waste (such as restaurant scraps and livestock manure) by 50–80%.
Fuel Performance and Standards
• Standard Compliance: BSFL-derived biodiesel has demonstrated fuel properties that are comparable to rapeseed-based biodiesel.
• Meeting Regulations: Analysis shows that BSF biodiesel meets most requirements of the European Biodiesel Standard (EN14214), including parameters for density, viscosity, flash point, and cetane index.
Integrated Biorefinery Potential
In a circular economy model, biodiesel production can be part of an integrated BSF biorefinery. For instance, 1,000 kg of wet food waste can be processed by larvae to yield approximately 23 kg of biodiesel while the remaining lipid-extracted larval biomass and frass (residue) are utilized for animal feed, biogas production, or organic fertilizers.
6. How do BSF larvae reduce harmful pathogens like Salmonella?
Black soldier fly larvae (BSFL) reduce harmful pathogens like Salmonella through a combination of biological, chemical, and physical mechanisms that occur during the bioconversion of organic waste. Research has shown that BSFL can significantly decrease Salmonella populations in various substrates, with reported reductions exceeding 4 to 7 log₁₀ units.
The primary ways BSFL reduce these pathogens include:
• Antimicrobial Peptides (AMPs) and Bactericidal Compounds: BSFL naturally produce and secrete a diverse repertoire of AMPs, such as cecropins and defensins, which induce lysis in the cell membranes of both Gram-negative and Gram-positive bacteria. These compounds are part of the larvae’s innate immune system, helping them survive and thrive in pathogen-rich environments.
• Alkaline Gut Environment and Enzymatic Action: The high pH levels (often between 8.5 and 9) in the posterior regions of the BSFL gut, combined with a suite of potent digestive enzymes (including proteases and lysozymes), act to inactivate and degrade ingested bacteria.
• Ammonia Generation: During the digestion of protein-rich wastes like manure, larval activity promotes an alkaline environment that facilitates the conversion of ammonium ions into uncharged ammonia (NH₃). This increased ammonia concentration is a major driver of antimicrobial effects, as it is toxic to many pathogenic microorganisms.
• Microbial Competition: The established gut microbiota of BSFL, which includes genera like Dysgonomonas and Bacillus, can outcompete and suppress pathogens for resources and niche space. These beneficial symbionts may also produce inhibitory molecules that further restrict the growth of Salmonella.
• Physical Ingestion: Larvae are voracious feeders that directly ingest huge volumes of organic matter, including the indigenous pathogenic bacteria present in the substrate.
Important Safety Considerations: While BSFL are highly effective at reducing the pathogenic load in the manure or waste substrate, the pathogens may not be entirely eliminated. Viable Salmonella can sometimes be detected within the larval gut at the end of the rearing period. Therefore, for the larvae to be used safely as animal feed, post-harvest decontamination—such as blanching or other heat treatments—is essential to ensure the final product is free of pathogens.
7. Can larvae safely process substrates contaminated with heavy metals?
Black soldier fly larvae (BSFL) are highly resilient and can process substrates contaminated with heavy metals, but the safety of the resulting products depends heavily on the specific element and concentration involved. While the larvae can survive and even gain weight in metal-rich environments, they do not eliminate these contaminants; instead, they redistribute them between their bodies and the residual frass.
Physiological Impact and Tolerance
BSFL show a remarkably high tolerance for heavy metals like cadmium, lead, and zinc, often completing their life cycle with minimal effects on survival under laboratory conditions. However, high concentrations can cause significant negative side effects, such as reduced growth rates, lower final larval weight, and decreased adult fecundity. For instance, elevated zinc levels in substrates have been linked to reduced egg yield and higher larval mortality, suggesting that “zinc poisoning” can disrupt colony stability.
Migration and Accumulation Patterns
The larvae act as biological filters, but their accumulation behavior is element-specific.
• Bioaccumulation: Metals like cadmium (Cd) and lead (Pb) are frequently bioaccumulated within the larval tissues, sometimes reaching concentrations that exceed those in the feeding substrate. Mercury (Hg) also shows a tendency for phased accumulation in the larval body as development progresses.
• Exclusion: Conversely, BSFL demonstrate a strong ability to exclude arsenic (As) and chromium (Cr), retaining over 90% of these metals in the frass (residue) rather than absorbing them into their biomass.
• Growth Dilution: A “growth dilution effect” has been observed, where the relative concentration of heavy metals per unit of body weight decreases as the larvae rapidly accumulate mass.
Safety of End Products
The distribution of heavy metals poses distinct risks for the two primary BSF coproducts:
• Larval Biomass: If larvae bioaccumulate toxic metals like Cd or Pb, the resulting meal may be unsuitable for animal feed or human food without strict quality control to ensure they meet international regulatory limits. However, extracted larval oil often maintains low metal concentrations even when the larvae are reared on contaminated sludge, potentially allowing its use as technical oil or for biodiesel.
• Frass (Residue): Because metals like As and Cr are concentrated in the frass, this byproduct may require safety assessments or stabilization treatments (such as chemical passivation or secondary composting) before it can be safely used as an agricultural fertilizer to prevent environmental accumulation.
In summary, while BSFL can effectively bioconvert contaminated waste, strict source control of feedstock, continuous monitoring of final products, and adherence to safety standards are essential to manage the risk of heavy metals entering the food chain.
8. How does BSF frass affect soil microbial diversity and pests?
Black soldier fly (BSF) frass significantly enhances soil microbial diversity and serves as a powerful biological control agent against pests by stimulating both the soil ecosystem and the plant’s internal defense mechanisms.
Impact on Soil Microbial Diversity
The application of BSF frass improves soil health by increasing microbial activity, biomass, and diversity.
• Beneficial Taxa Enrichment: Frass application has been shown to increase the abundance of beneficial microbial groups in the rhizosphere, such as mycorrhizal fungi, nitrogen-fixing bacteria, and Chitinophagaceae, which are associated with nutrient cycling and plant growth promotion.
• Community Reshaping: BSFL activity can markedly increase the number of microbial taxa in a substrate and reshape the community structure of the soil it is applied to. In specific trials using wheat straw frass, there was a notable increase in the phylum Bacteroidota and genera like Acinetobacter, which are efficient at degrading complex organic matter.
• Humification and Stability: The microbial succession during frass composting facilitates the decomposition of recalcitrant substances like lignin and cellulose, transforming unstable organic matter into stable humic substances that further support soil biological health.
Impact on Pests and Disease Suppression
BSF frass functions as an effective tool for biologically mediated pest regulation, often outperforming chemical fertilizers in minimizing plant damage.
• Predator Attraction: Frass application significantly increases the abundance of soil and foliar arthropods, particularly beneficial predators like ground beetles and spiders. These predators help maintain a balanced ecosystem and reduce herbivore pressure.
• Systemic Resistance Priming: A primary mechanism for pest suppression is the presence of chitin (from larval sheddings) and microbial fragments in the frass. These components trigger jasmonic acid (JA) and salicylic acid (SA) pathways in plants, priming systemic resistance against herbivores and pathogens.
• Significant Damage Reduction: In field trials with edamame, frass-treated plants maintained consistently low damage levels (~5–6%) compared to plants treated with NPK or compost, which reached damage levels of ~15–20%.
• Specific Pest Suppression: Chitin-fortified BSF frass has demonstrated the ability to suppress soil-borne pests such as potato cyst nematodes, reducing their reproduction rates by up to 75% while increasing crop yields.
• Pathogen Inhibition: The inherently alkaline pH of stabilized frass (often around 7.5–8.3) may further inhibit the growth of certain soil pathogens. Additionally, BSFL activity during the bioconversion process itself reduces pathogenic loads of E. coli and Salmonella in the initial organic waste, leading to a safer end product.
While BSF frass provides these benefits, researchers note that fresh frass can be phytotoxic due to high salinity or ammonium levels; therefore, proper stabilization or composting is recommended to maximize its agronomic value and safety.
9. Can BSF frass match the early-season growth of mineral fertilizers?
Whether Black Soldier Fly (BSF) frass can match the early-season growth of mineral fertilizers depends largely on its stabilization and the specific crop, though it is generally characterized as a slow-release organic fertilizer.
Slow-Release Dynamics and Early Growth
Most research indicates that BSF frass does not typically match the rapid nutrient delivery of mineral fertilizers in the early season:
• Reduced Synchrony: Nitrogen release from BSF frass is initially slower than mineral alternatives, which can reduce synchrony during the early stages of crop growth.
• Long-Term Benefit: Because much of its nitrogen is organically bound, frass is better suited as a long-term fertilizer that provides sustained nutrient availability during peak demand periods rather than an immediate burst.
• High C:N Ratio: A comparably high carbon-to-nitrogen ratio and low initial shares of ammonium nitrogen often result in limited immediate nutrient release.
Instances of Comparable Performance
Despite its slow-release nature, BSF frass can sometimes match mineral fertilizer performance under certain conditions:
• Immediate Ammonium Availability: Stabilized frass contains a portion of nitrogen as immediately available ammonium-N, which has been shown to support early canopy growth and chlorophyll status in Solanaceous crops like tomato and brinjal, matching the performance of mineral NPK programs.
• Comparable Growth to Ammonium Nitrate: Some studies have reported that BSF frass can produce plant growth comparable to ammonium nitrate (NH4NO3), suggesting that environmental factors like soil type, moisture, and microbial activity can accelerate nutrient mineralization.
• Higher Nitrogen Equivalence in Maize: In some field trials, BSF frass provided a higher nitrogen fertilizer equivalence than urea, resulting in greater nitrogen uptake and grain yield, even if the initial release was slower.
Optimization Strategies
To bridge the gap in early-season performance, researchers often recommend:
• Combined Application: The highest yields are frequently achieved by combining BSF frass with mineral NPK fertilizers, utilizing the mineral component for early growth and the frass for sustained nutrition and soil health.
• Proper Stabilization: Curing and composting the frass can help reduce phytotoxicity and optimize the nitrogen-release profile for better crop compatibility.
• Soil Incorporation: Incorporating frass into the topsoil ensures that mineral nitrogen is placed where it can be most effective as it mineralizes over time.
10. Why must fresh frass be stabilized before applying it to crops?
Fresh black soldier fly (BSF) frass must be stabilized before crop application primarily to mitigate phytotoxicity, ensure microbiological safety, and improve nutrient stability. While frass is a nutrient-rich byproduct of bioconversion, in its “fresh” or raw state, it often possesses characteristics that can inhibit plant growth or pose health risks.
