1. Introduction: The Strategic Role of Adjuvants in Modern Vaccinology
In the strategic development of modern prophylactic and therapeutic vaccines, pathogen antigens are frequently found to be insufficiently immunogenic when administered in isolation. Adjuvants are therefore a critical necessity to enhance the magnitude, quality, and duration of the adaptive immune response.

The term adjuvant is derived from the Latin adjuvare, meaning “to help” or “to sustain.” In the architecture of a modern vaccine, the antigen serves as the blueprint—providing the specific biochemical signature of a pathogen—but the adjuvant is the engine that drives the immune response.

The necessity for these helpers arises from a fundamental challenge in vaccinology: highly purified antigens, while safe, are often poorly immunogenic. They lack the inherent “danger signals” required to transition the immune system from a state of quiet surveillance to active, lasting memory. Without an adjuvant, the body may simply ignore the antigen or clear it before a robust defense can be mounted.
As defined within the current regulatory and scientific literature, an adjuvant is a substance used to “help or sustain” the immune system by accelerating, prolonging, or targeting the response toward specific immunological outcomes. By facilitating more robust antigen-specific immune responses, these agents enable essential dose-sparing strategies and the induction of high-affinity immunological memory. Their role effectively acts as a pharmacological catalyst to ensure the “message” of the vaccine is received and remembered.
1.1 1926: Serendipity and the Dawn of Alum
In the 1920s, vaccine manufacturing was a paradox. Researcher Alexander Glenny noticed that ultra-pure vaccine batches were often less effective than those considered “dirty” or “polluted.” While testing diphtheria toxoids, he found that adding aluminum salts—initially viewed as an impurity—actually sparked a significantly stronger antibody response in animal models.

The Great Shift in Perspective Scientists originally viewed impurities as “pollutants” to be purged. Glenny’s breakthrough revealed that these substances were actually “immune enhancers,” providing the necessary biological “spark” to wake up the immune system’s defenses.
To explain this, researchers proposed the “Depot Theory.” In plain language, this suggested that aluminum salts act as a slow-release reservoir (a “depot”) at the injection site. Instead of the antigen being immediately washed away, the adjuvant holds it in place, allowing Antigen-Presenting Cells (APCs)—the immune system’s “scout cells”—ample time to find, process, and report the threat. For nearly a century, this slow-release mechanism remained the dominant explanation for adjuvant function.
1.2. The Mid-Century “Gold Standard”: Aluminum Salts
Following Glenny’s work, aluminum-based mineral salts became the global “gold standard.” Their dominance was not merely a matter of efficacy; it was reinforced by a regulatory landscape that, for decades, did not foresee a framework for authorizing an adjuvant as a “stand-alone” product. Consequently, adjuvants were only licensed as part of a specific vaccine formulation.
| Year of Milestone | Adjuvant Type | Sample Brand Names/Vaccines |
|---|---|---|
| 1932 | Aluminum potassium sulfate | Pentacel, Quadracel, Adacel, TENIVAC, TDVAX |
| 1939 | Aluminum hydroxide | Havrix, Vaqta, Engerix-B, Recombivax HB, Infanrix, Pediarix, PedvaxHIB, ActHIB, Prevnar 13, Pneumovax 23 |
| 2000 | Calcium phosphate | Comvax, Hepavax, IPOL, BCG, MMR, Stamaral |
| 2006 | Amorphous aluminum hydroxyphosphate sulfate | Recombivax, PedvaxHIB, Gardasil, Vaxelis |
Despite the rise of new technologies, “Alum” remains the predominant global choice due to its robust safety profile and high efficiency in inducing humoral (antibody) responses. However, as science tackled more complex pathogens, it became clear that some threats required more than a “slow release”—they required a specific biological alarm signal.
1.3. The 1990s-2000s: The Emulsion and Virosome Revolution
In the late 1990s, the “Depot Theory” was expanded by the introduction of complex delivery systems: Virosomes (1994) and oil-in-water emulsions like MF59 (1997) and AS03 (2009). These are not passive reservoirs; they are active transporters.
