1. Introduction: The Strategic Shift Toward Ecological Soil Management
As the agricultural industry faces the mandatory phase-out of traditional chemical fumigants like methyl bromide, there is a critical need for sustainable agriculture practices that provide robust soilborne pathogen control without environmental degradation. Anaerobic Soil Disinfestation (ASD), also known as reductive soil disinfestation, has emerged as a premier non-chemical disinfestation strategy. By leveraging biological and chemical shifts in the soil environment, ASD offers an ecological alternative that aligns with the growing demand for sustainable specialty crop production.

This guide provides a technical overview of ASD, detailing its multifaceted role in managing a wide array of fungi, bacteria, and nematodes—including highly resilient pathogens like Rhizoctonia solani and Plasmodiophora brassicae—across vegetable and ornamental systems. We will explore the specialized biological phases that define the process, the precision implementation framework necessary for field success, and the specific efficacy outcomes observed in recent research. By synthesizing results from diverse production environments—including high-value muck soils and soilless substrates—this document establishes a roadmap for integrating ASD into modern integrated pest management (IPM) programs.

Understanding the fundamental biological and chemical processes is the first step toward optimizing ASD for diverse agricultural landscapes.
ASD vs. Chemical Fumigation: Strategic Differentiators
| Feature | Anaerobic Soil Disinfestation (ASD) | Chemical Fumigation |
|---|---|---|
| Environmental Impact | Low; leverages biological fermentation and organic byproducts. | High; risks of environmental leaching and regulatory restrictions. |
| Microbiota Selectivity | High; shifts communities toward beneficial antagonists (e.g., Firmicutes). | Low; indiscriminately kills both beneficial and pathogenic organisms. |
| Nutrient Availability | Increases Phosphorus (P) and Potassium (K) solubility. | Negligible impact on P/K; disrupts natural nutrient cycling. |
| Nitrogen Impact | Significant drop in Nitrate nitrogen (NO3-N); risk of immobilization. | Minimal impact on Nitrogen forms compared to biological shifts. |
| Cost and Labor | High labor; ~$3,450/acre (Wheat Bran); provides biological priming. | Lower initial labor; no long-term biological “legacy” benefits. |
2. The Mechanism of Action: The Biological and Chemical Transformation of Soil
For a Sustainable Agriculture Consultant, understanding the underlying mechanisms of ASD is not merely an academic exercise; it is the prerequisite for optimizing field results. Because ASD relies on a complex interaction of environmental variables, failing to achieve the necessary threshold of “anaerobicity” can lead to inconsistent pathogen suppression. Precision in managing the transition from aerobic to anaerobic states—often monitored using IRIS (Indicator of Reduction in Soils) tubes—is what determines the efficacy of the treatment.
The biological transformation during ASD occurs in three distinct physiological phases, characterized by shifts in microbial dominance and the accumulation of specific metabolites:
| Phase Name | Dominant Microbial Groups (Aerobic/Anaerobic) | Key Metabolites/Chemical Changes |
|---|---|---|
| Phase 1: Aerobic/Facultative | Aerobic & Facultative: Firmicutes (Bacillus), Actinobacteria (Streptomyces), Proteobacteria (Pseudomonas). | Rapid consumption of O2, amino acids, and polysaccharides; O2 levels drop below 1%. |
| Phase 2: Facultative Transition | Facultatively Anaerobic: Dominance of Firmicutes (specifically Bacillus spp.) and Zygomycota. | Peak lactic acid production; accumulation of acetone, acetic acid, and 1-butanol begins. |
| Phase 3: Strict Anaerobic | Strictly Anaerobic: High abundance of Clostridium species and members of the Firmicutes phylum. | Production of toxic metabolites: acetate, butyrate, p-cresol, methyl sulphides, and volatile hydrocarbons. |
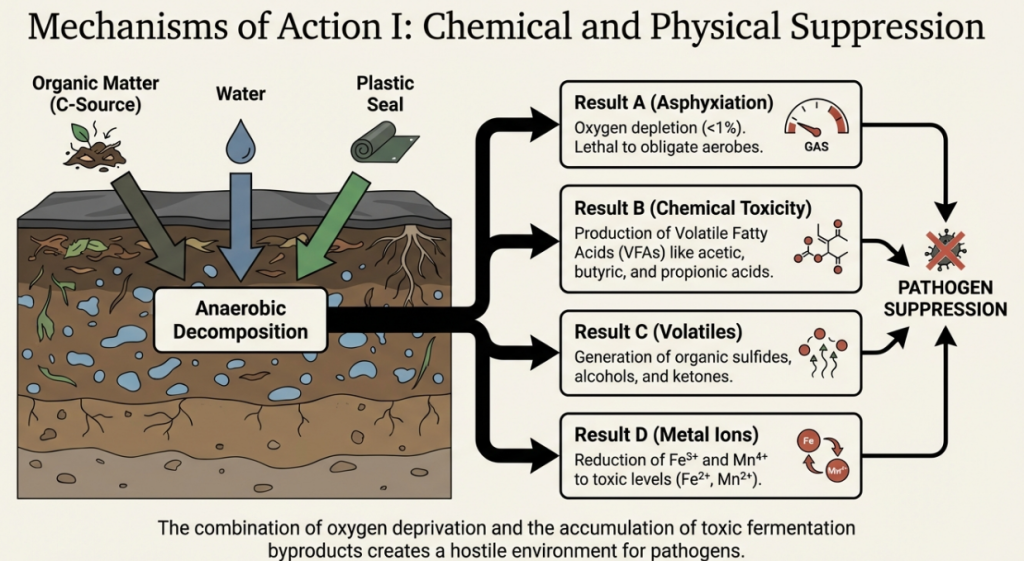
The “So What?” Layer: Creating a Lethal Environment
The efficacy of ASD is not derived from a single factor but from a “lethal cocktail” of conditions. As the soil redox potential (Eh) drops—reaching levels below -200 mV—the environment becomes hostile to specialized resistance structures. Pathogens that survive for decades via sclerotia (Stromatinia, Sclerotinia), chlamydospores (Fusarium, Phytophthora), or eggs and cysts (Meloidogyne) are neutralized through the combined pressure of oxygen depletion and the direct toxicity of volatile fatty acids and compounds like p-cresol. This multi-pronged attack ensures that even highly resilient propagules, such as root-knot nematode eggs, are significantly weakened or destroyed.
3. Implementation Protocol: A Five-Step Framework for ASD Success
The transition from theory to field application requires strict adherence to a precision framework. Because ASD is a biologically driven process, the quality of execution directly correlates with the intensity of the anaerobic conditions achieved.
1. Prepare the Soil: Clear the target area of all plant debris and ensure the surface is free of sharp objects. This is a critical preventive measure to ensure that the oxygen-impermeable tarps are not punctured during the process.
2. Incorporate Carbon Sources: Thoroughly mix organic matter—such as wheat bran, molasses, or soybean meal—into the soil profile. At this stage, install IRIS tubes to provide a visual confirmation of the reducing conditions during the treatment period.
3. Irrigate to Saturation: Apply water until the soil pores are filled to displace existing oxygen. This saturation creates the physical barrier necessary for anaerobic activity to commence and facilitates the movement of liquid amendments.
4. Cover with Plastic Mulch: Secure the area with oxygen-impermeable plastic tarps (Virtually Impermeable Film, VIF), ensuring the edges are buried deep. This prevents air exchange and maintains the reductive environment required for biocidal metabolite accumulation.
5. Incubate and Re-aerate: Leave the soil covered for 3 to 10 weeks, depending on soil temperature. After plastic removal, allow the soil to re-aerate for at least one week before planting to avoid phytotoxicity from accumulated organic acids.
The “So What?” Layer: Plastic Selection and Thermal Dynamics
The choice of plastic cover carries significant strategic implications. While dark plastics are the industry standard for ASD, transparent films can enable “biosolarization.” By capturing solar radiation, transparent plastics increase soil temperatures, which can synergistically accelerate pathogen mortality rates, particularly for oomycetes that typically require temperatures above 30°C for effective control.
3.1 The Ingredients of Transformation
To set the stage for this microbial alchemy, we must provide three physical inputs. These are the catalysts that shift the soil from a standard aerobic state into a powerful, pathogen-fighting anaerobic fortress.