1. Prevention of Phytotoxicity
Fresh frass is often “incompletely stabilized,” meaning it contains high levels of substances that are harmful to plants in concentrated amounts:
• Ammonia Toxicity: Raw frass frequently contains high levels of ammonia nitrogen (NH₄-N), which can induce osmotic stress, impair root respiration, and inhibit seed germination.
• High Salinity: The process of bioconversion can concentrate inorganic matter and ions, leading to high electrical conductivity (EC) levels in fresh frass. This high salinity is strongly correlated with reduced germination rates and can negatively affect soil health if not moderated through stabilization.
• Dissolved Organic Matter: Fresh frass contains high concentrations of dissolved organic carbon (DOC) and volatile organic acids that continue to decompose rapidly. This ongoing decomposition can deplete soil oxygen and produce toxic phenolic compounds that hinder plant development.
2. Microbiological Safety and Hygiene
Although BSF larvae significantly reduce the pathogenic load of the organic waste they consume, the resulting frass is not always sterile:
• Pathogen Persistence: Residual pathogens such as Salmonella spp., Escherichia coli, and various bacterial spores or helminth eggs can persist in fresh frass.
• Antibiotic Resistance: Manure-derived frass may still harbor antibiotic resistance genes (ARGs) that could be disseminated into agricultural soils if the material is not properly treated.
• Stabilization Methods: Standardized stabilization, such as aerobic composting or heat treatment (e.g., 70 °C for 60 minutes), is effective at reducing these microbial risks to safe levels.
3. Enhanced Nutrient and Biological Stability
Stabilization processes like curing and composting transform the frass into a more beneficial agricultural input:
• Humification: Composting promotes the formation of humic substances, converting labile (unstable) organic matter into stable humus that improves soil structure, water retention, and microbial activity.
• Nitrogen Conservation: Stabilization helps convert inorganic nitrogen into more stable, organically bound forms. This reduces the risk of ammonia volatilization—where nitrogen is lost to the atmosphere as gas—thereby preserving the fertilizer’s nutritional value.
• Maturity Indicators: A common metric for safety is the Germination Index (GI); stabilized frass typically reaches a GI of ≥80%, indicating it is fully mature and no longer phytotoxic to sensitive crops.
4. Regulatory Compliance
In many jurisdictions, stabilization is a legal requirement. For example, under European Union regulations, insect frass intended for use as a fertilizer must undergo heat treatment at 70 °C for 60 minutes to ensure it is free of Salmonella and that Enterobacteriaceae levels are sufficiently reduced.
11. What are the technical limits to extracting lipids for fuel?
The technical limits to extracting lipids from black soldier fly larvae (BSFL) for fuel primarily involve trade-offs between extraction efficiency, oxidative stability, and environmental impact, along with the high sensitivity of advanced extraction methods to process parameters.
1. Extraction Efficiency and Recovery Rates
• Entrapment in Emulsions: While aqueous extraction is a more environmentally sustainable “green” method, it often yields lower lipid recovery than solvent-based methods. This is because lipids frequently become trapped within soluble solids or form oil-in-water emulsions, making them difficult to separate even with high centrifugation forces.
• Solvent Residues vs. Yield: Traditional organic solvent-based methods are highly efficient at extracting total lipids but can introduce chemical residues. These residues may be unsuitable if the defatted biomass is intended for concurrent use in feed applications.
2. Chemical and Oxidative Stability
• Free Fatty Acid Formation: Mechanical pressing and thermal extraction methods can lead to the formation of free fatty acids and oxidative degradation depending on the shear forces and temperatures used.
• Oxidative Instability: Early methods involving thermal extraction posed concerns regarding oxidative stability. High temperatures and exposure to air during processing can degrade the quality of the lipids, which is critical since lipid quality directly determines the quality of the resulting biodiesel.
3. Process Sensitivity and Optimization
• Parameter Precision: Advanced techniques like enzyme-assisted or supercritical CO₂ extraction can achieve superior defatting efficiency, but they are highly sensitive. Minor deviations in temperature or enzyme concentrations can profoundly alter the resulting lipid composition and fatty acid profile.
• Refining Requirements: Extracted larval oil often requires additional processing to remove odors, colors, phospholipids, and free fatty acids to meet industrial standards or increase applicability in specific markets.
4. Infrastructure and Economic Constraints
• Capital and Energy Intensity: Building and running industrial-scale BSFL production and lipid extraction systems is capital intensive. The drying stage, necessary before many extraction processes, is a significant point of high energy consumption, which can impact the overall global warming potential (GWP) of the fuel unless renewable energy sources are used.
• Scalability Challenges: Translating successful laboratory-scale extraction to an industrial scale is difficult due to the heterogeneity of the waste substrates used to rear the larvae, which can lead to inconsistent lipid yields and quality.
Despite these limits, BSFL lipids are considered an ideal feedstock for biodiesel because they are naturally rich in medium-chain saturated fatty acids (like lauric acid), which provides greater fuel stability compared to high-PUFA (polyunsaturated fatty acid) sources.
12. How does larval density impact waste reduction and growth?
Larval density is a critical operational parameter in black soldier fly (BSF) bioconversion that significantly influences growth rates, individual biomass, survival, and waste reduction efficiency through a complex interplay of competition and cooperation. The impact of density is not universal but is highly dependent on the specific type and nutritional quality of the feeding substrate.
Impact on Larval Growth and Development
• Individual Weight vs. Total Yield: Higher larval densities typically lead to decreased individual growth rates and smaller final prepupal weights due to intense competition for limited food and space. However, while individual weight may decline, total larval yield (biomass per unit area) often increases with density up to a certain threshold.
• Substrate-Specific Responses: In nutrient-dense substrates like chicken feed, individual weight consistently decreases as density increases. Conversely, on high-fiber or low-quality substrates (such as vegetable waste or wheat straw), higher densities may actually enhance growth compared to very low densities.
• Social Digestion: On fibrous materials, larvae benefit from “social digestion” or aggregation effects, where the combined secretion of extracellular enzymes and increased microbial activity facilitate the breakdown of complex polysaccharides.
• Developmental Timing: Increasing larval density generally prolongs the developmental period because nutrient limitations and competition slow the rate at which larvae reach the maturity required for pupation.
Impact on Waste Reduction and Bioconversion
• Mass Reduction: Higher larval densities generally lead to greater total waste reduction because a larger population can process the material more voraciously. For example, in experiments with wheat straw, a medium density (300 larvae per group) achieved an optimal conversion rate of 42.9%, outperforming lower densities.
• Substrate Underutilization: Low densities may fail to fully exploit the bioconversion potential of the waste, leaving significant portions of the substrate unconsumed.
• Inhibitory Effects of Overcrowding: Excessive crowding can be counterproductive; it can lead to the buildup of excreted waste and unconsumed feed, which creates sticky, anaerobic conditions that hinder larval movement and oxygen availability.
• Optimal Ranges: Commonly reported optimal densities range from 5 larvae per gram for agricultural residues to 8–10 larvae per gram for protein- and carbohydrate-rich food waste.
Physiological and Environmental Interactions
• Metabolic Heat: High stocking densities generate significant metabolic heat, which can increase substrate temperatures to over 45°C. While this can improve feed assimilation in cool environments, it risks causing lethal heat stress (above 47°C) and accelerates moisture loss through evaporation.
• Survival Rates: Survival typically remains stable across various densities in high-quality feeds but declines markedly in high-moisture or low-quality substrates at elevated densities due to poor aeration and microbial proliferation.
• Nutritional Composition: Larval density can modulate the biochemical profile of the larvae, specifically affecting crude fat content more than protein levels. Higher densities can sometimes trigger lipid accumulation as a metabolic response to carbon-rich diets.
Strategic Density Management
• Goal-Oriented Density: For producers aiming to maximize individual larval size (e.g., for specific animal feed markets), lower densities are preferable. If the goal is maximal waste volume reduction, higher densities should be employed.
• Tailored Approaches: Research suggests that food industry wastes are better utilized at higher densities, whereas fiber-rich agricultural residues perform optimally at lower to moderate densities.
13. What happens to heavy metals and toxins during bioconversion?
During black soldier fly (BSF) bioconversion, heavy metals and toxins are redistributed between the larval biomass and the residual frass through a dynamic management strategy characterized by selective exclusion, phased accumulation, and growth dilution. While BSF larvae (BSFL) are highly resilient and can process contaminated substrates, they do not eliminate these substances; instead, they either metabolize and degrade them or concentrate them in specific system components.
1. Heavy Metals and Inorganic Contaminants
The migration of heavy metals is element-specific, meaning BSFL handle different metals in distinct ways:
• Bioaccumulation: Metals such as cadmium (Cd) and lead (Pb) are frequently accumulated within larval tissues. Cd, in particular, shows strong enrichment with bioaccumulation factors as high as 9.62. Pb often accumulates significantly in the larval exoskeleton, and its concentration in the body can be reduced through the molting process.
• Exclusion/Rejection: BSFL demonstrate a powerful ability to exclude arsenic (As) and chromium (Cr), with over 90% of these metals being retained in the frass rather than being absorbed into the larval biomass.
• Growth Dilution Effect: As larvae rapidly gain mass, the relative concentration of heavy metals per unit of body weight often decreases. This means that harvesting larvae when their biomass is maximal but before pupation can minimize metal risks in derived products.
• Impact on Larvae: High concentrations of metals like zinc (Zn) can lead to “zinc poisoning,” which reduces larval growth rates, survival, and adult fecundity.
2. Antibiotics and Antibiotic Resistance Genes (ARGs)
BSFL-mediated bioconversion acts as a biological filter for medical and veterinary residues:
• Gene Mitigation: BSFL treatment significantly reduces the relative abundance of tetracycline resistance genes (tetA, tetB, tetG) and pathogenic hosts associated with them in manure residues.
• Active Degradation: Larvae and their associated gut microbiota can degrade up to 97% of tetracycline, along with other drugs like roxithromycin and trimethoprim.
• Gut Retention: While manure residues become safer, certain ARGs (such as sul1 and qnrB) and β-lactamase-related genes can accumulate within the larval gut. This retention highlights the critical need for post-harvest decontamination if larvae are intended for feed.
3. Mycotoxins and Pesticides
BSFL possess robust innate detoxification pathways for many organic toxins:
• Non-Accumulation: Studies indicate that BSFL do not accumulate mycotoxins such as Aflatoxin B1, Zearalenone, Ochratoxin A, or Deoxynivalenol (DON) in their biomass even when exposed to high levels in their feed.