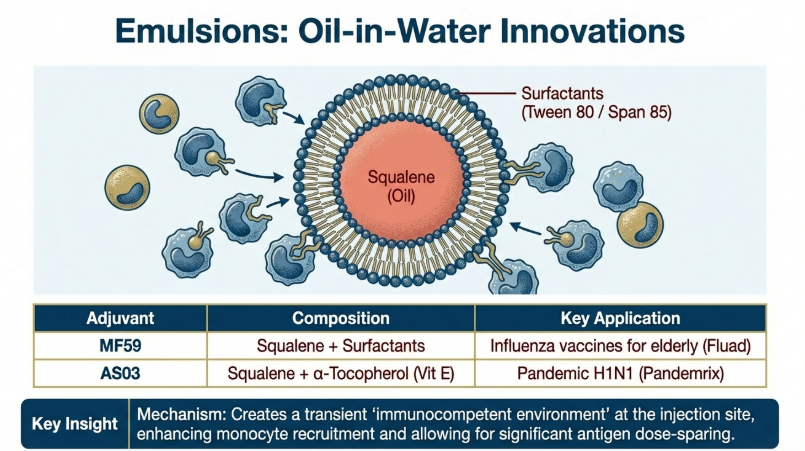
The Components of MF59:
1. Squalene: A natural oil found in humans, plants, and animals.
2. Polysorbate 80: An emulsifier that stabilizes the oil-water mixture.
3. Sorbitan trioleate: A surfactant that ensures the stability of tiny oil droplets.
Unlike Alum, which primarily stays at the injection site, emulsions are more aggressive. They induce Damage-Associated Molecular Patterns (DAMPs) and trigger Pattern Recognition Receptors (PRRs), which act as “natural warning signals.” These signals recruit immune cells more effectively, leading to faster and more robust responses, particularly in seasonal and pandemic influenza vaccines.
1.4 Modern Precision: Synthetic Agonists and LNPs
We have entered a “New Age” where adjuvants are precision-engineered. Instead of general stimulation, scientists use Synthetic Agonists and Lipid Nanoparticles (LNPs) to engage specific immune pathways.

Traditional vs. Modern Adjuvants
| Feature | Traditional (e.g., Alum) | Modern (e.g., CpG 1018, LNPs, Matrix M) |
|---|---|---|
| Mechanism of Action | Antigen Depot / Slow release | Direct molecular signaling (TLR/PRR Agonists) |
| Targeted Response | Primarily Humoral (Th2/Antibodies) | Balanced Th1/Th2; Restrains high-stability peptides to select low-affinity peptides for MHC II, focusing the T-cell response. |
| Recent Examples | DTP, Hep B | COVAXIN (Alhydroxiquim-II), Novavax (Matrix M), Shingrix (AS01B) |
The “LNP Revolution” of 2020 redefined the field. LNPs are not just helpers; they are “biological protective envelopes.” They shield delicate mRNA “instruction manuals” from being shredded by the body’s enzymes, ensuring they safely reach the cell to begin vaccine production.
2. Taxonomy of Vaccine Adjuvants: From Delivery Systems to Immunomodulators
A rigorous classification of adjuvants by their functional modality—delivery system versus immune stimulation—is vital for rational vaccine design and predictable clinical outcomes. From a regulatory perspective, it is critical to distinguish between “adjuvants,” which are integral to the vaccine formulation, and “immunomodulators,” which the EU explanatory note defines as compounds administered separately or at different time points to pre-condition the host immune environment.

The primary taxonomic categories include:
2.1 Delivery Systems:
These include oil-in-water emulsions (e.g., MF59, AS03), mineral salts, microparticles, liposomes, and virosomes. Their strategic role is to act as a carrier material that facilitates antigen presentation to Antigen Presenting Cells (APCs) while prolonging the bioavailability of the antigen.
Delivery systems act as sophisticated carriers. Their primary role is to organize the vaccine components into a physical format that the immune system can easily recognize and process. The most prevalent versions of these are Mineral Salts and Emulsions.
The Timed-Release Reservoir: The Depot Effect
A primary mechanism of delivery systems is the “Depot Effect.” Rather than allowing the body to rapidly metabolize a fleeting signal, these systems create a timed-release reservoir at the injection site. This prevents the immune system from “overheating” with too much information at once or, conversely, ignoring the antigen entirely. By releasing the antigen slowly, the adjuvant ensures a sustained level of exposure, which is critical for developing high-affinity immune responses.