The ASD Toolkit
| Component | Role in the Process | Examples from Research |
|---|---|---|
| Carbon Source | Provides the “fuel” for microbes; stimulates rapid respiration. | Wheat bran, molasses, ethanol, tomato pomace, and cover crops (rye or mustard). |
| Water | Displaces oxygen from soil pores to create a liquid environment. | Irrigation applied until the soil reaches total saturation. |
| Plastic Mulch | Acts as an oxygen-impermeable barrier to trap heat and gases. | Oxygen-impermeable tarps (VIF), black embossed mulch, or transparent films. |

3.2 Phase 1: The Aerobic Frenzy (Rapid Oxygen Depletion)
Within the first 24 to 48 hours after sealing the soil, a high-speed biological race begins. Because we have provided an abundance of carbon “food” and restricted the air supply, the indigenous aerobic microbes enter a feeding frenzy.
• Microbial Spotlight: This phase is dominated by aerobic and facultative bacterial phyla, particularly Firmicutes (specifically Bacillus species) and Proteobacteria (specifically Pseudomonas).
• The Action: Imagine these microbes gasping for the last breaths of oxygen as they rapidly consume available amino acids and polysaccharides.
This frenzy is critical because it creates the “biological vacuum”—a total exhaustion of oxygen—necessary for the next stage of specialists to take over.
As the last of the oxygen vanishes, the ecosystem reaches a tipping point where a new group of specialists takes the lead.
3.3 Phase 2: The Transitional Shift (The Rise of Fermentation)
Once the oxygen is depleted, the soil enters a transitional period. The strict oxygen-breathers go dormant, and the facultatively anaerobic microbes—those gifted with the ability to survive with or without air—begin to dominate the landscape.
• Chemical Insight: This phase is defined by the rise of fermentation. We observe a significant peak in lactic acid production, which correlates strongly with the continued proliferation of Bacillus species.
• The Baton Pass: As the environment shifts, we see the early accumulation of other metabolic byproducts like acetic acid and 1-butanol.
Think of this as a relay race: the aerobic microbes have finished their lap, and the fermenters are now building the chemical momentum needed for the most protective stage.
This transitional period builds the momentum needed to enter the final, most protective stage of soil disinfestation.
3.4 Phase 3: The Anaerobic Fortress (Organic Acid Production)
This is where the true alchemy happens. In this final phase, the soil becomes a highly reductive environment where strict anaerobes—organisms that cannot survive in oxygen—become the masters of the domain. Chief among these are the Clostridium species.
The Chemical Shield
• Organic Acids: The microbes produce high concentrations of acetate and butyrate. These lower the soil pH and serve as the first line of defense.
• Volatile Organic Compounds (VOCs): The soil accumulates powerful “natural fumigants,” including methyl sulfides (specifically dimethyl disulfide and trisulfide) and p-cresol.
• Indicator of Success: To prove these conditions are being met, researchers use IRIS tubes. These are coated with Goethite (a synthetic iron oxide). When the soil’s Redox potential (Eh) drops sufficiently, the orange Goethite dissolves, providing visual proof that the “anaerobic fortress” has been established.
These compounds are the weapons forged by the microbes themselves to protect the health of your future crop.
4. Carbon Source Optimization: Evaluating Material Efficacy and C: N Dynamics
The carbon source acts as the strategic driver of microbial respiration. Selecting a source requires balancing regional availability with biological stability and the post-treatment crop’s specific nutritional needs.
| Carbon Source | Relative Efficacy | Key Contextual Application |
|---|---|---|
| Wheat Bran | High (Industry Standard) | Widely used in the US ($3,450/acre); effective for Rhizoctonia and Meloidogyne. |
| Wheat Midds | High | Economical alternative ($2,180/acre) with similar performance to wheat bran. |
| Soybean Meal | High | High nitrogen content; effective for specialty flowers and oomycete suppression. |
| Ethanol | Very High | Preferred in Japan/Brazil; highly labile liquid source for rapid anaerobicity. |
| Tomato Pomace | Promising | Emerging regional byproduct (Midwest US); highly effective against R. solani. |
| Molasses | Moderate to High | Often used in liquid systems (C:N 81:1); may require combining with solid sources. |
The “So What?” Layer: C: N Ratios and Substrate Specificity
The C: N ratio of the amendment is a critical horticultural factor. While high C: N sources (e.g., molasses at 81:1) can lead to nitrogen immobilization, recent research into soilless substrates (Promix BX) reveals a different risk: ASD can significantly decrease NO3–N availability, necessitating precise post-treatment fertilization. Additionally, while mineral soils often see a pH drop during ASD, soilless media may experience a significant pH increase (from 5.6 to 6.8), which can alter nutrient solubility and disease dynamics for ornamental growers.

| Carbon Source | C:N Ratio | State | Pathogen Target Efficiency |
|---|---|---|---|
| Wheat Bran | 17:1 | Solid | High; remarkably effective against Rhizoctonia solani and Meloidogyne hapla. |
| Molasses | 81:1 | Liquid | Variable; highly effective against nematodes but can be inconsistent if used alone. |
| Ethanol | 100% Labile | Liquid | Exceptional; as a non-nitrogenous source, it penetrates deep layers to suppress Fusarium. |
| Tomato Pomace | ~9% Cellulose | Solid | High for Rhizoctonia; high cellulosic content specifically favors antagonistic microbes. |
| Soybean Meal | ~6% Cellulose | Solid | Moderate; high protein can produce toxic ammonia but risks nitrogen immobilization. |
The “So What?” of C: N Ratios The Carbon-to-Nitrogen (C: N) ratio is the governor of the “burn rate.” A high C: N ratio, such as Molasses (81:1), can lead to nitrogen immobilization, where microbes exhaust available nitrogen, potentially starving the subsequent crop. Conversely, low C: N ratios can drive rapid pH shifts. Selecting the right ratio ensures we disinfest the soil without compromising the nutrient availability for the next planting.
5. Pathosystem Efficacy: Comparative Analysis of Pathogen Suppression
ASD is not a universal cure; its success is pathogen-specific and influenced by inoculum density and environment.
High Efficacy Successes
• Meloidogyne hapla (Root-knot Nematode): Ohio field trials demonstrated that ASD with wheat bran or molasses reduced egg mass production by over 90%.
• Rhizoctonia solani: In specialty cut flower trials (zinnias), tomato pomace and wheat bran amendments reduced disease incidence to as low as 8.3% compared to 100% in controls.
• Plasmodiophora brassicae (Clubroot): While field results were less consistent due to soil mixing, bioassays showed significant reductions in clubroot severity for mustard greens following ASD with wheat bran-based amendments.

Technical Limitations and Failures
• Phytophthora drechsleri: ASD was notably ineffective against this oomycete in gerbera daisy trials when inoculum levels were high. In flooded conditions, the lack of oxygen actually increased plant susceptibility.
• Inoculum Thresholds: Success against P. drechsleri was only observed at low inoculum levels (3.4–6.8 kg/m³). At higher densities, ASD alone is insufficient without supplemental heat.
The “So What?” Layer: Inoculum and Ontogenic Resistance
The starting inoculum threshold often determines the outcome of ASD. For aggressive oomycetes, ASD should be viewed as a tool for maintaining low populations rather than eradicating high-density infestations. Furthermore, “ontogenic resistance” is a critical factor; gerbera daisies older than 6 weeks at transplant showed significantly slower disease progress, suggesting that ASD efficacy is maximized when integrated with strategic crop timing.

5.1 Success in ASD is not universal; it is a matching game between carbon, soil, and the specific target.
| Pathogen | Crop | Best Variable Combination | Control Result |
|---|---|---|---|
| Meloidogyne hapla | Lettuce | Wheat Bran or Molasses | Significant Reduction in galling and egg masses. |
| Rhizoctonia solani | Zinnia | Tomato Pomace or Wheat Bran | Inconsistent; bioassays showed a reduction, but field results varied significantly by year. |
| Phytophthora drechsleri | Gerbera | Any standard carbon source | Failure; motile zoospores utilized the water medium, while flooding stress weakened the host. |
| Plasmodiophora brassicae | Mustard Greens | Wheat Bran + Molasses | Inconsistent; bioassays showed reduction, but field results varied significantly by year. |
6. Strategic Challenges: Muck Soils, Soilless Substrates, and Economic Realities
Adopting ASD in commercial production requires addressing regional environmental and economic hurdles.