• Residue Contamination: While the larvae remain low in mycotoxin content, the frass often remains highly contaminated. For example, in a study using DON-contaminated feed, the larvae actively detoxified the toxin, but the frass remained unsuitable for downstream agricultural use due to high DON concentrations.
• Pesticide Stability: BSFL are susceptible to certain insecticides like moxidectin and cyromazine, which can be lethal. However, they generally do not concentrate pesticides, and the bioconversion process can actually shorten the half-lives of pesticides in the resulting compost compared to untreated substrates.
4. Pathogens and Sanitization
BSFL improve the hygiene of organic waste through several mechanisms:
• Pathogen Reduction: Larval activity can reduce populations of Salmonella spp. and E. coli by 4 to 7 log₁₀ units.
• Antimicrobial Action: This sanitization is driven by the secretion of antimicrobial peptides (AMPs), competitive gut microbiota, and the generation of an alkaline gut environment (pH 8.5–9.0).
• Persistence: Despite overall reductions in the substrate, some pathogens may persist within the larval gut, making post-harvest treatments like blanching or heat treatment essential for safety.
14. Are there specific crops that perform best with frass?
Research across various studies indicates that BSF frass is an effective organic fertilizer for a wide array of crops, often achieving yield parity with chemical fertilizers while improving soil health.
1. Solanaceous Crops (Tomato, Eggplant, and Chili)
Solanaceous species have shown exceptional performance with BSF frass application. Greenhouse trials demonstrate that tomato, brinjal (eggplant), and chili yields treated with frass are statistically comparable to those achieved with mineral NPK fertilizers. For these crops, the agronomic optimum was identified at an application rate equivalent to 150 kg of plant-available nitrogen per hectare. Cured frass provides immediately available ammonium-N that supports early canopy growth and chlorophyll status in these species.
2. Legumes (Edamame and Clover)
• Edamame (Soybean): Field evaluations found that edamame plants in frass-treated plots produced an average yield of 84.27 g per plant, which did not significantly differ from the 85.05 g per plant produced in commercial NPK plots.
• Red Clover: In nutrient-poor sandy soils, frass application significantly increased total nitrogen and carbon, leading to improved plant cover and vigor.
3. Leafy Greens (Lettuce, Arugula, and Pak Choi)
Leafy greens respond rapidly to the nutrients in frass:
• Lettuce and Arugula: These crops achieved significantly higher fresh and dry biomass when grown in frass-amended media compared to standard peat-only controls. Researchers found that frass can effectively replace 10–20% of peat in potting substrates without compromising performance.
• Pak Choi: Successful growth has been documented, though users are cautioned that high application rates of unstabilized frass may temporarily inhibit seed germination due to salt or ammonium toxicity.
4. Cereals and Field Crops (Maize, Wheat, and Canola)
• Maize: Field studies compared BSF frass to commercial organic fertilizers and urea, finding that frass provided a higher nitrogen fertilizer equivalence, resulting in superior nitrogen uptake and grain yield.
• Wheat and Canola: Both crops showed significant nutrient contributions and improved soil fertility, even in sandy soils low in nutrients, following frass application at rates up to 10 t/ha.
• Ryegrass: Frass consistently outperformed exclusive chemical fertilization in promoting soil fertility resilience, leading to sustained or increased production in subsequent cultivation cycles.
5. Other Specialized Applications
• Radish: Seedlings treated with frass derived from wheat straw bioconversion exhibited the best growth performance, characterized by an increased number of leaves and an enriched abundance of beneficial soil microbial groups.
• Reforestation Species: Calibrated frass application doubled the height and significantly increased the biomass of the fast-growing tree species Dodonaea madagascariensis in nursery trials.
Key Performance Factor: Stabilization
While many crops perform well, the sources emphasize that fresh frass can be phytotoxic. To ensure the best performance across all crop types, frass should be stabilized through composting or curing until it reaches a germination index of ≥80%. Properly stabilized frass functions as a slow-release nitrogen source, supporting sustained nutrient availability during peak demand periods.
15. How is frass stabilized to prevent toxicity to plants?
Frass stabilization is primarily achieved through aerobic composting, curing, and thermal hygienisation, which transform raw larval residue into a mature organic fertilizer. This process is necessary because “fresh” or raw frass often contains high levels of ammonia, salts, and organic acids that can cause osmotic stress or “nutrient burn” in plants.
The following steps are required to effectively stabilize frass and eliminate its phytotoxicity:
1. Aerobic Composting and Curing
Composting is the most common method used to stabilize frass by allowing microorganisms to break down labile organic matter into stable humus.
• Thermo-Enzymatic Degradation: Using a mechanical reactor can accelerate the process, maintaining temperatures between 55–65°C to rapidly degrade organic matter. This thermophilic phase promotes the breakdown of phytotoxins like volatile fatty acids and phenolics.
• Curing Phase: After the initial high-heat phase, frass should undergo aerobic curing (typically 7–14 days) at moisture levels of 35–45%. This period allows for the completion of short-term oxidation and reduces ammoniacal phytotoxicity.
• Biological Synergy: The process benefits from the synergistic action of BSF intestinal microbes and exogenous composting microorganisms, which together facilitate nitrogen retention and humification.
2. Thermal Hygienisation
To meet safety and regulatory standards (such as those in the EU), frass must be heat-treated to ensure it is free of pathogens like Salmonella.
• Standard Parameters: Effective stabilization involves heating the frass to 70°C for 60 minutes.
• Alternative Method: Maintaining the coldest point of the material at ≥55°C for at least 72 hours with regular turning also effectively improves hygiene and stability.
3. Substrate Modification (C:N Ratio and Bulking)
Adjusting the chemical and physical structure of the frass during rearing or post-harvest can significantly improve its final quality.
• C:N Ratio Adjustment: Straw or sawdust can be added as bulking agents to reach a carbon-to-nitrogen ratio of 15:1 to 30:1. This balance is critical for microbial metabolism and avoids the slow decomposition associated with high-lignin straw alone.
• Salinity Reduction: Supplementing substrates with materials like rice straw can help reduce the electrical conductivity (EC) of the final frass, making it safer for salt-sensitive crops.
4. Maturity Indicators
The effectiveness of the stabilization process is measured using the Germination Index (GI).
• Stability Threshold: Frass is considered fully mature and non-phytotoxic when the GI reaches ≥80%.
• Enhanced Results: Properly stabilized frass can achieve a GI as high as 180%, meaning its water extract not only lacks toxicity but actively promotes seed germination and seedling vigor.
Through these methods, the nitrogen in the frass is converted from volatile ammonia into stable, organically bound forms or nitrates, providing a slow-release nutrient source that sustains plant growth without the risks associated with fresh manure.
16. What are the specific legal hurdles for BSF in food?
The legal hurdles for utilizing Black Soldier Fly (BSF) in human food are significant and vary widely by region, primarily revolving around novel food classifications, strict rearing substrate restrictions, and lack of international standardization.
1. European Union: The Novel Food Barrier
In the EU, the primary legal hurdle for BSF as human food is the Novel Food Regulation (EU) 2015/2283.
• Authorization Process: Any insect intended for human consumption must undergo a rigorous safety assessment by the European Food Safety Authority (EFSA). This requires extensive data on the insect’s composition, production methods, toxicological safety, and allergenic potential.
• Pending Status: While BSF is authorized for use in animal feed (aquaculture, poultry, and swine) under Regulation (EU) 2017/893 and (EU) 2021/1925, it has not yet been added to the list of authorized edible insects for human consumption in the EU.
• Substrate Restrictions: Even for animal feed, the EU prohibits rearing insects on “catering waste” (household/restaurant waste), manure, or sewage sludge. They must be reared on plant-based or a limited set of non-ruminant animal-origin materials (like eggs or honey) to prevent the risk of transmitting pathogens or prions.
2. United States: Regulatory Complexity
In the U.S., the landscape is divided between food and feed regulators:
• FDA and AAFCO: The Food and Drug Administration (FDA) considers insects food if they are produced specifically for that purpose under Current Good Manufacturing Practices (cGMP). However, the Association of American Feed Control Officials (AAFCO) has historically been more restrictive, initially approving dried BSFL only for salmonid fish and requiring they be raised exclusively on feed-grade materials.
• Defect Action Levels: Legally, the U.S. still characterizes insects in general as a “defect” or impurity in food up to a certain threshold, rather than explicitly defining them as a standard food source.
3. Global and International Standardization
• Codex Alimentarius: One of the largest global hurdles is that insects are not currently listed as “food” in the Codex Alimentarius, the international standard for food safety. Instead, they are only mentioned as impurities that contaminate other products.
• Halal and Religious Compliance: In regions with large Islamic communities, there are ongoing legal and religious debates regarding the halal status of meat from animals (or the insects themselves) fed on substrates like manure or feces.
• Genetic Engineering: In the EU, any BSF improved through genome-editing (like CRISPR/Cas9) is classified as a Genetically Modified Organism (GMO) under Directive 2001/18/EC. This subjects these strains to exceptionally strict environmental risk assessments and approvals, which can delay or block their industrial use.
4. Safety and Traceability Concerns
• Allergenic Potential: Legal frameworks often require labeling due to cross-reactive allergens, particularly for consumers allergic to crustaceans or mites, as BSFL contain tropomyosin and arginine kinase.
• Contaminant Accumulation: Regulatory authorities remain cautious about BSFL because they can bioaccumulate heavy metals (especially cadmium, lead, and mercury) from contaminated waste streams. Confirming that these toxins do not enter the human food chain is a prerequisite for legalizing waste-fed BSF.
17. How do BSF larvae compete with microbes in organic waste?
Black soldier fly larvae (BSFL) utilize a complex combination of physical, chemical, and biological strategies to compete with and suppress microbial populations within organic waste.
1. Physical Ingestion and Direct Competition
BSFL are voracious detritivores that directly compete with microbes by rapidly consuming massive amounts of organic matter. During this process, they ingest the microbes present in the waste, effectively using them as a nutrient source. High larval densities further intensify this competition, as a large population can process material faster than indigenous microbes can metabolize the available nutrients.
2. Secretion of Antimicrobial Peptides (AMPs)
A primary chemical defense mechanism is the production of potent antimicrobial peptides (AMPs), such as cecropins and defensins. These peptides are secreted into the environment or act within the larval gut to induce lysis in the cell membranes of both Gram-negative and Gram-positive bacteria. The BSF genome contains a diverse repertoire of these genes, which are often expressed in a diet-dependent manner to help the larvae thrive in pathogen-rich environments.