Facilitating the Frontline Hand-off
Delivery systems are essential for Antigen Presentation. They bundle the antigen into particulate forms that are easily “captured” by Antigen Presenting Cells (APCs)—the immune system’s frontline intelligence analysts.
The Nuance of Mineral Salts: Beyond “Alum”
As a specialist, it is vital to note that “Alum” is not a monolithic substance. Synthesis conditions, specifically pH, dictate its effectiveness. When synthesized under acidic or near-neutral conditions (pH 5–7), Aluminum Hydroxide exhibits anisotropic growth, resulting in low-crystallinity (amorphous) nanoparticles. Critically, these amorphous forms preserve protein stability far better than the highly crystalline versions produced in basic (pH 9+) environments.
| Type | Physical Form | Primary Licensed Example |
|---|---|---|
| Mineral Salts | Suspension of Nanoparticles (Amorphous or Crystalline) | Alum (Aluminum Hydroxide) |
| Emulsions | Oil-in-water or water-in-oil droplets | MF59 or AS03 (Squalene-based) |
But even the best delivery does not matter if the body fails to realize it is under attack; this is where our “Alarm Systems” come in.
2.2 Immunomodulators:
Comprising toll-like receptor agonists (TLR agonists such as MPL and CpG) and NOD agonists, these act as “danger signals.” They activate APCs by binding to specific pattern recognition receptors, signaling a transition from innate to adaptive immunity.

Immunomodulators, or immunostimulants, trigger “danger signals” by binding to specific receptors. They do not just carry the vaccine; they actively prime the APCs to respond more robustly.
Focusing the Magnifying Glass: MHC II Presentation
A transformative “how” of immunomodulators involves MHC II complexes. Adjuvants like TLR agonists actually change the “peptide repertoire” shown to T-cells. By restraining the presentation of high-stability/high-affinity peptides and favoring low-affinity ones, adjuvants focus the immune response like a magnifying glass. This forces the immune system to target specific, potentially more effective regions of the antigen that it might otherwise overlook.
The 3 Key Benefits of Activation:
1. Faster Response: Identifying the threat and initiating defense protocols immediately.
2. Stronger Immune Memory: Promoting the development of specialized memory T-cells for years of protection.
3. Vulnerable Population Support: Overcoming “poor responsiveness” in the elderly or immunocompromised.
The Spectrum of Stimulants
We categorize these alarms by the specific Pattern Recognition Receptors (PRRs) they trigger:
• TLR Agonists (Toll-like Receptors)
◦ TLR 1/2: Stimulated by molecules like L-pampo.
◦ TLR 3: Triggered by Poly I:C (mimicking viral RNA).
◦ TLR 4: Triggered by MPL (Monophosphoryl lipid A).
◦ TLR 5: Triggered by Flagellin (bacterial protein).
◦ TLR 7/8: Triggered by R837 or R848.
◦ TLR 9: Triggered by CpG (synthetic DNA sequences).
• NOD Agonists (Nucleotide-binding oligomerization domain)
◦ NOD Stimulants: Triggered by molecules like MDP.
Often, a single alarm isn’t enough for complex pathogens, leading scientists to develop “team-based” solutions.
2.3 Combination Adjuvants:
Combined adjuvants (the “AS” or Adjuvant System series) utilize multiple molecules to work synergistically. By pairing a delivery system with one or more immunomodulators, we can achieve results greater than the sum of their parts.

Steering the Immune Bias
The true power of combinations is the ability to steer the immune bias. A simple Alum adjuvant typically triggers a Th2 (humoral/antibody) response. However, fighting diseases like Malaria or Shingles requires a Th1 (cytotoxic/cellular) response—the “killer” T-cell arm of the immune system. Synergistic teams allow us to “dial in” the exact type of cellular response required for the target disease.
“Power Team” Examples:
• AS01: Combines a TLR4 ligand (MPL) and a saponin (QS-21). This is the engine behind the Shingles vaccine, specifically designed to elicit a massive Th1/Killer T-cell response.