• Muck Soil Challenges (Histosols):
◦ Pesticide Binding: High organic matter (>20%) binds traditional pesticides, making ASD an attractive non-chemical alternative.
◦ Pathogen Mobility: Muck soils are low-lying and prone to flooding/wind erosion, which can rapidly reintroduce P. brassicae or Phytophthora propagules post-treatment.
◦ Infrastructure: A major barrier in muck systems (lettuce/greens) is the lack of existing plasticulture infrastructure (drip and tarping equipment) compared to strawberry systems.
The “So What?” Layer: Economic Trade-offs
The economic viability of ASD hinges on amendment costs. Using wheat bran at standard rates (3,450/acre) may be prohibitive for some growers. Transitioning to wheat midds (2,180/acre) or utilizing regional by-products such as tomato pomace is essential for commercial feasibility. Growers must weigh these costs against the treatment’s “legacy effect”.
7. Tarping Protocols and Environmental Monitoring
Gas-impermeable barriers are essential to maintaining the toxic metabolic environment required to neutralize resistant structures such as sclerotia, cysts, and nematodes.
Tarping Specifications
• Material: Use black embossed or transparent (VIF – Virtually Impermeable Film) plastic mulch.
• Sealing: All edges must be buried deep to prevent air exchange. Any puncture or seal failure will allow oxygen intake, neutralizing the Phase 3 strict anaerobes.
Induction Monitoring: IRIS Tubes
Verify the reductive state using IRIS (Indicator of Reduction in Soils) Tubes. The primary metric for success is the intensity of the red-to-gray/clear transition of the iron oxide paint. While effective wheat bran treatments generally show high paint loss, variability is common; the visual stripping of the paint serves as confirmation that a sufficiently reductive (anaerobic) state has been reached.
Operational Parameters for Incubation
• Duration: 3 to 10 weeks. Standardize at 4 weeks for temperate environments (25°C).
• Temperature Thresholds: Efficacy is highly temperature-dependent, ideally between 16°C and 30°C+.
CRITICAL WARNING: At standard temperatures (approx. 25°C), ASD is ineffective against Phytophthora drechsleri. In some cases, these conditions actually enhance disease progression compared to aerobic controls. Effective control of P. drechsleri requires soil temperatures reaching approximately 40°C.
Termination of the treatment must be handled with care to protect subsequent crop health.
7.1 The Environment (Soil Type and Temperature)
The physical medium dictates how well the “anaerobic state” is maintained and how pathogens react to the treatment.
• Muck Soils (Histosols): Containing >20% organic matter, these soils are light and prone to wind erosion. Their high organic matter can bind chemical pesticides, but in ASD, it provides a secondary carbon reservoir. However, muck soils are prone to flooding, which can reintroduce pathogens like Plasmodiophora brassicae post-treatment.
• Soilless Substrates: Often used in specialty flowers (e.g., peat-based), these have lower microbial complexity. Researchers must monitor for significant pH spikes when adding carbon like wheat bran, as there is less “biological buffering” than in mineral soils.
The Role of Temperature: The Catalyst Temperature is the accelerator for ASD. While 16°C is the minimum “floor” for efficacy, the standard 3-to-6-week window is predicated on 25°C. At “biosolarization” levels (40°C), the timeframe for lethal metabolite accumulation is significantly compressed, potentially allowing for faster crop turnover and higher efficiency against aggressive oomycetes.
Environmental Control Checklist for Researchers
1. Monitor Temperature: Aim for at least 25°C; 40°C combinations allow for a shorter “clock.”
2. Verify Saturation: All soil pores must be water-filled to prevent aerobic “oxygen pockets.”
3. Secure Tarps: In light muck soils, bury mulch edges deeply to prevent wind-driven air exchange.
4. Post-Treatment Fertility Check: Test for soluble salts and nitrogen availability before the next crop.
Once the environment is set, the biological battle follows a predictable temporal sequence.
7.2 The Clock (Incubation and Tarping Period)
The standard incubation window ranges from 3 to 10 weeks. Shortening this window too much risks re-aerating the soil before the pathogens are dead.
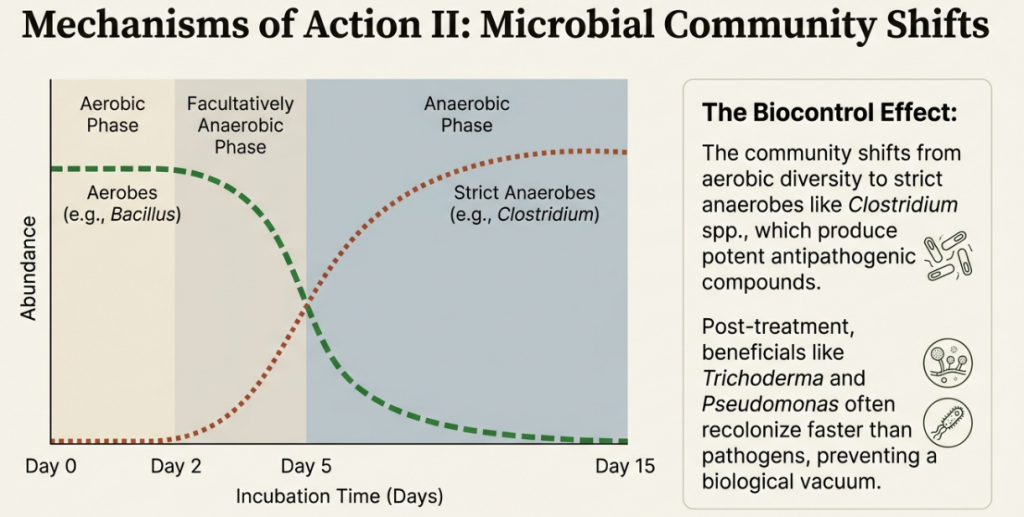
The Three Physiological Phases of the Soil Microbiome (15-Day Interaction)
1. Aerobic Phase (Day 0–3): Rapid O2 consumption. Aerobic bacteria like Pseudomonas dominate as they feast on sugars and amino acids.
2. Facultatively Anaerobic Phase (Day 3–7): Oxygen drops below 1%. Lactic acid levels peak, directly correlating with a massive proliferation of Bacillus species.
3. Anaerobic Phase (Day 7–15+): Strict anaerobes like Clostridium take over. This is the peak of the “kill zone,” where the soil is flooded with butyrate, acetate, and methyl sulphides.
If the tarp is lifted prematurely, we lose the “Phase 3” chemistry that serves as the final blow to resilient pathogen structures. But how do we know the “magic” is happening if we can’t see beneath the tarp? We look for the chemical signatures of a battle won.

8. Post-Treatment Verification and Crop Integration
The “Legacy Effects” of ASD include the soil microbiome’s transition back to aerobic stability, characterized by a surge in beneficial microorganisms and an increase in plant-available Phosphorus.
Termination and Aeration Protocol
Upon tarp removal, a mandatory 5-to-7-day aeration period is required. This allows for the dissipation of toxic organic acids (acetic and butyric acid) and volatile organic compounds (VOCs) that would otherwise cause phytotoxic “seedling burn.”
Crop-Specific Outcomes and Integration
• Lettuce and Mustard Greens: High efficacy against Root-Knot Nematode (M. hapla) and consistent (though occasionally variable) reductions in Clubroot (P. brassicae).
• Specialty Cut Flowers (Zinnia/Gerbera): ASD provides excellent control of Rhizoctonia solani. For Phytophthora management, transplanting older, more resilient seedlings (6–8 weeks old) is recommended, as they demonstrate significant ontogenic resistance compared to 4-week-old seedlings.
Nutrient Risks and Verification
ASD drastically alters nitrogen chemistry. Managers must expect a drastic decrease in NO3-N following carbon incorporation and anaerobicity. Post-ASD soil testing is mandatory to check for nitrogen stabilization and phosphorus solubility before final fertilization adjustments and planting.
Anaerobic Soil Disinfestation, when implemented with rigorous adherence to carbon ratios and monitoring protocols, serves as the sustainable cornerstone of an Integrated Pest Management (IPM) system for modern commercial operations.

8.1 Verification Variables: Measuring What We Cannot See
To confirm that the soil has reached the necessary “reductive” state, researchers utilize IRIS Tubes (Indicator of Reduction in Soils). These 30-cm PVC pipes are coated with iron oxide paint.
• The Grid Method: Paint loss is quantified using a visual grid method. Researchers evaluate both the upper and lower halves of the 30-cm tube to ensure that reducing conditions are uniform throughout the soil profile, not just at the surface.