3. Environmental Modification (pH and Ammonia)
Larval activity profoundly alters the physical chemistry of the substrate to make it less hospitable for many microbes:
• Alkaline Shift: BSFL actively shift the pH of their substrate toward highly alkaline levels (often reaching pH 9.0 or higher).
• Ammonia Toxicity: This alkaline environment facilitates the conversion of ammonium ions into uncharged ammonia (NH3), which is highly toxic to many pathogenic microorganisms and is a major driver of sanitization.
• Temperature Elevation: In high-density rearing environments, the intense metabolic activity of the larvae generates significant heat, often raising substrate temperatures to over 45°C, which can inhibit the growth of heat-sensitive bacteria.
4. Aeration and Aerobic Competition
The constant movement of larvae aerates the substrate, which disrupts the anaerobic conditions required by many fermentative microbes. This aeration specifically suppresses methanogenesis (the production of methane) and other fermentative microbiological processes that occur under oxygen exclusion.
5. Synergies with Gut Microbiota
The larvae’s internal gut microbiota (predominantly phyla such as Firmicutes, Proteobacteria, and Bacteroidetes) acts as a biological filter. These beneficial symbionts outcompete opportunistic pathogens for resource space and may produce their own inhibitory molecules that further restrict the growth of external microbes.
Impact on Pathogens
Through these combined mechanisms, BSFL bioconversion can significantly reduce the populations of harmful bacteria such as Salmonella enterica and Escherichia coli, with reported reductions of 4 to 7 log₁₀ units in various manure and food waste substrates. While the substrate is sanitized, it is important to note that some pathogens may persist within the larval gut, necessitating post-harvest treatments like blanching or heat drying if the larvae are intended for feed.
18. How does social digestion help larvae break down fibrous wastes?
Social digestion is a density-mediated facilitation process that allows Black Soldier Fly larvae (BSFL) to more effectively process structurally complex, fibrous materials like lignocellulose and cellulose. While high larval densities usually lead to competition for food in nutrient-rich substrates, they provide significant advantages when larvae are confronted with difficult-to-digest fibrous wastes.
Social digestion assists the breakdown of fibrous waste through the following mechanisms:
• Aggregation and Collective Secretion: At higher densities, larvae tend to aggregate, which leads to the collective secretion of extracellular enzymes. This combined enzymatic force is more effective at breaking down the tough tissue network of complex polysaccharides than individual feeding efforts.
• Microbial Synergy: Aggregation promotes increased microbial activity within the substrate. Larvae and their associated gut microbiota work collaboratively to degrade cellulose and hemicellulose. Larval activity also reshapes the microbial community in the waste, enriching for beneficial, cellulolytic taxa like the phylum Bacteroidota and genera such as Acinetobacter.
• Physical Texture Modification: Larval movement and muscular activities physically disrupt the compact and rigid surfaces of fibrous materials. This mechanical action transforms the waste into a rougher, more porous texture, significantly increasing the surface area available for both larval enzymes and microbial colonization.
• Metabolic Heat Generation: High stocking densities generate significant metabolic heat, which can raise the temperature within the substrate to over 45°C. This increased temperature can enhance digestive processes and the activity of certain enzymes, such as proteases, which reach their peak activity at these higher temperatures.
• Improved Aeration: Continuous larval movement through the waste provides constant aeration, shifting the environment from anaerobic to aerobic. This shift promotes the growth of aerobic microbial communities that further assist in the breakdown of recalcitrant substances.
In summary, for fiber-rich substrates like vegetable waste or wheat straw, higher larval densities facilitate a “social” approach where physical disruption, collective enzyme production, and microbial stimulation work together to improve nutrient accessibility and conversion efficiency.
19. How do different drying methods affect the larvae’s nutritional quality?
Drying methods significantly influence the nutritional and physical quality of black soldier fly (BSF) larvae by affecting protein solubility, lipid oxidation, digestible amino acid scores, and physical appearance. While substrate composition typically has a larger overall impact on the larvae’s final nutritional profile, the chosen drying process determines how well those nutrients are preserved and utilized.
1. Oven-Drying
Oven-drying is a versatile method usually conducted at temperatures between 50°C and 120°C.
• Optimal Temperature: Drying at 60°C is considered ideal for maintaining protein solubility and minimizing tissue collapsing, shrinkage, and the Maillard reaction (browning).
• High-Heat Effects: Excessive heat can lead to the degradation of heat-sensitive components and reduced nutrient stability.
2. Freeze-Drying
Freeze-drying is a non-thermal process that removes moisture while the larvae are frozen.
• Visual Quality: It is the most effective method for preserving the natural color of the larvae, resulting in the “whitest” powder because it completely avoids the Maillard reaction. Whole larvae also appear “inflated” rather than shrunken.
• Nutritional Drawbacks: It induces the greatest level of lipid oxidation among all drying methods, which can lead to quality loss. Additionally, it causes a roughly 10% reduction in protein solubility.
• Mitigation: Blanching the larvae before freeze-drying can reduce lipid oxidation by half.
3. Microwave-Drying
Microwave-drying is the fastest method, taking only 10 to 15 minutes.
• Digestibility Issues: This method induces protein polymerization, which results in a lower digestible amino acid score and overall lower digestibility compared to conventional oven-drying.
• Physical Traits: Like freeze-drying, it produces inflated whole larvae, but it allows for browning and results in a powder with larger particle sizes.
4. Sun-Drying
Sun-drying is a traditional, low-cost method characterized by low temperatures and extended processing times.
• Nutritional Stability: In controlled trials, sun-dried larvae showed proximate nutritional values broadly comparable to those dried in a low-temperature oven.
• Safety Risk: While cost-effective for smallholders, solar drying alone may sometimes be insufficient to fully reduce bacterial and yeast populations depending on environmental conditions.
5. Hot-Air Puffing
This method involves exposing larvae to very high heat (150°C) for a very short period (1–3 minutes).
• Concentration Effects: It achieves a very low residual moisture content (below 10%), which can cause slight shifts toward higher measured protein and lower measured fat due to concentration and analytical matrix effects.
Summary of Key Impacts
| Method | Speed | Impact on Protein | Impact on Lipids | Physical Appearance |
|---|---|---|---|---|
| Oven (60°C) | Moderate | Preserves solubility | Stable | Shrunken |
| Freeze-Drying | Very Slow | 10% solubility loss | High oxidation | Inflated, light color |
| Microwave | Very Fast | Lower digestibility | Stable | Inflated, brown |
| Sun-Drying | Very Slow | Comparable to oven | Stable | Shrunken |
20. Can BSF larvae be used in food for human consumption?
Yes, Black Soldier Fly (BSF) larvae can be used for human consumption, and they are already eaten in certain cultures, though they face significant legal, safety, and social hurdles in Western markets.
1. Current Consumption and Potential Applications
• Traditional Use: Humans have eaten BSFL in parts of Southeast Asia, such as Thailand and Vietnam. A notable example is the Kadazan-Dusun community in Sabah, Malaysia, who consume the larvae raw along with a fermented beverage called tapai.
• Modern Food Products: Research is actively investigating incorporating BSFL into familiar foods to improve acceptance. Examples include:
◦ Bakery and Snacks: Flour made from BSFL has been successfully used in cookies, tortilla chips, and snack bars (specifically in Indonesia to combat childhood malnutrition).
◦ Meat Substitutes: BSFL protein has been used to partially substitute pork lean meat in low-salt sausages, improving the flavor profile without compromising texture at levels up to 15%.
◦ Alternative Staples: BSFL meal has been used to fortify whole wheat bread, significantly increasing protein and lipid content while remaining sensorially acceptable to consumers at a 10% inclusion level.
2. Nutritional Profile
BSFL are a nutrient-dense food source, typically containing 35–53% crude protein and 15–41% lipids on a dry matter basis.
• Proteins: They have a complete essential amino acid profile that aligns well with WHO reference standards, being particularly rich in lysine.
• Fats: They are high in lauric acid, which has antimicrobial properties, though they generally contain more saturated fats and fewer polyunsaturated fats than other edible insects like crickets or mealworms.
• Minerals: BSFL are exceptional sources of calcium (comparable to milk), iron, and zinc.
3. Safety and Health Risks
Despite their potential, several safety concerns must be managed:
• Allergenic Potential: BSFL contain tropomyosin and arginine kinase, which can trigger allergic reactions in people already allergic to crustaceans (shrimp) or dust mites. In animal models, BSF protein triggered stronger allergic reactions than mealworms or silkworms.
• Contaminants: Because BSFL can be reared on organic waste, there is a risk of bioaccumulating heavy metals (especially cadmium and lead) or harboring pathogens. However, they do not seem to accumulate mycotoxins or pesticides.
• Need for Processing: Raw larvae can harbor high microbial loads; therefore, blanching or heat treatment is essential to ensure food safety.
4. Legal and Social Hurdles
• Regulatory Status: In the European Union, BSFL are classified as a “novel food” and are currently pending approval for human consumption. In the United States, while not explicitly banned, they must be produced specifically for human use under Current Good Manufacturing Practices (cGMP).
• Social Stigma: A major barrier is the “law of contagion”—the psychological taboo of eating an organism known to consume waste and manure. To overcome this, experts suggest using powdered or non-visible forms of the insect in food products.
21. What are the common challenges when scaling to industrial production?
Scaling Black Soldier Fly (BSF) bioconversion from laboratory or pilot settings to industrial production involves navigating a complex array of economic, technical, regulatory, and social hurdles.
1. Economic and Financial Constraints
• High Initial Capital: Industrial-scale facilities require significant investment in infrastructure, specialized rearing equipment, and automation technologies to ensure consistency.
• Operational Costs: Labor and substrate acquisition can account for up to 90% of total costs. Energy-intensive processes, such as the drying required for shelf stability or thermal steps for enzyme conditioning, further strain profitability.
• Market Competitiveness: To be viable, BSF products must compete in price and quality with established, highly efficient industries like fishmeal and soybean meal.
2. Technical and Logistical Challenges
• Heterogeneous Feedstocks: Maintaining consistent conversion efficiency and product quality is difficult when processing variable or seasonal waste streams rather than standardized laboratory diets.