• AS04: Combines Alum (the transporter/depot) with MPL (the alarm). Used in HPV and Hepatitis B vaccines to provide both stability and high antibody titers.
• AS03: A squalene-based emulsion team that recruits immune cells and enhances cytokine production for pandemic influenza.
While these teams provide systemic protection, some vaccines must be the first line of defense at our body’s physical “entry points.”
2.4 Mucosal Adjuvants:
Mucosal adjuvants are specialized compounds that strengthen defenses in the “wet” tissues of the respiratory and digestive systems.
These aids are unique because they provide both local protection (secreting antibodies exactly where the germ arrives) and systemic protection (body-wide). Because the mucosal environment is harsh, these adjuvants—such as Chitosan or CT (Cholera Toxin derivatives)—act as gatekeepers that ensure the antigen is not washed away or degraded before the immune system can react.
Mucosal aids are the essential “secret sauce” for intranasal (nasal spray) or oral (swallowed) vaccines. They are critical for preventing the initial attachment of pathogens to the lungs or gut lining.
Selecting the appropriate class requires a deep analytical evaluation of its physicochemical impact on the vaccine antigen, as the surface microenvironment dictates both shelf-life and immunological “focus.”
3. Physicochemical Engineering: Tuning Aluminum Hydroxide for Antigen Stability
The “depot theory” historically suggested that aluminum salts functioned solely as an antigen reservoir for sustained release. However, modern molecular immunology recognizes that the local microenvironment of the adjuvant surface is the primary determinant of vaccine shelf-life and efficacy. For developers, the synthesis conditions of aluminum hydroxide nanoparticles are critical for maintaining the structural integrity of bound proteins like Bovine Serum Albumin (BSA) or Beta-Lactoglobulin (BLG).
Analytical findings demonstrate that synthesis pH governs the spatial arrangement of surface hydroxyl groups, which in turn dictates interfacial hydration. Nanoparticles synthesized under acidic or neutral conditions (AH1/AH1-N) exhibit low crystallinity and higher interfacial hydration. In contrast, basic synthesis (AH2) results in high crystallinity and an enriched density of surface hydroxyls. Crucially, high hydroxyl density leads to limited water accessibility because extensive hydrogen bonding between surface hydroxyls restricts the structuring of water at the interface. This “restricted water accessibility” is a primary mechanism behind the reduced thermal stability of antigens bound to highly crystalline surfaces.
Comparison of Aluminum Hydroxide Nanoparticle Properties
| Synthesis pH | Adjuvant Label | Crystallinity | Hydration Level | Impact on Antigen Stability | Synthesis Temp / Duration | Model Antigen |
|---|---|---|---|---|---|---|
| Acidic (pH ≈ 5.0) | AH1 | Low | High | Improved Stability | 150°C / 24h | BSA / BLG |
| Neutral (pH ≈ 7.1) | AH1-N | Intermediate | High | Improved Stability | 150°C / 24h | BSA / BLG |
| Basic (pH ≈ 9.0) | AH2 | High | Reduced | Reduced Stability | 150°C / 24h | BSA / BLG |
Ensuring stability at the molecular level through precise physicochemical tuning is a prerequisite for generating a predictable immune response during Phase III clinical trials.
4. Mechanistic Insights: How Adjuvants Fine-Tune the T-Cell Response
The conceptualization of adjuvants has shifted from simple “helpers” to precise modulators of the MHC II peptide repertoire. Adjuvants like MPLA and CpG do not merely boost the magnitude of the response; they actively influence antigen processing within the APC.
A significant finding in molecular immunology (Li et al., 2024) is that adjuvanted conditions actually restrain the presentation of high-stability/high-affinity peptides. Instead, adjuvants favor the presentation of peptides with low affinity for MHC II molecules. By narrowing the antigen repertoire in this manner, the adjuvant “focuses” the T-cell response on specific epitopes that might otherwise be overlooked. As stated in the source context:
“Adjuvants, beyond increasing the magnitude of a given immune response, may also fine-tune the portion of the antigen targeted by T cells.”
This focusing of the peptide repertoire ensures that the resulting CD4+ T-cell help is targeted toward regions of the antigen most likely to induce high-quality, neutralizing antibodies.