• Redox vs. pH: While pH tracking is common, it is an inconsistent indicator, especially in muck soils where pH may not drop despite successful disinfestation. Iron oxide paint loss is the gold standard; if the paint is gone, the soil has become sufficiently reductive to kill pathogens.
9. Conclusion: The Future of ASD in Integrated Pest Management
Anaerobic Soil Disinfestation represents a significant advancement in soil health management, offering a path to restore biological balance while reducing chemical reliance. By effectively suppressing pathogens like root-knot nematodes and Rhizoctonia, ASD provides a viable alternative for high-value specialty crops.

9.1 Strategic Summary
Match the Fuel to the Pathogen. High-cellulose sources (tomato pomace) are your best weapon against fungi like Rhizoctonia, while labile sources (ethanol) are superior for deep-seated nematodes.
The Environment is the Governor Never ignore the soil type. Muck soils require deeper tarp edges, while soilless substrates require a watchful eye on pH and nitrogen levels post-treatment.
Measurement is the Only Truth. A sealed tarp is not a guarantee of success. Always use IRIS tubes to verify the reduction grid before concluding your trial.
Future Directions The next frontier of “translational” ASD involves investigating Cover Crops as “trap crops” to lure pathogens to the surface before the tarp is laid. Economically, we are searching for lower-cost carbon sources like wheat midds. Most importantly, the industry is transitioning to Bioplastics to replace VIF (Virtually Impermeable Film), ensuring that the ecological promise of ASD is not undermined by plastic waste.
Actionable Insight: The true value of ASD lies in the re-establishment of beneficial microbiota. Post-treatment soils are enriched with pathogen antagonists (Trichoderma) and microorganisms that trigger N2 fixation and sulfur cycling pathways. This legacy effect not only suppresses disease but also enhances plant vigor and nutrient availability throughout the production cycle.
Image Summary






Questions/Answers
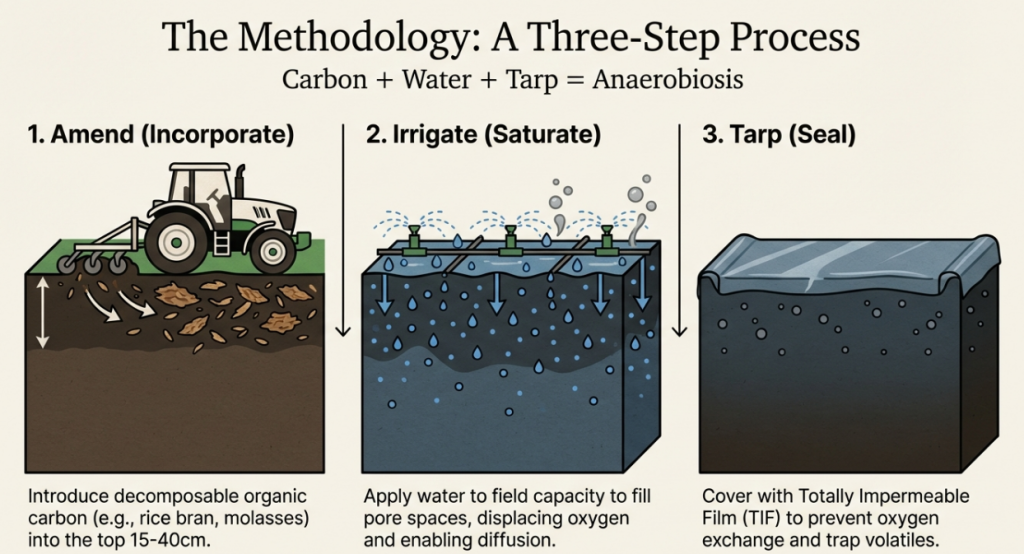
1. What are the three essential steps for performing ASD?
Anaerobic soil disinfestation (ASD) is a three-step process designed to create an oxygen-depleted environment to manage soilborne pathogens, pests, and weeds. The three essential steps are as follows:
1. Soil Amendment (Incorporating a Carbon Source): The first step involves incorporating easily decomposable, carbon-rich organic materials into the soil. These materials, such as wheat bran, molasses, or cover crops, provide the energy source for beneficial soil microorganisms. The carbon source should be spread evenly and typically incorporated to a depth of 15 to 20 cm (6 to 8 inches).
2. Soil Irrigation (Saturation): The second step is to irrigate the soil to saturation or field capacity. This fills the soil pores with water, which displaces and reduces the available oxygen in the soil, creating the initial conditions necessary for anaerobiosis.
3. Soil Tarping (Covering): The final step is to tarp the treated area with an airtight, gas-impermeable plastic mulch or film to prevent oxygen from being resupplied to the soil. Suitable covers include thick polyethylene or Virtually Impermeable Film (VIF). The soil generally remains covered for a period of 3 to 10 weeks, during which the lack of oxygen and the production of toxic organic acids and volatile compounds work to kill pathogens.
Once the treatment period is complete, the tarp is removed or punctured, and planting is typically delayed for 5 to 7 days to allow the soil to re-aerate and dry.
2. How do different carbon sources affect pathogen suppression results?
The selection of a carbon source is a critical determinant of the success of Anaerobic Soil Disinfestation (ASD), as it acts as the fuel for microbial respiration, which depletes oxygen and triggers the release of toxic metabolic byproducts. Different carbon sources influence pathogen suppression through their physical state, their specific chemical composition, and the unique spectrum of volatile compounds they produce during decomposition.
Liquid vs. Solid Carbon Sources
The physical state of the carbon amendment significantly impacts the depth and uniformity of disinfestation:
• Solid Materials (e.g., cereal brans, cover crops): These are typically tilled into the soil, but their suppressive effect is often limited to the depth of incorporation, usually approximately 20–30 cm.
• Liquid Materials (e.g., molasses, ethanol): These can be applied through irrigation systems, allowing them to penetrate deeper and more uniformly into the soil profile. Ethanol, in particular, is noted for being a pure substance with a stable, uniform composition that allows for precise application.
Efficacy Against Specific Pathogens
Pathogen suppression results vary widely based on the specific carbon source used:
• Cereal Brans (Wheat and Rice): Wheat bran has proven effective in reducing the incidence of spinach wilt and bacterial wilt of tomato caused by F. oxysporum and R. solanacearum, respectively. Rice bran is a popular choice for managing Verticillium dahliae in strawberry fields, often achieving 85–100% reduction in microsclerotia.
• Molasses: In Florida, molasses (often combined with composted broiler litter) provided excellent control of Phytophthora capsici, F. oxysporum, and Macrophomina phaseolina. However, research in California found molasses alone was less effective than rice bran for strawberry production and failed to induce necessary shifts in the soil microbial community.
• Ethanol: This source is often more effective than solid cereal brans; for instance, it required only 3 to 9 days to reach undetectable levels of F. oxysporum in lab settings, compared to 9 days for wheat bran.
• Cover Crops and Green Manures: The results for cover crops are variable. For example, broccoli and perennial ryegrass were found to effectively inactivate F. oxysporum, R. solani, and V. dahliae. However, crops like pearl millet were less effective than molasses at suppressing F. oxysporum.
• Specialized Industrial Byproducts: Tomato pomace and wheat bran demonstrated the potential to control Rhizoctonia stem rot in both soilless and soil-based substrates, while soybean meal was less effective.
Mechanisms of Variation
The differences in suppression results are driven by several factors related to the carbon source:
• Volatile Organic Compounds (VOCs): Each carbon source produces a unique spectrum of volatile compounds (such as alcohols, organic sulfides, and esters) and organic acids (primarily acetic and butyric acids). For instance, while some rice bran volatiles inhibit Pythium ultimum, they may have no activity against F. oxysporum.
• C:N Ratio: While the specific type of carbon is often more important for generating anaerobicity than the exact Carbon-to-Nitrogen (C:N) ratio, the ratio affects nutrient availability. Sources with low C:N ratios can lead to high ammonium levels, which may be toxic to some pathogens, while high C:N ratios (like molasses) may cause nitrogen immobilization, requiring increased post-treatment fertilization.
• Temperature Interaction: The effectiveness of a carbon source is often temperature-dependent. For example, lower carbon amendment rates may fail to decrease pathogen viability if soil temperatures are moderate or low (15–25 °C), whereas higher rates or more labile sources might compensate for cooler conditions.