• Climate and Process Control: Large-scale systems face challenges in precise climate control and space optimization. High larval densities generate significant metabolic heat, which can lead to lethal heat stress if not properly dissipated.
• Automation and Harvesting: Transitioning from manual handling to automated management of larval cycles and feedstock transportation is a major engineering barrier. Efficiently separating larvae from residues at scale remains an operational difficulty.
3. Regulatory and Institutional Barriers
• Substrate Restrictions: In many regions, including the European Union, rearing insects on “waste” such as manure, catering waste, or sewage sludge is prohibited for animals intended for human food chains, limiting the circularity of the process.
• Lack of Standardization: There is a notable absence of harmonized international safety and quality standards for insect-derived protein and frass fertilizer, which complicates trade and market entry.
• GMO Regulations: In the EU, genome-edited BSF strains (e.g., using CRISPR/Cas9) are classified as Genetically Modified Organisms (GMOs), subjecting them to extremely strict environmental and food safety assessments.
4. Social and Cultural Obstacles
• Social Stigma: Public perception often links BSF to filth and pathogens because they consume waste, leading to a “yucky factor” that hinders consumer acceptance.
• Environmental Nuisance: Large facilities can generate offensive odors and attract pests, leading to opposition from nearby communities if not managed through proper ventilation and hygiene.
• Religious and Halal Concerns: There is ongoing debate regarding the halal compliance of meat from animals fed on insects reared on feces or manure.
5. Risk Management and Safety
• Contaminant Accumulation: Ensuring that heavy metals (like cadmium and lead) or pharmaceutical residues from waste streams do not bioaccumulate to dangerous levels in the larval biomass is a persistent safety concern.
• Pathogen Transmission: While larvae can reduce many pathogens in the substrate, some may persist in the larval gut, necessitating reliable post-harvest decontamination steps like blanching or heat treatment.
22. How do you test if frass is safe for plants?
To test if Black Soldier Fly (BSF) frass is safe for plants, researchers and producers primarily use a Germination Index (GI) bioassay alongside chemical and microbiological analyses to identify potential phytotoxicity (toxicity to plants). Fresh or “raw” frass often contains high levels of ammonia and salts that can inhibit growth if applied before it is properly stabilized.
1. The Seed Germination Index (GI) Bioassay
The most common biological test for frass safety is the Germination Index, which evaluates both seed germination and root elongation.
• Procedure: A 10% (w/v) aqueous extract of the frass is prepared and used to hydrate sensitive indicator seeds, such as cabbage, radish, or cucumber.
• The Formula: The GI is calculated by multiplying the relative seed germination percentage by the relative primary root length (both compared to a distilled water control).
• Safety Thresholds:
◦ GI ≥ 80%: Generally indicates the frass is mature and has no phytotoxicity.
◦ GI < 50%: Indicates strong phytotoxicity; the material is unsafe for direct application.
◦ GI > 100%: Some stabilized frass reaches indices as high as 180%, meaning the extract not only lacks toxicity but actively promotes seedling vigor.
2. Chemical Indicators of Safety
Chemical analysis identifies specific components in the frass that may cause “nutrient burn” or osmotic stress.
• Ammonium Nitrogen (NH4+-N): High concentrations of ammonium in fresh frass are toxic to many plants. Safety increases as this nitrogen is converted into more stable organic forms or nitrates during stabilization.
• Electrical Conductivity (EC): This measures salinity. High EC (salinity) in frass derived from certain substrates (like restaurant waste) can induce osmotic stress. A decline in EC to below 2 mS/cm often indicates an improvement in fertilizer suitability.
• pH Levels: Fresh frass can be acidic, but stabilized, plant-safe frass typically exhibits a near-neutral to slightly alkaline pH (around 7.5 to 8.3).
• Dissolved Organic Carbon (DOC): High DOC in raw frass indicates unstable organic matter that is rapidly decomposing, which can deplete soil oxygen and hinder root respiration.
3. Microbiological and Contaminant Screening
Safety testing also extends to ensuring the frass does not introduce pathogens or toxins into the environment.
• Pathogen Testing: Regulatory standards (such as those in the EU) require frass to be tested for Salmonella (which must be absent in 25g) and Enterobacteriaceae (which must be below 1,000 cfu/g).
• Heavy Metal Analysis: Because BSFL can concentrate heavy metals from their feed, frass must be screened for arsenic, cadmium, lead, mercury, and chromium to prevent their accumulation in agricultural soils.
• Antibiotic Resistance Genes (ARGs): Recent research suggests monitoring frass for ARGs, particularly when larvae are reared on manure, to prevent their dissemination into the food chain.
4. Physical Maturity Signs
Beyond laboratory tests, the physical state of the frass provides clues to its safety.
• Texture and Stability: Safe, stabilized frass should be dry, friable, and relatively odorless.
• Stabilization Requirement: If frass does not meet safety criteria, it must undergo further treatment such as aerobic composting, curing, or heat treatment (e.g., 70°C for 60 minutes) to achieve biological stability and hygiene.
23. What are the legal requirements for using frass in agriculture?
Legal requirements for using Black Soldier Fly (BSF) frass in agriculture primarily focus on thermal treatment to ensure microbiological safety and nutrient composition standards for labeling, particularly within the European Union.
1. Mandatory Heat Treatment
In the European Union, insect frass intended for use as a fertilizer must undergo a specific stabilization process to eliminate pathogens.
• Standard Parameters: Under EU Regulation 142/2011, frass must be heat-treated at 70 °C for at least 60 minutes.
• Alternative Stabilization: Effective hygienization can also be achieved by maintaining the material at ≥55 °C for at least 72 hours with regular turning.
2. Microbiological Safety Criteria
Legal frameworks mandate strict limits on residual pathogens to prevent the dissemination of diseases into the soil and food chain:
• Salmonella spp.: Must be completely absent in a 25 g sample of the final product.
• Enterobacteriaceae: These must be reduced to levels below 10 cfu/g (some guidelines cite a limit of 1,000 cfu/g for hygiene verification).
• Clostridium perfringens: The treatment process must effectively reduce vegetative cells of this bacterium.
3. Labeling and Nutrient Thresholds
In the EU, frass is governed by Regulation (EU) 2019/1009 and is typically categorized as a solid organic fertilizer (PFC 1 (A) (I)). To be legally marketed as such, it must meet minimum nutrient thresholds:
• Individual Nutrients: The frass must contain at least 1% (by mass) of total nitrogen (Nt), phosphorus (P2O5), or potassium (K2O).
• Total Nutrient Content: The sum of these three primary nutrients must constitute at least 4% by mass.
4. Rearing Substrate Restrictions
The legality of the frass is often tied to what the larvae were fed. For instance, in the EU, larvae intended to produce frass for the commercial market must be reared on approved substrates, which currently exclude “catering waste” (restaurant/household food waste), manure, or sewage sludge for most agricultural applications. Using prohibited substrates restricts the resulting frass to research or experimental purposes rather than broad commercial use.
5. Institutional and Operational Standards
• Coordination: Producers may face hurdles related to a lack of coordination between headquarters and state-level regulatory authorities regarding documentation and certifications.
• Hygiene Management: Facilities are often required to implement HACCP (Hazard Analysis and Critical Control Points) and Good Manufacturing Practices (GMP) to ensure consistent safety and quality.
• Odour Control: While specific national guidelines may be lacking in some regions, producers are increasingly required to manage bad odours from rearing areas through regular inspections and stabilization before application.
24. How does composting frass improve its performance as a fertilizer?
Composting Black Soldier Fly (BSF) frass improves its performance as a fertilizer by transforming a raw, potentially toxic byproduct into a stable, mature, and nutrient-rich organic amendment. While fresh frass contains high levels of nutrients, it is often “incompletely stabilized” and can hinder plant growth if applied directly.
Composting improves frass performance through the following mechanisms:
1. Elimination of Phytotoxicity
Fresh BSF frass often exhibits strong phytotoxicity (toxicity to plants) that can inhibit seed germination and root elongation. Composting mitigates this by:
• Reducing Ammonia Nitrogen (NH4+-N): High concentrations of ammonia in raw frass can cause “nutrient burn”. Pilot-scale composting has been shown to decrease NH4+-N levels by nearly 65% through volatilization and microbial denitrification.
• Lowering Salinity: Composting can reduce the electrical conductivity (EC) of frass by over 46%, preventing osmotic stress in sensitive crops.
• Degrading Organic Acids: Labile organic matter, such as volatile fatty acids and phenolic compounds, is broken down during the thermophilic phase of composting, reaching levels that are safe for plants.
2. Enhanced Nutrient Stability and Slow-Release Dynamics
Composting transforms the nutrient profile of frass into a format better suited for long-term soil health:
• Humification: Composting promotes the formation of humic substances, converting unstable organic matter into stable humus. It increases the humic acid (HA) content, which enhances the fertilizer’s ability to complex heavy metals and improve soil fertility.
• Nitrogen Retention: The synergistic action between BSF intestinal microbes and exogenous composting microorganisms facilitates nitrogen retention, converting it into more stable, organically bound forms.
• Slow-Release Mechanism: Composted frass acts as a slow-release fertilizer, providing sustained nutrient availability during peak plant demand periods rather than an immediate, volatile burst.
3. Biological and Structural Soil Improvement
Composted frass provides benefits beyond basic chemistry by fostering a healthier soil ecosystem:
• Microbial Enrichment: Composting reshapes the microbial community, significantly increasing the abundance of beneficial, cellulose-degrading taxa such as the phylum Bacteroidota and families like Chitinophagaceae.
• Soil Structure: The application of composted frass can promote the formation of stable granular soil structures, which improves water retention, aeration, and permeability.
• Increased Vigor: Maturity tests (Germination Index) show that composted frass extracts can achieve values as high as 180%, meaning they don’t just lack toxicity but actively promote seedling vigor.
4. Superior Crop Response
Biological validation in pot experiments demonstrates that crops treated with composted frass consistently outperform those receiving raw materials. For instance, radish seedlings amended with composted wheat straw frass showed a significantly higher number of leaves and better overall growth compared to control groups. In solanaceous crops like tomato and chili, stabilized frass supported yields statistically comparable to—and sometimes exceeding—mineral NPK fertilizers.
25. How do consumer attitudes toward eating insects vary globally?
Consumer attitudes toward eating insects vary from deep-rooted cultural acceptance to significant psychological aversion, influenced by regional traditions, food processing methods, and demographic factors.
1. Traditional Entomophagous Cultures
In many parts of Asia, Africa, and the Americas, eating insects is a long-standing practice, with more than 1,900 to 2,000 species consumed globally.