5. EU Regulatory Framework: Clinical and Non-Clinical Evaluation Standards
In the European Union, the legal landscape—governed by Directive 2001/83/EC—dictates that adjuvants are treated as constituents of the medicinal product rather than stand-alone active substances. This means an adjuvant cannot be authorized independently; it must be evaluated within the context of a specific Marketing Authorization Application (MAA).

Under European legislation, specifically Directive 2001/83/EC, a rigid legal distinction is drawn:
• Adjuvant: Defined as a constituent of the vaccine that is not the active substance, intended to enhance, speed up, prolong, or target the immune response.
• Active Substance: The primary agent (e.g., antigen) intended to elicit the specific immune response.
It is a common misconception—particularly among US-based firms accustomed to different FDA pathways—that an adjuvant can be authorized as a stand-alone product via a “Master File” approach. In the EMA jurisdiction, there is No Stand-Alone Authorization.
“So What?”: The Integration Mandate The EMA does not recognize an independent “Adjuvant Master File.” Because the adjuvant is legally a constituent of the final medicinal product, comprehensive data on its quality, safety, and efficacy must be integrated directly into the specific vaccine dossier. Any modification—including the addition or replacement of an adjuvant in a licensed vaccine—is treated as a major variation requiring new confirmatory studies. Strategically, this means your CMC and clinical data must be tethered to a specific antigen from day one.
While the adjuvant is not the “active substance,” the EMA treats it with nearly equal rigor in the Chemistry, Manufacturing, and Controls (CMC) section of the dossier, necessitating a technical foundation of the highest fidelity.
The regulatory requirements for the Dossier are structured across three phases:
1. Quality Requirements: Comprehensive characterization of physical and biochemical traits (e.g., surface charge, viscosity, pH). Purity must be established (endotoxin/bioburden), with a specific focus on ruminant-origin risks (TSE compliance).
2. Non-Clinical Development: Dose-response studies in two species (typically one rodent and one non-rodent) are required. Developers must provide “proof-of-concept” challenge studies to justify the adjuvant-antigen ratio.
3. Clinical Phase Requirements:
◦ Phase I/II: Evaluation of safety and dose-finding in healthy adults and the target population.
◦ Phase III: Randomized, double-blind trials are required. For a new vaccine, the safety database must typically include a minimum of 3,000 subjects.
◦ Nuance: For pediatric indications, the regulator expects a sample size of at least 300 subjects per specific pediatric age group to ensure adequate stratification of safety data.
Rigorous vaccine pharmacovigilance is the only way to detect rare, immune-mediated adverse events that only emerge after widespread population exposure.
5.1 Quality Standards: Physicochemical Characterization and Manufacturing
Structural integrity at the nanoscale is the bedrock of clinical success. For an adjuvanted vaccine, the manufacturing process must be defined with granular precision to ensure that structural outcomes dictate—rather than disrupt—long-term shelf-life and antigen stability.
Critical Parameters for Characterization
To demonstrate technical command during a CHMP review, manufacturers must monitor specific physicochemical metrics:
• Physical Metrics: Visual appearance, density, viscosity, pH, surface charge (Zeta potential), and particle size distribution.
• Biochemical Metrics: Compositional analysis and the identification of specific crystalline planes. For aluminum-based systems, this requires confirming the presence of (020), (120), (031), (051), and (151) planes of γ-AlOOH (boehmite).
• Purity Metrics: Endotoxin content, bioburden, and manufacturing residuals (e.g., surfactants like Polysorbate 80).
Correlation of Synthesis Environment and Structural Outcomes
Data from aluminum hydroxide nanoparticle research proves that synthesis pH is the primary lever for tuning adjuvant-antigen interactions. High-crystallinity surfaces can actually be detrimental to the vaccine’s profile.
| Synthesis pH | Structural Outcomes (Crystallinity, Density, Hydration) | Antigen Stability Impact |
|---|---|---|
| Acidic (pH ~5.0) | Low crystallinity (amorphous/poorly ordered); low hydroxyl density; high interfacial hydration. | High: Superior preservation of native protein thermal stability. |
| Basic (pH ~9.0) | High crystallinity (ordered γ-AlOOH); hydroxyl-dense surfaces; markedly reduced hydration. | Low: Promotes partial unfolding and reduced thermal stability. |
Basic synthesis environments create hydroxyl-dense, highly crystalline surfaces that restrict water accessibility. This lack of hydration and high crystallinity creates a rigid microenvironment that often leads to reduced protein stability. Neutral or acidic synthesis is generally preferred for maintaining the integrity of the antigen.