3. How does ASD change soil chemistry and microbial communities?
Anaerobic Soil Disinfestation (ASD) induces significant biological and physiochemical shifts in the soil by creating an oxygen-depleted environment through carbon amendment, irrigation, and airtight tarping. This process stimulates a cascade of chemical reactions and a dramatic restructuring of the soil microbiome.
Changes in Soil Chemistry
The primary chemical change during ASD is a rapid reduction in oxidation-reduction potential (redox potential, Eh). As soil microorganisms consume the added carbon, they quickly deplete available oxygen, forcing a shift to anaerobic respiration. This leads to the consumption of other electron acceptors in the soil matrix, such as nitrate (NO3−), manganese (MnO2), ferric iron (Fe3+), and sulfate (SO42−).
Key chemical modifications include:
• Production of Volatile Fatty Acids (VFAs): Fermentation of the organic material releases short-chain organic acids, primarily acetic, butyric, propionic, and lactic acids. In their non-ionized forms, these acids are highly toxic to many soilborne pathogens and pests.
• Soil pH Fluctuations: ASD typically decreases soil pH in many systems due to the accumulation of organic acids and dissolved carbon dioxide. However, in naturally acidic soils, pH may actually increase as reduction processes consume protons or as the decomposition of organic amendments releases basic cations.
• Nitrogen Cycling: ASD causes substantial changes to nitrogen status. Nitrate (NO3−) is often rapidly depleted through leaching, denitrification, or dissimilatory nitrate reduction to ammonium (DNRA). Concurrently, levels of ammonium (NH4+) typically increase.
• Release of Metal Ions and Gases: Strong reducing conditions lead to the accumulation of ferrous iron (Fe2+) and manganese (Mn2+) ions in the soil solution. Additionally, toxic gases such as ammonia (NH3), hydrogen sulfide (H2S), methane (CH4), and ethylene are produced during anaerobic decomposition.
• Nutrient Availability: ASD can enhance the availability of phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg), partly because many amendments (like molasses) are rich in these elements and partly because reduction processes can increase P solubility in acidic soils.
Changes in Microbial Communities
ASD dramatically induces changes in the soil microbial community composition by removing niches for obligate aerobes and favoring those that thrive in low-Eh environments.
The microbial shift occurs in distinct phases:
1. Aerobic Phase: Initially, the addition of labile carbon stimulates the rapid growth of resident aerobic and facultative microbes, such as members of Bacillus, Streptomyces, and Pseudomonas.
2. Anaerobic Phase: As oxygen is depleted, the community structure shifts to dominance by facultative and obligate anaerobes. The most dramatic change is a significant increase in the Firmicutes phylum, specifically members of the Clostridia and Bacilli classes. Some Clostridium species contribute to pathogen control by producing chitinase to degrade fungal cell walls.
3. Persistence and Recovery: While microbial diversity often decreases during the anaerobic phase due to reduced niche space, it can foster a “disease-suppressive” environment. After the plastic is removed and oxygen returns, aerobic communities slowly return to pre-treatment levels, but some treatment-specific shifts—such as increased populations of the beneficial fungus Trichoderma—can persist through the growing season.
The specific carbon source used plays a major role in shaping these communities; for example, rice bran may increase the abundance of Acidobacteria and Burkholderia, while alfalfa meal favors different genera like Flavisolibacter and Rhodanobacter. Ultimately, ASD does not create a biological vacuum; instead, it replaces a pathogen-conducive community with a more diverse, suppressive microbiome that can enhance long-term system resistance to re-infestation.
4. Can ASD be combined with solarization for better pest control?
Yes, Anaerobic Soil Disinfestation (ASD) can be combined with soil solarization, a practice often referred to as “biosolarization” or “biodisinfection”. Combining these two methods can improve the overall efficacy of pest control and overcome the specific limitations of each treatment when applied individually.
The benefits and mechanisms of combining ASD with solarization include:
Enhanced Soil Temperatures
While standard ASD does not strictly require high temperatures, the addition of a solarization component significantly boosts its effectiveness.
• Clear Plastic Efficacy: Using clear, UV-stabilized plastic film facilitates solarization, which can elevate soil temperatures by up to 10°C more than opaque (black or green) plastic.
• Synergistic Inactivation: Pathogen suppression is often temperature-dependent; for example, Verticillium dahliae populations were found to be significantly lower under clear plastic compared to opaque plastic.
• Treatment Duration: In high-temperature environments (above 30°C), the required duration for ASD to suppress certain pathogens like Fusarium oxysporum may be reduced to as little as 14 days.
Improved Management of Specific Pests
The combination has proven particularly effective against hardy pathogens and pests:
• Nematodes: Solarization combined with an added carbon source (ASD) resulted in significantly lower rates of root galling from root-knot nematodes (Meloidogyne incognita) than solarization alone.
• Fungal Pathogens: In Florida field studies, ASD combined with summer solarization achieved mortality rates for Fusarium oxysporum and Phytophthora capsici that were equivalent to traditional methyl bromide soil fumigation.
• Weeds: While ASD alone often provides limited weed control, the combination with solarization creates a synergistic effect that better manages difficult species. For example, the use of composted broiler litter with solarization was more effective at controlling nutsedge (Cyperus spp.) than anaerobic conditions alone.
Nutrient Dynamics
Combining these methods can also impact soil chemistry. The use of compost or organic amendments with solarization has been shown to significantly increase soil inorganic nitrogen concentrations and enhance the accumulation of biopesticidal organic acids.
Practical Considerations
The decision to combine these treatments is often dictated by environment and timing. In warm regions like Florida or California, solarization can be performed for most of the year, whereas in cooler regions like the Netherlands, it is limited to summer months. In some commercial systems, growers use clear plastic to facilitate solarization and then cover it with opaque plastic at the end of the ASD period to provide the cooler soil temperatures needed for crop production.
5. How long should the plastic tarp remain on the soil?
The duration for which the plastic tarp should remain on the soil during Anaerobic Soil Disinfestation (ASD) typically ranges from 3 to 10 weeks, depending on the target pathogens, soil temperature, and carbon source used. While some sources cite a standard treatment range of 3 to 6 weeks, the specific duration is often tailored to environmental conditions.
Key factors influencing the tarping duration include:
Target Pathogens and Pests
• Fungi, Bacteria, and Oomycetes: A meta-analysis of 222 studies found that a 3-week incubation period was sufficient to suppress these populations by approximately 64%.
• Nematodes: Managing plant-parasitic nematodes generally requires a longer duration; incubation periods of 4 to 6 weeks have proven most effective, achieving over 90% suppression.
• Persistent Pathogens: In specific cases, such as managing Verticillium dahliae in tree nurseries or using certain carbon sources like fresh broccoli, treatment times have been extended to 10 to 15 weeks.
Temperature and Minimum Requirements
• High Temperatures: ASD is more effective and can be performed faster in warmer conditions. If soil temperatures are consistently above 85°F (29.4°C), a 3-week tarping period is often effective for most pathogens. In Japan, researchers found that just 14 days were required to suppress Fusarium oxysporum when soil temperatures exceeded 30°C.
• Minimum Threshold: Regardless of temperature, the soil should generally remain covered for at least 15 days to ensure the adequate generation and accumulation of antimicrobial compounds like organic acids and methyl sulfides.
• Regional Standards: In regions with temperate climates like Ohio, a 4-week tarping period has been used successfully for high tunnel and field production.
Termination and Planting
Once the treatment period is complete, the plastic is either removed or punctured. Planting should typically be delayed for 5 to 7 days after the tarp is removed or breached to allow the soil to re-aerate, dry, and breathe.
6. How do liquid and solid carbon sources differ in depth?
In anaerobic soil disinfestation (ASD), the physical state of the carbon source—liquid or solid—is a primary factor in determining the depth and uniformity of disinfestation throughout the soil profile.
Solid Carbon Sources
Solid amendments, such as cereal brans (rice or wheat), mustard meal, and cover crops, are typically incorporated into the soil through mechanical tilling or rotovating.
• Restricted Depth: The suppressive effect of solid materials is generally limited to the depth of physical incorporation, which is typically the upper 15 to 20 cm (6 to 8 inches). Depending on the equipment used, this may extend up to 30 or 40 cm.