• Southeast Asia: Traditions are well-established in countries like Thailand and Vietnam, where larvae are typically boiled, roasted, or fried. In Malaysia (Sabah), the Kadazan-Dusun community traditionally consumes BSF larvae raw alongside fermented beverages.
• Shifting Trends: Even in these regions, attitudes are changing. Younger generations in Malaysia often find the practice “disgusting or weird,” and entomophagy is gradually diminishing as urbanization increases. In China, purchase decisions are increasingly influenced by age and economic status rather than just tradition.
2. Western and Non-Traditional Markets
In Europe, the United States, and Australia, insects are generally viewed with taboo, disgust, or apprehension.
• Food Neophobia: Acceptance is largely hindered by “food neophobia,” a reluctance to try novel or foreign foods. For example, Italian consumers show strong resistance due to deep-seated cultural and culinary traditions.
• Motivations: Where interest does exist in Western markets, it is often driven by curiosity, sustainability concerns, or novelty. Dutch consumers, for instance, are more likely to be motivated by environmental benefits and animal welfare than by taste.
3. The Impact of Food Processing
Consumer acceptance improves significantly when insects are processed or disguised rather than served whole.
• Invisible Forms: Western consumers are much more willing to accept insects in powdered or extracted forms added to familiar products like cookies, bread, burgers, or protein bars.
• Hybrid Products: Consumers often prefer “hybrid” meat products that blend conventional livestock meat with insect protein over items that are composed entirely of insects.
4. Specific Challenges for Black Soldier Fly (BSF)
The BSF faces unique hurdles due to its role as a waste decomposer.
• The Law of Contagion: Many consumers apply the psychological “law of contagion,” perceiving the larvae as “filthy” because they consume manure and decaying matter. This stigma persists even if the larvae are reared on clean, food-grade substrates for human use.
• Feed vs. Food: BSF is currently perceived more favorably as animal feed (for poultry, fish, or pets) than as direct human food. For instance, Americans are generally more willing to consume meat from animals fed with insects than to eat the insects directly.
5. Demographic and Economic Influences
• Age and Gender: A study in Spain during the COVID-19 pandemic found that men were more than three times as likely as women to buy insect-protein products, with the 18–39 age group being the most accepting.
• Income Status: In some European contexts, individuals with lower income levels were found to be twice as likely to purchase products containing insect protein.
• Knowledge: Awareness plays a critical role; in many regions, the primary barrier to consumption is simply a lack of knowledge regarding the existence or benefits of insect-based foods.
26. How do larvae physically and chemically suppress pathogenic microbes?
Black soldier fly larvae (BSFL) suppress pathogenic microbes through a sophisticated combination of biochemical secretions and physical environmental modifications.
Chemical Suppression Mechanisms
• Antimicrobial Peptides (AMPs): The BSF genome contains a diverse repertoire of at least 57 AMP genes, including cecropins and defensins. Cecropins act by inducing lysis in the cell membranes of both Gram-negative and Gram-positive bacteria, while defensins primarily disrupt the membranes of Gram-positive bacteria by altering their electrostatic charge. Other identified AMPs, such as sarcotoxin and stomoxyn, have demonstrated antimicrobial activity against bacteria and filamentous fungi.
• Alkaline Gut and Ammonia Toxicity: BSFL maintain a highly alkaline gut environment, with pH levels reaching 8.5 to 9.0 in certain regions. This alkaline environment promotes the conversion of ammonium ions into uncharged ammonia (NH3), which is toxic to many pathogenic microorganisms and is a major driver of sanitization in substrates like manure.
• Bioactive Lipids: BSFL are naturally rich in lauric acid (C12:0), a medium-chain fatty acid that exhibits potent antimicrobial, antiviral, and antifungal properties. Lauric acid can enter bacterial cell membranes, causing structural damage and increasing permeability to other antibacterial compounds.
• Digestive Enzymes: The larvae produce a suite of potent digestive enzymes, including proteases and lysozymes, which assist in the degradation and inactivation of ingested bacteria.
Physical Suppression Mechanisms
• Direct Ingestion and Competition: BSFL are voracious feeders that physically compete with microbes by rapidly consuming vast quantities of organic matter. During this process, they directly ingest the indigenous microbes present in the waste, using them as a primary nutrient source.
• Substrate Aeration: The constant mechanical movement of the larvae through the waste aerates the substrate. This physical disruption shifts the environment from anaerobic to aerobic, effectively inhibiting methanogenesis and the growth of other anaerobic pathogenic microbes.
• Metabolic Heat Generation: In high-density rearing systems, the intense metabolic activity of the larvae generates significant heat, often raising substrate temperatures to over 45°C. This elevated temperature can enhance digestive processes while simultaneously suppressing heat-sensitive pathogenic bacteria.
• Interspecific Competition: BSFL naturally outcompete and repel other disease-vectoring insects, such as the house fly (Musca domestica). They produce chemical signals (allomones) and physically modify the habitat to inhibit house fly oviposition.
Microbiome-Mediated Suppression
The gut microbiota of the larvae, which includes beneficial genera like Bacillus and Lactobacillus, plays a critical role in pathogen suppression. These beneficial symbionts competitively exclude pathogens within the gut and may produce additional inhibitory molecules that contribute to the overall sanitization of the waste substrate.
While these mechanisms are highly effective at reducing the pathogenic load in the substrate—reporting reductions of 4 to 7 log₁₀ units for Salmonella and E. coli—some pathogens may persist within the larval gut, necessitating post-harvest decontamination such as blanching if the larvae are used for feed.
27. Does high-density rearing work well for all types of waste?
High-density rearing does not work well for all types of waste; its effectiveness is highly substrate-dependent, influenced by the nutritional profile and physical characteristics of the material. While high densities can improve waste reduction for some materials, they can hinder individual growth or lead to lethal conditions in others.
The Role of Substrate Quality
• Nutrient-Dense Waste (e.g., chicken feed, chowmein waste): In high-quality substrates, increased larval density typically leads to decreased individual prepupal weights. This is due to intense competition for limited space and nutrients, which increases metabolic stress. For these substrates, lower densities are generally better for maximizing the size of individual larvae.
• Fiber-Rich/Low-Quality Waste (e.g., vegetable waste, wheat straw): Conversely, higher densities can enhance growth and conversion efficiency on low-quality substrates. This is attributed to “social digestion” or aggregation effects, where the collective secretion of extracellular enzymes and increased microbial activity help break down complex polysaccharides like cellulose and lignin.
Performance Trade-offs
• Waste Reduction vs. Individual Yield: Higher densities generally result in greater total waste reduction because a larger population can process material more voraciously. However, while the total biomass yield may increase, the final weight of each individual larva often declines.
• Optimal Densities: Reported optimal densities range from 5 larvae/cm² for fiber-rich agricultural residues or mixed vegetable waste to 8–10 larvae/g of substrate for protein- and carbohydrate-rich food waste.
Risks of High-Density Rearing
• Lethal Heat Buildup: High densities generate significant metabolic heat, which can raise substrate temperatures above 45°C. While this can accelerate digestion, temperatures above 47°C are lethal and can cause mass mortality if the heat is not properly dissipated.
• Anaerobic Conditions: Overcrowding in high-moisture substrates (like fruit and vegetable scraps) can create sticky, anaerobic conditions. This buildup of unconsumed feed and excreta restricts larval movement and oxygen availability, often leading to increased mortality or larvae attempting to escape the system.
• Moisture Dynamics: High densities accelerate water evaporation due to increased larval activity and heat. If the substrate becomes too dry (below 60%), larval growth and the ability to consume organic matter are severely hindered.
Impact on Nutritional Quality
Larval density significantly influences the crude fat content of the larvae while having a more limited impact on protein levels. High densities combined with carbohydrate-rich substrates often promote lipid accumulation as a metabolic response, which may be desirable if the larvae are intended for biodiesel production rather than high-protein animal feed.
28. Does processing BSF into powder improve its social acceptance?
Processing black soldier fly (BSF) larvae into powder or non-visible forms significantly improves their social acceptance, particularly in Western cultures where whole insects are often met with disgust or apprehension.
The “Ideal Method” for Acceptance
• Disguising the Origin: Grinding BSF larvae into a powder or flour is considered the ideal method to introduce insects to consumers in regions without a tradition of entomophagy. This is because it successfully hides the insect’s appearance, which is a primary driver of food neophobia (the reluctance to try novel foods).
• Familiar Food Matrices: Consumers are much more likely to accept BSF when it is incorporated into familiar products like cookies, bread, sausages, or protein bars rather than being served as whole larvae.
Improving Visual and Sensory Appeal
• Color Optimization: Processing techniques such as blanching before drying result in a lighter brown color in the final powder by inactivating enzymatic browning.
• Defatting Benefits: Removing fat from BSF flours also contributes to a lighter, more desirable color and a lower browning index, which is more visually appealing in food products than dark brown unprocessed powders.
• Aroma Reduction: While dried whole BSF larvae can have a pungent, fishy aroma, milling them into powder and further processing them—such as through oil extraction—can help minimize unappetizing odors, making the ingredient more versatile for human food applications.
Overcoming Psychological Barriers
• The Law of Contagion: A major barrier to accepting BSF is its role as a waste decomposer. The psychological “law of contagion” leads people to view the larvae as “filthy,” but processing them into an “invisible” powder helps to mitigate this stigma.
• Gradual Normalization: Studies suggest that while direct consumption of whole insects remains a challenge, consumers are increasingly open to indirect consumption (eating animals fed with BSF) or eating them in highly processed “hybrid” meat products.
While processing into powder provides a viable technological path to improve acceptance, large-scale utilization still faces significant challenges in terms of overcoming deep-seated psychological barriers and navigating strict food regulations.
29. How do larvae physically aerate waste to inhibit anaerobic microbes?
Black soldier fly larvae (BSFL) physically aerate waste through their constant mechanical movement and burrowing, which transforms the substrate’s structure and suppresses anaerobic microbial activity.
Mechanism of Physical Aeration
• Constant Movement and Burrowing: BSFL are highly active and instinctively burrow into their feeding substrate to a depth of approximately 2 cm. This continuous mechanical action acts as a form of natural “turning,” which consistently introduces oxygen into the material.
• “Fountain” Feeding Behavior: Larvae often aggregate around food sources, feeding in a specialized “fountain” pattern where they flow in a cone-shaped or circular motion. This collective movement maximizes their contact with the waste and ensures the entire substrate is physically disrupted.