• Management Challenges: Because many plant-parasitic nematodes can migrate into the crop root zone from depths greater than 1.5 meters, the relatively shallow incorporation of solid amendments may be insufficient to provide commercially viable control in certain systems, such as tree-crop nurseries.
• Uniformity: Solid amendments can sometimes suffer from a lack of uniformity in distribution during field application, which can lead to inconsistent pathogen suppression.
Liquid Carbon Sources
Liquid amendments, such as ethanol and molasses, offer distinct advantages regarding soil penetration because they are frequently applied through irrigation systems.
• Deeper Penetration: Because they are dissolved in or diluted with irrigation water, liquid sources can seep deeper and more uniformly into the soil profile than solid materials.
• Buried Delivery: Delivering soluble carbon sources through buried irrigation lines has been proposed as a method to generate anaerobicity at significantly greater depths than traditional surface tilling.
• Application Efficacy: In laboratory settings, liquid ethanol has reached undetectable levels of certain pathogens like Fusarium oxysporum in as little as 3 to 9 days, whereas solid wheat bran required a minimum of 9 days.
Depth and Pathogen Control
Research indicates that the depth of the soil profile significantly influences the overall effectiveness of the ASD treatment.
• Deep vs. Shallow: Studies have shown that nematode populations can be suppressed by 82% in deep soil profiles and 70% in shallow profiles.
• Pathogen Specificity: While a depth of 15–20 cm is often sufficient to manage many bacterial and fungal pathogens, it is frequently noted as a limitation for nematode management, as these pests move more freely through the soil matrix.
• Observed Limits: In preliminary field trials, consistent pathogen suppression has been found down to 46 cm, with more limited suppression observed as deep as 76 cm.
7. Why is ethanol considered more effective than solid cereal brans?
Ethanol is often regarded as more effective than solid cereal brans due to its superior depth of penetration and uniformity within the soil profile. While the suppressive effects of solid cereal brans are typically limited to the depth of mechanical incorporation (approximately 20–30 cm), liquid ethanol applied via irrigation systems can seep deeper and more evenly throughout the treated area. Ethanol also demonstrates faster results in pathogen inactivation; for example, in laboratory settings, it reduced Fusarium oxysporum to undetectable levels in as little as 3 to 9 days, whereas solid wheat bran required at least 9 days.
Chemically, ethanol is a pure substance with a uniform and stable composition, which provides more consistent results than cereal brans that can suffer from uneven distribution or variable chemical makeup. Furthermore, ethanol is highly advantageous for nutrient management because it lacks nitrogen. This prevents the potential issues of excessive nitrogen addition or immobilization that can occur when using high rates of solid cereal brans, which may complicate post-treatment fertilization. Finally, ethanol is highly effective at triggering a rapid drop in redox potential when applied to saturated soil, which is a critical mechanism for generating the anaerobic conditions needed to manage soilborne pests.
8. How does ASD enhance phosphorus and potassium availability in soil?
Anaerobic soil disinfestation (ASD) enhances the availability of phosphorus (P) and potassium (K) through the direct addition of nutrients from organic amendments, chemical solubilization triggered by soil reduction, and microbial mobilization.
Direct Nutrient Addition from Amendments
The most immediate increase in soil P and K comes from the organic carbon sources incorporated at the beginning of the ASD process. Many of the materials utilized are naturally nutrient-dense:
• Rice Bran: Applying rice bran at a common rate of 20 Mg ha⁻¹ provides approximately 360 kg ha⁻¹ of phosphorus and 340 kg ha⁻¹ of potassium.
• Molasses: In Florida field studies, the application of molasses led to dramatic increases in exchangeable soil potassium, raising levels from under 50 mg K/kg soil to over 500 mg K/kg soil.
• Chicken Manure: The decomposition of amendments like composted chicken manure and molasses provides a significant nutritional boost, which has been linked to improved plant vigor and higher biomass in crops like sweetpotato.
Chemical Solubilization via Reducing Conditions
The anaerobic environment itself facilitates the release of nutrients already present in the soil matrix. As soil microorganisms consume the added carbon, they deplete oxygen and lower the redox potential (Eh). This shift to a strongly reduced state can increase phosphorus solubility, especially in acidic soils. These chemical changes alter the solubility of various soil minerals and cause a shift in the range of small organic molecules present in the soil solution.
Microbial Mobilization and Cycling
ASD induces a massive restructuring of the soil microbiome, favoring beneficial organisms that assist in nutrient availability.
• Specialized Cycling Communities: Post-treatment soils are often enriched with microbial taxa specifically associated with phosphorus and potassium cycling.
• Nutrient Mobilization: These shifted microbial communities positively correlate with soil nutrient mobilization, helping to transition minerals into forms that are more easily absorbed by plants.
• Persistence: These beneficial shifts can persist through the growing season, contributing to a more disease-suppressive and fertile environment.
By combining these biological and physiochemical effects, ASD can significantly reduce the need for external fertilizer inputs, potentially lowering overall production costs.
9. How does ASD affect plant growth and crop yields?
Anaerobic soil disinfestation (ASD) serves as a preplant treatment developed not only to control soilborne pathogens but also to manage yield decline across various crop production systems. Research generally indicates that ASD provides yield results and plant growth improvements that are comparable to, or exceed, those achieved with traditional chemical fumigation.
Impact on Crop Yields
In many studies, ASD has demonstrated the ability to maintain or increase marketable yields relative to fumigated and non-amended controls:
• Strawberries: Field trials in California showed that strawberry yields using ASD with rice bran were comparable to soil fumigation with 1,3-dichloropropene plus chloropicrin. In three of four trials, ASD produced marketable fruit yields equivalent to bed fumigation.
• Vegetables: In Florida, ASD with molasses combined with solarization produced bell pepper and eggplant yields equal to or higher than those from methyl bromide fumigation. Tomato yields in some locations were also significantly higher in ASD plots than in soil fumigation plots.
• Germplasm Compatibility: Studies on sweetpotato genotypes found that ASD combined with chicken manure and molasses resulted in significantly higher yields and disease tolerance in specific resistant genotypes compared to susceptible controls.
Effect on Plant Biomass and Vigor
ASD frequently stimulates superior plant growth, often measured through increased biomass and vigor:
• Biomass Increases: In sweetpotato experiments, ASD-treated plants had 47% higher dry above-ground biomass and 25% higher dry below-ground biomass compared to controls. Similarly, lettuce and mustard greens grown in ASD-treated muck soils exhibited significantly higher biomass in bioassays compared to those in non-treated soils.
• Plant Vigor: ASD has been shown to improve the vigor of various crops, including tomatoes, peppers, and sweetpotatoes. This increased vigor is often attributed to improved plant nutrition and water-holding capacity.
• Growth Promotion: Beyond killing pathogens, the process enhances beneficial soil microbial communities, such as Trichoderma and plant growth-promoting rhizobacteria (PGPR), which can produce phytohormones and antimicrobial substances that directly foster plant health.
Nutrient Dynamics and Fertility
The decomposition of organic amendments during ASD plays a major role in these growth benefits by modifying soil fertility:
• Enhanced Nutrient Availability: ASD can significantly increase the availability of essential elements, including phosphorus, potassium, calcium, and magnesium.
• Nitrogen Cycling: The impact on nitrogen is highly dependent on the amendment’s C: N ratio. Amendments with low C: N ratios can result in substantial inorganic nitrogen (ammonium) following treatment, providing a boost to crop nutrition.
Potential Factors Limiting Growth
While generally beneficial, ASD can occasionally have neutral or negative impacts on growth depending on management:
• Nitrogen Immobilization: Carbon sources with high C: N ratios, such as molasses, may cause nitrogen immobilization in microbial biomass, potentially leading to leaf nitrogen deficiency in crops like bell peppers unless post-treatment fertilization is increased.
• Phytotoxicity: Certain amendments, particularly those derived from Brassicaceae seed meals, can sometimes cause phytotoxicity, which may reduce seedling survival despite successful pathogen suppression.
• Pathogen Sensitivity: While ASD effectively manages many pathogens to promote growth, it is less effective against some aggressive oomycetes like Phytophthora drechsleri in gerbera daisy unless inoculum thresholds are very low.
10. How do transparent versus dark plastics affect soil temperature?
The type of plastic mulch used during anaerobic soil disinfestation (ASD) significantly impacts soil temperature, with transparent and dark plastics serving different functional roles based on climate and management goals.