• Modification of Substrate Texture: As larvae feed and move, they disrupt the compact and rigid surfaces of the waste, transforming it into a rougher, more porous, and granular texture. By breaking up feed clumps and destroying the highly ordered tissue networks of the waste, they create channels that allow air to penetrate deeper into the substrate.
Inhibition of Anaerobic Microbes
• Suppression of Methanogenesis: The primary effect of this constant aeration is the inhibition of fermentative microbiological processes that occur in the absence of oxygen, most notably methanogenesis (the production of methane). By shifting the environment from anaerobic to aerobic, larvae significantly reduce the generation of methane gas.
• Enrichment of Aerobic Communities: The aerobic environment created by larval movement promotes the growth of beneficial, aerobic microbial communities while causing a decrease in the overall diversity of anaerobic taxa.
• Odor Mitigation: Aeration helps prevent the formation of anaerobic zones that produce foul-smelling volatile organic compounds, such as 2-methylpropanoic acid and various indoles commonly associated with manure and rotting waste.
Operational Considerations for Aeration
To maintain this natural aeration, producers must manage the bed depth of the substrate. Keeping the waste layer relatively shallow (typically between 5 cm and 12 cm) prevents the formation of localized anaerobic zones at the bottom that larval movement alone might not be able to oxygenate. Additionally, adding absorbent materials like bran or straw can help manage excess moisture that might otherwise lead to waterlogging and sticky, low-oxygen conditions.
30. What happens if a farmer uses unstabilized fresh frass?
If a farmer applies unstabilized fresh black soldier fly (BSF) frass to crops, it can lead to severe phytotoxicity, microbial contamination, and environmental nuisances. While frass is a valuable nutrient source, in its raw state, it contains high concentrations of substances that can be lethal or inhibitory to plants.
1. Immediate Phytotoxicity and Plant Damage
Fresh frass is often “incompletely stabilized,” meaning it contains reactive compounds that hinder plant development:
• Ammonia Toxicity: Raw frass frequently contains high levels of ammonia nitrogen (NH4+-N), which can induce osmotic stress and cause “nutrient burn,” leading to reduced seedling vigor and inhibited seed germination.
• High Salinity: The bioconversion process concentrates inorganic matter and ions, resulting in high electrical conductivity (EC). This high salinity can induce osmotic stress in crops, particularly salt-sensitive species, ultimately reducing yields.
• Inhibited Germination: Biological tests show that the Germination Index (GI) of fresh frass is often extremely low; a GI below 50% indicates the material is unsafe for direct application.
• Oxygen Depletion: Fresh frass contains high levels of dissolved organic carbon (DOC) and volatile organic acids that continue to decompose rapidly in the soil. This ongoing decomposition can deplete soil oxygen, impairing root respiration and producing toxic phenolic compounds.
2. Microbiological and Safety Risks
Raw frass may serve as a reservoir for harmful biological and chemical contaminants:
• Pathogen Persistence: While BSF larvae reduce the pathogenic load of waste, fresh frass can still harbor residual pathogens such as Salmonella spp., Escherichia coli, and various bacterial spores or helminth eggs.
• Antibiotic Resistance: If the larvae were reared on animal manure, the resulting unstabilized frass may contain antibiotic resistance genes (ARGs) that could be disseminated into agricultural soils.
• Heavy Metal Concentration: In certain substrates, heavy metals such as arsenic and chromium are primarily retained in the frass rather than the larvae, necessitating safety evaluations before soil application.
3. Environmental and Operational Nuisances
Applying unstabilized material creates practical challenges for the farmer and the community:
• Foul Odors: Fresh frass can emit offensive putrid odors caused by the ongoing decomposition of organic matter and anaerobic conditions.
• Pest Attraction: Raw residues can attract nuisance flies and other pests, posing a nuisance to residents and potentially spreading diseases if not managed through proper stabilization.
• Poor Handling: Fresh frass often has a high moisture content and can become sticky or viscous, making it difficult to distribute evenly in a field compared to dry, friable stabilized material.
To avoid these outcomes, frass should be stabilized through aerobic composting or curing until it reaches a germination index of ≥80%. Properly treated frass then functions as a slow-release nitrogen source that supports sustained nutrient availability without the risks of phytotoxicity.
31. How can larvae naturally repel disease-vectoring pests like house flies?
Black soldier fly larvae (BSFL) naturally repel disease-vectoring pests like the common house fly (Musca domestica) through a combination of chemical signaling, physical habitat modification, and intense resource competition. Research has shown that when BSFL are present in moderate to high abundances, house fly populations are significantly reduced or eliminated.
1. Chemical Inhibition of Oviposition
The primary mechanism by which BSFL repel house flies is through oviposition inhibition.
• Allomones: BSFL are believed to produce chemical signals known as allomones. These chemicals communicate to gravid (pregnant) female house flies that a specific habitat—such as manure—is already heavily infested with BSF larvae.
• Deterrence: Upon detecting these chemical cues, female house flies will avoid laying their eggs in that location. This natural repulsion is highly effective; one study reported house fly control rates between 94% and 100% in manure management systems using BSFL.
2. Interspecific Competition and Habitat Modification
BSFL outcompete house flies for both space and nutrients, effectively rendering the waste unsuitable for other fly species.
• Voracious Feeding: BSFL are voracious feeders that rapidly consume and process organic waste, leaving behind a residue (frass) that is less nutritionally available for house fly larvae.
• Physical Agitation: The constant mechanical movement and burrowing of BSFL physically disrupt the substrate. This physical presence makes it difficult for house fly larvae to establish themselves or survive if they share the same substrate.
• Environmental Shifts: BSFL activity alters the physical chemistry of the substrate, such as shifting the pH toward alkaline levels (often above pH 9.0) and generating significant metabolic heat. These conditions can be hostile to the development of house fly larvae.
3. Odor and Microbial Mitigation
BSFL significantly change the olfactory profile of the waste, which reduces its attractiveness as a breeding ground for pests.
• Odor Reduction: BSFL can reduce odorous volatile organic compounds (VOCs) and malodorous emissions from livestock manure by 87% or more. Since house flies are primarily attracted to the smell of decaying organic matter and manure, this reduction in odor makes the site less enticing for colonization.
• Microbial Control: BSFL modify the microflora of the waste and secrete antimicrobial peptides that suppress pathogenic bacteria like Salmonella and E. coli. By sanitizing the waste and altering its microbial diversity, they reduce the suitability of the “filth” environment that house flies require to thrive.
4. Factors Influencing Success
The effectiveness of this natural repulsion depends on several operational parameters:
• Larval Density: A dense population of BSFL is required to optimally repel house fly oviposition. Low BSF densities may allow house flies to successfully complete their development.
• Colonization Timing: BSFL are more effective at preventing house fly colonization if they are established first in the waste.
• Substrate Quality: While house flies can colonize various manures and human excrement, they are often outcompeted by the larger and more resilient BSFL, which can digest a much wider range of organic materials.
32. Which nutrients in BSF larvae shift most based on density?
Nutrient shifts in black soldier fly (BSF) larvae due to rearing density are primarily observed in crude fat (lipids) and ash content, while crude protein levels are often reported to be less sensitive or highly dependent on the specific type of waste being processed.
1. Crude Fat (Lipids)
Crude fat is the nutrient that shifts most consistently and significantly based on larval density.
• General Increase with Density: In most substrates, higher larval densities correlate with increased crude fat content. For example, in vegetable waste, fat content was significantly higher at elevated densities (5 and 7 larvae/cm²) compared to lower densities (1 and 3 larvae/cm²).
• Substrate Interaction: High densities combined with carbohydrate-rich substrates (such as food industry waste like chowmein) promote the most substantial lipid accumulation. This is often a metabolic response to dietary imbalances, where larvae synthesize fat de novo from excess carbohydrates.
• Nutritional Trade-off: There is often an inverse relationship between fat and protein accumulation; as density increases and stimulates fat storage, relative protein content may decrease due to “lipid dilution”.
2. Crude Protein
While some research suggests density has a limited effect on protein compared to fat, newer studies show that protein levels shift significantly when larvae are reared on specific waste types.
• Fibre-Rich Substrates: On vegetable waste, protein content follows the opposite trend of fat; it is highest at lower densities (e.g., 3 larvae/cm²) and declines as the population becomes more crowded.
• Food Industry Waste: On chowmein waste, protein content was initially low but increased slightly at the highest densities (7 larvae/cm²).
3. Ash (Mineral Content)
Larval density also has a significant, though sometimes non-linear, impact on ash content.
• Peak at Intermediate Densities: In some trials, ash content was lowest at extreme densities (1 and 7 larvae/cm²) and peaked at intermediate densities (5 larvae/cm²).
• Substrate Variance: Prepupae reared on vegetable waste generally exhibited higher ash content (up to 20%) compared to those on more nutrient-dense substrates like chowmein waste, regardless of density.
4. Moisture Content
Unlike fat and protein, the moisture content of the larvae themselves appears to be unaffected by rearing density, instead being driven almost entirely by the moisture levels of the feeding substrate.
Summary of Shifts
| Nutrient | Shift Pattern | Influencing Factor |
|---|---|---|
| Crude Fat | Typically increases with higher density. | Synergizes with carbohydrate-rich diets. |
| Crude Protein | Variable; often higher at low densities in fibrous waste. | Determined by substrate-density interaction. |
| Ash | Significant non-linear shifts. | High levels found in larvae fed vegetable waste. |
| Moisture | No significant shift based on density. | Driven by substrate water availability. |
Overall, while fat is the most dynamic nutrient across different stocking levels, the entire nutritional profile is determined by density-dependent responses tailored to the specific quality of the biowaste.
33. How do larvae modify the physical texture of fibrous waste?
Black soldier fly larvae (BSFL) modify the physical texture of fibrous waste through a combination of mechanical disruption and enzymatic action, transforming compact, rigid materials into a rougher, more porous, and granular state.
The specific mechanisms of this physical transformation include:
• Disruption of the Tissue Network: Larvae use their muscular activities and movement to physically break down and homogenize ingested feed mixtures. Scanning electron microscopy (SEM) reveals that biomass samples with regular, smooth surfaces before treatment are converted into a corrugated and porous texture. This process effectively destroys the highly ordered structural network of the fibers.
• Surface Morphology Alteration: On substrates like wheat straw and dairy manure, BSFL treatment significantly disrupts the surface structure of the fibers. This mechanical breakdown transforms the material from a compact state into a rougher texture.
• Increased Surface Area for Microbes: By breaking down and shredding fibrous tissue, larvae exponentially increase the surface area available for microbial colonization. These physical modifications create favorable conditions for both the larvae’s own gut enzymes and external bacteria to more effectively colonize and degrade recalcitrant organic matter.
• Reduction in Crystallinity: Larval activity has been shown to reduce the crystallinity of lignocellulosic fibers (such as wheat straw), making the structurally complex material less rigid and more susceptible to further biological decomposition.
• Aeration through Movement: The constant burrowing and mechanical movement of larvae act as a natural turning process that aerates the substrate, further disrupting its physical consistency and preventing the formation of dense, anaerobic clumps.
These physical alterations are a prerequisite for efficient bioconversion, as they lower the enzymatic burden on the larvae by making the structural components of the waste easier to ingest and assimilate.
What are the primary health and environmental benefits of using insect-based aquafeed?
The primary benefits of using insect-based aquafeed, specifically derived from Black Soldier Fly Larvae (BSFL), include significant improvements in environmental sustainability and the nutritional health of aquatic species.
1. Environmental Benefits
The environmental advantages are rooted in the ability of BSFL to function as bio-converters, aligning aquaculture with circular bioeconomy principles.
• Reduced Dependence on Marine Stocks: BSFL oil (BLO) and meal serve as strategically advantageous replacements for fish oil (FO) and fishmeal (FM), which are increasingly scarce and associated with overexploitation of marine ecosystems.
• Waste Valorization: BSFL can upcycle low-value organic waste—including food leftovers, agricultural by-products, and manure—into high-quality nutrient streams, effectively closing nutrient loops.
• Lower Greenhouse Gas (GHG) Emissions: Utilizing BSFL for organic waste management can lead to up to a 90% reduction in GHG emissions compared to conventional strategies.
• Resource Efficiency: Producing 100 kg of BSFL protein requires vastly less land and water than traditional protein sources like cattle or chickens, making it a highly efficient alternative.
• Green Extraction Technologies: Emerging patented cold-aqueous extraction methods for insect oil are designed to be low-energy and low-residue, further minimizing the environmental footprint of feed production.
2. Health and Nutritional Benefits
Research on various species, including zebrafish, salmon, and carp, indicates that insect-based feeds support or enhance biological performance.
• Superior Growth Performance: Studies have demonstrated that complete substitution of FO with BLO can yield pronounced somatic growth, significantly elevated specific growth rates, and more efficient feed conversion without compromising survival.
• Rich Nutrient Profile: BSFL biomass typically contains 35–45% crude protein and 15–35% lipids. It provides a complete essential amino acid profile, particularly rich in lysine and methionine, which are critical for neural development and immune function.
• Bioactive Properties of Lauric Acid: BSFL are naturally enriched with lauric acid (C12:0), a medium-chain fatty acid known for its potent antimicrobial, antiviral, and antifungal capacities. This can reduce the dependency on antibiotics in intensive production systems.
• Enhanced Immunity and Gut Health: Insect-based feeds have been shown to modulate gut microbiota balance, enhance pathogen resistance (e.g., against Vibrio alginolyticus), and improve intestinal health by increasing the activity of beneficial microbes.
• Oxidative Stability and Antioxidants: Optimized processing of BSFL can enhance antioxidant enzyme activity and reduce lipid oxidation in species such as European seabass and rainbow trout.
• Endogenous FA Synthesis: While insect oils are often lower in omega-3 fatty acids like DHA, certain freshwater species show a compensatory capacity to maintain tissue DHA levels through endogenous biosynthesis when fed BLO.
References
Al Anas, M., Mubarok, M. A., Adi, V. B., Aprianto, M. A., Hidayaturrohman, N., Sapan, Y., Fakhruddin, D., Abrar, A., & Atapattu, N. S. B. M. (2026). Optimizing broiler feather waste as a nitrogen-rich substrate to enhance black soldier fly larvae growth and nutrient composition. Journal of Agriculture and Food Research, 102681. https://doi.org/10.1016/j.jafr.2026.102681
Dallaire-Lamontagne, M., Saucier, L., Rivera Rodríguez, M. P., Koné, M., Gallant, F., Allard Prus, J. M., Lavoie, J., Vandenberg, G. W., & Deschamps, M. H. (2026). Valorisation of fermented hatchery residues through black soldier fly larvae (Hermetia illucens) bioconversion. Cleaner Waste Systems, 13. https://doi.org/10.1016/j.clwas.2026.100476
Dörper, A., Nicolai, K., Gort, G., de Jong, I. C., van Harn, J., Veldkamp, T., & Dicke, M. (2026). Feeding live or processed black soldier fly larvae to slow-growing broilers impacts their behaviour differently. Animal, 101748. https://doi.org/10.1016/j.animal.2025.101748
Dorper, A., Veldkamp, T., & Dicke, M. (2021). Use of black soldier fly and house fly in feed to promote sustainable poultry production. Journal of Insects as Food and Feed, 7(5), 761–780. https://doi.org/10.3920/JIFF2020.0064
Eskandari, K., Kargar, Z., Sanei, M. E., Hashamfirooz, M., Momeniha, F., & Akbarzadeh, K. (2026). The role of black soldier fly in food waste management and their environmental impacts: a systematic review. Sustainable Futures, 11. https://doi.org/10.1016/j.sftr.2026.101696
Ivanova, L., Dhaoui, N., Tukun, F.-L., Byhrø, E., & Kruse Fæste, C. (2026). Multi-mycotoxin method applicable for the safety assessment of waste-fed black soldier fly (Hermetia illuscens) larvae on the way to circular bioeconomics. Talanta, 302, 129432. https://doi.org/10.1016/j.talanta.2026.129432
Lee, D. J., Park, J., Kim, K. Y., Kim, J. Y., Koo, B., Park, K., & Kwon, E. E. (2026). Tracking carbon and nitrogen flow in black soldier fly larvae cultivation: Effects of feed carbohydrate-to-protein ratio. Journal of Environmental Management, 397. https://doi.org/10.1016/j.jenvman.2025.128357
Li, M., Quan, J., Liu, B., & Yuan, Z. (2026). Insight into insect protein production and gut microbiota from mixed food waste bioconversion by Black soldier fly (Hermetia illucens L.) larvae. Waste Management, 210. https://doi.org/10.1016/j.wasman.2025.115239
Oliveira, J., Guilgur, L. G., Assunção, R., Murta, D., & Trindade, A. (2026). Genomic Insights and Bioconversion Potential in the Black Soldier Fly (Hermetia illucens): Current Advances and Future Directions. Insects, 17(1), 70. https://doi.org/10.3390/insects17010070
Omar, E. M., Aly, M. Z. Y., Osman, K. S. M., & Tawfik, A. I. (2026). Compositional changes in preimaginal stages of black soldier fly (Hermetia illucens) reared on fermented water hyacinth with and without supplementation. European Food Research and Technology, 252(2). https://doi.org/10.1007/s00217-025-04979-5
Sharma, R. D., & Thangadurai, P. (2026). Process engineering and scale-up of an alkaline protease from Bacillus velezensis to enhance black soldier fly bioconversion. Bioresource Technology Reports, 33. https://doi.org/10.1016/j.biteb.2026.102599
Sharma, R. D., Velusamy, A., & Thangadurai, P. (2026). Single-enclosure black soldier fly system for decentralized conversion of mixed organic residues into feed-grade larvae and stabilized frass. Biocatalysis and Agricultural Biotechnology, 71. https://doi.org/10.1016/j.bcab.2025.103905
Shen, K., Liu, L., Niu, Y., Chen, Yue, Wang, F., Chen, Yuliang, Lin, L., Aye, S. L., Liu, Z., Wu, D., & Li, X. (2026). Food waste treatment by black soldier fly larvae (BSFL) and pilot-scale aerobic composting of the BSFL frass using horizontal mechanical reactor. Process Safety and Environmental Protection, 207. https://doi.org/10.1016/j.psep.2026.108413
Su, H., Zhang, B., Yang, R., Shi, J., He, S., Dai, S., Wu, D., & Zhao, Z. (2026). The Valorization of Food Waste into High-Value Biomass and Organic Fertilizers Through Bioconversion Using Black Soldier Fly Larvae (Hermetia illucens). Recycling, 11(1), 8. https://doi.org/10.3390/recycling11010008
Tian, Y., Zeng, X., Zheng, Y., Huang, Y., Wang, B., & Fu, X. (2026). Partial substitution of pork lean meat with black soldier fly larvae protein: Enhancing the quality and volatile flavor of low-salt sausages. Food Research International, 230. https://doi.org/10.1016/j.foodres.2026.118539
Ungureanu, N., & Vlăduț, N.-V. (2026). Black Soldier Fly (Hermetia illucens) Larvae and Frass: Sustainable Organic Waste Conversion, Circular Bioeconomy Benefits, and Nutritional Valorization. Agriculture, 16(3), 309. https://doi.org/10.3390/agriculture16030309
Wang, J., Liu, S., Zhang, N., Duan, Y., & Ma, Y. (2026). Bioconversion of restaurant food waste via black soldier fly larvae into a high-value protein source for improving meat quality in finishing pigs. Waste Management, 213. https://doi.org/10.1016/j.wasman.2026.115369
Wang, X., Wang, R., Wei, J., Hu, R., Wang, Z., Li, H., Chen, P., Qu, J., & Li, M. (2026). Conversion of wheat straw by black soldier fly larvae and fertilization effects of frass. Journal of Asia-Pacific Entomology, 29(1). https://doi.org/10.1016/j.aspen.2026.102527
Wu, Y. Y., & Lin, Y. Y. (2026). Mitigation of antibiotic resistance genes and pathogens via black soldier fly-mediated bioconversion of chicken manure. Science of the Total Environment, 1015. https://doi.org/10.1016/j.scitotenv.2026.181409
Yudistira, D. H., Sandi, Y. U., Wirabumi, B. A., Wikandari, P., Ramadhan, M. P., Rahmaningtyas, D. U., Fukushi, T., Gunawan, F. A., & Sato, S. (2026). Comparison of black soldier fly Hermetia illucens (Diptera: Stratiomyidae) frass to chemical and organic fertilizers on arthropod abundance and edamame plant performance. Applied Entomology and Zoology. https://doi.org/10.1007/s13355-026-00960-0