Transparent Plastics and Solarization
Transparent (clear) plastics are primarily used to facilitate soil solarization, a process that traps solar radiation to heat the soil.
• Temperature Increase: Clear plastic film can elevate soil temperatures by up to 10°C more than opaque (black or green) plastics.
• Synergistic Effects: When combined with organic amendments in ASD, the use of clear plastic creates a process often called “biosolarization” or “biodisinfection”. This combination uses both high temperatures and anaerobic conditions to enhance the suppression of hardy pathogens like Verticillium dahliae and certain nematodes.
• Regional Use: In warm climates like Florida, clear UV-stabilized solarization film is frequently used to maximize heat during the treatment period.
Dark Plastics and Weed Control
Dark-colored plastics, such as black or green polyethylene, are widely used because they are cost-effective and provide superior weed control.
• Lower Relative Temperatures: While dark plastics still help maintain soil moisture and prevent air exchange, they do not provide the same dramatic heat boost as clear films because they absorb solar radiation rather than allowing it to pass through to the soil.
• Specific Applications: In some regions, such as the Netherlands, black plastic is used to ensure adequate soil temperatures for microbial activity during the ASD process.
• Management Flexibility: In high-temperature regions, growers may apply clear plastic first to facilitate solarization and then cover it with opaque plastic at the end of the ASD period. This provides the cooler soil temperatures necessary for successful crop production and transplanting.
Observed Temperature Ranges
Field data across different studies highlight these temperature differences:
• Transparent PE: In various trials, soil temperatures under transparent films reached ranges of 30–40°C in Florida and 18.1–35.5°C in Japan.
• Black PE: In a Tennessee trial, soil temperatures under black polyethylene were recorded at a lower average of approximately 21°C.
Ultimately, while ASD does not strictly require high temperatures to function, the higher heat provided by transparent plastics often reduces the time required for effective disinfestation.
11. What is the recommended delay before planting after tarping?
After removing the plastic tarp following anaerobic soil disinfestation (ASD), planting should typically be delayed for five to seven days. This waiting period is necessary to allow the soil time to dry and breathe.
Removing the cover is essential to reestablish soil aeration and to stimulate the degradation of anaerobic decomposition by-products. In specific research applications, such as field trials for lettuce and mustard greens or greenhouse studies for sweetpotato, planting was performed one week after treatment termination.
If ASD is applied to raised beds, holes can be cut into the plastic to allow the soil to breathe before transplanting. Additionally, it is recommended to grow a crop immediately following the delay to prevent the leaching of nutrients that may have become available during the disinfestation process.
12. Are there specific plastic types that prevent oxygen exchange better?
Specific types of plastic films are essential for performing anaerobic soil disinfestation (ASD) because they serve as an airtight barrier to prevent the resupply of oxygen to the soil. The sources identify Virtually Impermeable Film (VIF) and Totally Impermeable Film (TIF) as specialized materials that prevent gaseous exchange better than standard plastics. VIF is specifically recommended for its strength and high degree of airtightness, which is necessary to maintain an oxygen-free atmosphere. Similarly, TIF is used in some systems and offers the advantage of remaining in the field throughout the subsequent crop production cycle.
In addition to these specialized films, thick polyethylene with a thickness of 0.15 to 0.40 mm (commonly used for silage) is considered a suitable alternative. Standard thinner plastics are generally deemed insufficient because they are not airtight enough to achieve the required level of anaerobicity. The effectiveness of the seal can also be enhanced through multi-layering; for example, using three layers of thin plastic film can reduce oxygen permeability from 1500 mL/m²/day/bar to 240 mL/m²/day/bar compared to a single layer. Finally, the sources suggest using a heavier grade or embossed mulch to prevent damage and tearing, which would allow oxygen to re-enter the soil and compromise the treatment.
13. Can liquid amendments be applied through existing drip irrigation?
Yes, liquid amendments can be applied through existing irrigation systems, including drip lines. Liquid carbon sources such as molasses and ethanol are frequently introduced this way because they can be diluted and pumped through the lines after the plastic tarp has been installed.
Applying amendments via drip irrigation offers several practical and efficacy advantages:
• Ease of Application: In Japan, ethanol is widely applied through irrigation systems in both commercial fields and plastic houses.
• Deeper Soil Penetration: Because they are dissolved in irrigation water, these amendments can seep deeper and more uniformly into the soil profile than solid materials like cereal brans.
• Flexibility in Tarping: Soil can be covered with plastic mulch before irrigation if drip tape is placed underneath, allowing for precise delivery of the carbon source and water to reach saturation.
• Commercial Availability: In regions like Florida, double-filtered molasses is commercially available specifically for use in ASD because it is filtered to prevent clogging and can be applied directly via drip lines.
However, researchers have noted some potential limitations. Application through irrigation lines may sometimes result in a less even distribution of the carbon source compared to surface spraying. Additionally, if using solid amendments like molasses in a concentrated form, they must be properly diluted (often 1:3 or 1:4 with water) before application to ensure they flow correctly through the system.
14. What is the recommended soil depth for cereal bran incorporation?
For the incorporation of cereal brans (such as wheat or rice bran) during Anaerobic Soil Disinfestation (ASD), the recommended soil depth generally falls between 15 and 20 cm (6 to 8 inches). However, this depth can vary based on the specific pathogens being targeted and the type of equipment used:
• Standard Incorporation: Cereal brans are typically spread evenly over the treatment area and incorporated into the upper 15 to 20 cm of soil using a tractor-drawn or hand-pushed rototiller.
• Deeper Requirements: If the target pathogens infect the entire root system, incorporation may be required to a deeper level, such as 40 cm. In some cases, operating depths have been noted as deep as 80 cm, though the amount of amendment must be adjusted accordingly (e.g., 80 tons/ha for an 80 cm depth).
• Typical Practical Range: Other sources suggest a practical range of 20 to 30 cm, depending on the method of bed formation and mechanical incorporation.
• Specific Trial Examples: In field trials for strawberries, rice bran has been successfully mixed into premade beds at a depth of 0 to 15 cm.
Important Limitation: The suppressive effect of solid materials like cereal bran is generally limited to the depth of incorporation. This can be a challenge for managing pests like plant-parasitic nematodes, which may reside deeper in the soil profile (sometimes greater than 1.5 meters) than standard tilling equipment can reach. In contrast, liquid carbon sources are often preferred when deeper penetration is required because they can seep further into the soil profile via irrigation.
15. Can ethanol treat deep-rooted pathogens better than solid amendments?
Yes, ethanol can treat deep-rooted pathogens more effectively than solid amendments because of its ability to penetrate much further into the soil profile. While solid materials like cereal brans are typically limited to the depth of physical incorporation (generally the upper 15 to 30 cm), liquid amendments applied via irrigation systems can seep deeper and more uniformly throughout the soil matrix.
This superior depth of penetration is critical for managing specific pests, such as plant-parasitic nematodes, which can migrate into the crop root zone from depths exceeding 1.5 meters. Because solid amendments cannot reach these depths, they are often insufficient for commercially viable nematode control in systems like tree-crop nurseries.
Key advantages of using ethanol for deep-soil treatment include:
• Irrigation Application: In Japan, ethanol is widely diluted and applied through irrigation lines in both open fields and plastic houses to ensure deep and even distribution.
• Enhanced Reach: Delivering soluble carbon sources through buried irrigation lines has been proposed as a method to generate anaerobic conditions at significantly greater depths than traditional surface tilling allows.
• Faster Inactivation: In laboratory settings, ethanol has been shown to reduce pathogens like Fusarium oxysporum to undetectable levels in as little as 3 to 9 days, outperforming solid wheat bran which required at least 9 days.
16. Which amendments best provide phosphorus and potassium for organic crops?
Based on the sources, several organic amendments used in anaerobic soil disinfestation (ASD) are highly effective at increasing the availability of phosphorus (P) and potassium (K) for crops.
Highly Effective Amendments
The sources highlight specific amendments for their significant nutrient contributions:
• Rice Bran: This is a particularly nutrient-dense option; applying rice bran at a rate of 20 Mg ha⁻¹ provides approximately 360 kg ha⁻¹ of phosphorus and 340 kg ha⁻¹ of potassium.
• Molasses: Liquid molasses has been shown to cause dramatic increases in exchangeable soil potassium. In Florida field studies, molasses applications increased exchangeable K from less than 50 mg/kg to over 500 mg/kg.
• Chicken Manure and Molasses (CM + M): In organic sweetpotato production, the combination of composted chicken manure and molasses significantly increased phosphorus and potassium values after the ASD process. This nutrient boost was linked to improved plant vigor and increased dry biomass.
• Tobacco Stems: Research indicates that using tobacco stems as a carbon source significantly increases the available potassium and organic matter levels in the soil.
• Wheat Bran and Tomato Pomace: In trials involving soil-based substrates, both wheat bran and tomato pomace significantly increased exchangeable potassium compared to non-amended controls. Wheat bran also increased phosphorus concentrations in soilless substrates.
Mechanisms for Nutrient Increase
The increase in these essential nutrients occurs through three primary mechanisms described in the sources:
1. Direct Nutrient Addition: Many organic amendments naturally contain high amounts of P and K, which are released as the material decomposes.
2. Chemical Solubilization: The anaerobic environment created during ASD triggers soil reduction processes. These processes can increase phosphorus solubility, particularly in acidic soils, by altering the chemical nature of the soil matrix.
3. Microbial Mobilization: ASD induces a shift in the soil microbiome, favoring microbial communities capable of phosphorus and potassium cycling. These beneficial microbes positively correlate with soil nutrient mobilization, making minerals more accessible to the plants.
Because ASD can significantly improve the availability of these nutrients, the sources suggest that growers may be able to reduce or eliminate pre-plant fertilizers, thereby lowering production costs for organic operations. However, it is recommended to perform soil analyses after the ASD incubation period to adjust fertilization programs based on the specific nutrients released.
17. How do carbon-to-nitrogen ratios affect post-treatment fertilizer needs?
The carbon-to-nitrogen (C: N) ratio of the organic amendments used in Anaerobic Soil Disinfestation (ASD) is a primary driver of soil nitrogen availability and dictates whether post-treatment fertilizer needs will increase or decrease.
Low C: N Ratios (<20:1)
Amendments with a low C: N ratio generally mineralize nitrogen, releasing it into the soil in a form plants can use.
• Reduced Fertilizer Needs: When amendments with large amounts of nitrogen and low C: N ratios are used, substantial inorganic nitrogen (ammonium) can remain in the soil post-treatment.
• Case Studies: Applying rice bran (which has a moderate C: N ratio) at high rates can provide approximately 460 kg ha⁻¹ of nitrogen, which may allow for the complete elimination of pre-plant fertilizers.
• Management Risks: Excessive inorganic nitrogen following low C: N treatments must be managed carefully to provide adequate nutrition while preventing environmental degradation through leaching or denitrification.
High C: N Ratios (>20:1)
Amendments with a high C: N ratio typically lead to nitrogen immobilization, where soil microorganisms consume available inorganic nitrogen to process the added carbon.
• Increased Fertilizer Needs: High C: N ratios tie up nitrogen in microbial biomass, making it temporarily unavailable to crops. This often necessitates increased post-treatment nitrogen fertilization to maintain crop productivity.
• Case Studies: In trials using molasses (a high C: N source), bell peppers and eggplants often exhibited leaf nitrogen deficiency despite being planted in treated soil, requiring supplemental fertilization.
• Mitigation strategies: Growers can mix high C: N sources like molasses with lower C: N sources (such as composted poultry litter or rice bran) to provide a better nutritional balance and reduce overall costs.
Other Nutrient Considerations
While the C: N ratio specifically impacts nitrogen, the decomposition of organic amendments also releases other essential minerals:
• Many organic amendments are naturally high in phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S).
• The release of these nutrients, combined with the chemical solubilization triggered by anaerobic conditions, can significantly reduce overall fertilizer costs for these elements regardless of the nitrogen status.
Ultimately, researchers recommend performing a soil analysis after the ASD incubation period but before planting to adjust fertilization programs based on the specific mineralization or immobilization patterns of the carbon source used.
18. How do you avoid clogging irrigation lines with liquid molasses?
To avoid clogging irrigation lines when using liquid molasses for Anaerobic Soil Disinfestation (ASD), growers should focus on two primary strategies: proper filtration and significant dilution.
Filtration and Material Selection
The most effective way to prevent obstructions is to use a specific grade of the amendment. The sources highlight that double-filtered molasses is commercially available specifically for ASD applications, as it is processed to ensure it can be delivered through drip lines without causing issues. Utilizing high-quality, filtered feed-grade blackstrap molasses is a standard practice in regions like Florida.
Dilution Ratios
Because molasses is naturally thick and viscous, it cannot be applied in its concentrated form through existing irrigation systems. It must be diluted with water before application. The recommended dilution ratios are as follows:
• 1:3 to 1:4 (molasses to water): This specific range is suggested to ensure the liquid is thin enough to flow through drip tapes and emitters correctly.
• Applying the molasses as a diluted solution through double drip tape placed under the plastic mulch ensures the carbon source reaches the necessary saturation levels to trigger anaerobiosis.
Practical Benefits of Drip Application
Applying molasses through drip lines rather than surface spraying not only helps manage the viscosity of the material but also improves the uniformity and depth of penetration into the soil profile. In some systems, the plastic mulch is installed first, and the diluted molasses is then pumped through the lines, allowing for precise delivery and saturation of the treated area. However, researchers note that even with dilution, application through irrigation lines can sometimes result in less even distribution compared to surface sprays, requiring careful monitoring of the delivery system.
References
Liu, D., Samtani, J. B., & Butler, D. M. (2025). Effect of Anaerobic Soil Disinfestation with Yeast Amendment on Weed Control and Strawberry Yield. HortScience, 60(9), 1510–1520. https://doi.org/10.21273/HORTSCI18692-25
Lopes, E. A., Canedo, E. J., Gomes, V. A., Vieira, B. S., Parreira, D. F., & Neves, W. S. (2022). Anaerobic soil disinfestation for the management of soilborne pathogens: A review. In Applied Soil Ecology (Vol. 174). Elsevier B.V. https://doi.org/10.1016/j.apsoil.2022.104408
Meshram, S., Philbrick, A. N., & Adhikari, T. B. (2024). Anaerobic soil disinfestation: a biologically-based solution for sustainable crop production. In Frontiers in Horticulture (Vol. 3). Frontiers Media SA. https://doi.org/10.3389/fhort.2024.1436782
Rosskopf, E. N., Serrano-Pérez, P., Hong, J., Shrestha, U., Rodríguez-Molina, M. del C., Martin, K., Kokalis-Burelle, N., Shennan, C., Muramoto, J., & Butler, D. (2015). Anaerobic Soil Disinfestation and Soilborne Pest Management (pp. 277–305). https://doi.org/10.1007/978-3-319-23075-7_13
Shennan, C., Muramoto, J., Koike, S., Baird, G., Fennimore, S., Samtani, J., Bolda, M., Dara, S., Daugovish, O., Lazarovits, G., Butler, D., Rosskopf, E., Kokalis-Burelle, N., Klonsky, K., & Mazzola, M. (2018). Anaerobic soil disinfestation is an alternative to soil fumigation for control of some soilborne pathogens in strawberry production. Plant Pathology, 67(1), 51–66. https://doi.org/10.1111/ppa.12721
Singh, S., Cutulle, M., Rutter, W., Wadl, P. A., Ward, B., & Khanal, C. (2025). Anaerobic Soil Disinfestation as a Tool for Nematode and Weed Management in Organic Sweetpotato. Agronomy, 15(3). https://doi.org/10.3390/agronomy15030548
Strauss, S. L., & Kluepfel, D. A. (2015). Anaerobic soil disinfestation: A chemical-independent approach to pre-plant control of plant pathogens. In Journal of Integrative Agriculture (Vol. 14, Number 11, pp. 2309–2318). Editorial Department of Scientia Agricultura Sinica. https://doi.org/10.1016/S2095-3119(15)61118-2
Testen, A. L., & Miller, S. A. (2019). Anaerobic soil disinfestation to manage soilborne diseases in muck soil vegetable production systems. Plant Disease, 103(7), 1757–1762. https://doi.org/10.1094/PDIS-09-18-1578-RE
Yanez, D. G., Testen, A. L., & Hand, F. P. (2024). Evaluation of Anaerobic Soil Disinfestation to Reduce Soilborne Diseases in Soilless and Soil-Based Substrates for Specialty Cut Flower Production. Plant Disease, 108(4), 908–919. https://doi.org/10.1094/PDIS-05-23-0857-RE