Association Stability
The dossier must provide rigorous proof of Association Stability. It is not enough to show that an antigen binds to the adjuvant; you must prove it does not dissociate during its shelf-life. EMA evaluators expect sophisticated methodologies to measure this, such as centrifugation-based assays to separate and quantify free versus bound antigen, or in-situ Synchrotron X-Ray Diffraction (SXRD) to evaluate structural integrity in the dispersed aqueous phase.
5.2 Non-Clinical Toxicological Strategy and Proof-of-Concept (PoC)
Animal models serve the strategic role of de-risking human trials and setting safe starting doses. However, a “one-size-fits-all” approach to animal testing is a regulatory red flag.
The Two-Species Requirement and the Extrapolation Risk
The EMA generally requires testing in two species (one rodent, one non-rodent). This is mandatory for adjuvants with low species-specificity, such as oil-in-water emulsions (MF59, AS03).
• Justification for Single Species: For highly species-specific biological classes (e.g., TLR agonists or certain cytokines), a single-species justification may be accepted if a second species provides no relevant biological data.
• The Alum Extrapolation Gap: As a Director, I must warn that animal models are imperfect predictors. For example, aluminum hydroxide is known to enhance the immunogenicity of split virion vaccines in mice, ferrets, and macaques, yet it often fails to show the same enhancement in humans. This discrepancy must be addressed in your non-clinical summary to manage expectations for Phase I outcomes.
The Toxicological Program
1. Repeated-Dose Toxicity: Must mirror the clinical schedule and use at least one additional dose than planned for human use.
2. Immunotoxicity: Specifically focusing on hypersensitivity. Assays like Passive Cutaneous Anaphylaxis (PCA) and Active Systemic Anaphylaxis (ASA) are essential to detect adjuvant-induced IgE increases.
3. Local Reactogenicity: Evaluation of granulomas and injection site reactions via full necropsy.
4. Pyrogenicity: Mandatory in vitro tests for fever-inducing substances.
Proof-of-Concept (PoC) Requirements
For novel adjuvants, PoC must be comparative. You must assess the vaccine with the new adjuvant against the antigen-only control or a well-established benchmark (e.g., standard Alum). You must establish that the adjuvant effectively enhances the response in a relevant challenge model before the first-in-human (FIH) trial.
5.3 Clinical Development: Safety Milestones and Dose Finding
Clinical development marks the shift from immunogenicity magnitude to the qualitative “fine-tuning” of the T-cell response.
“Fine-Tuning” via MHC II Peptide Presentation
Strategic value is found in how adjuvants like MPLA and CpG influence the specificity of the response.
• The Mechanism: Adjuvants activate pattern recognition receptors (PRRs) on Antigen Presenting Cells (APCs), which actually restrains the presentation of high-stability, high-affinity peptides.
• The Benefit: By focusing the immune system on a narrower repertoire of low-affinity MHC II peptide sequences, adjuvanted vaccines “fine-tune” the T-cell response. Conversely, unadjuvanted vaccines often lead to a broader repertoire of presented sequences, which is less efficient and lacks the targeted activation required for difficult pathogens.
The 3,000-Subject Safety Rule
The EMA mandates a safety database reflecting at least 3,000 subjects who received the final vaccine formulation. This database must be stratified by age to reflect the target population.
Clinical Researcher Checklist for Pediatric Stratification: To satisfy EMA guidelines, pediatric databases must include at least 300 subjects in each of the following cohorts:
• Infants (0–23 months)
• Toddlers (2–3 years)
• Young Children (4–8 years)
• Children 9–11 years
• Adolescents 12–14 years
• Adolescents 15–17 years
6. Case Study in Safety: Pandemrix, AS03, and the Narcolepsy Association
The 2009 H1N1 pandemic influenza vaccine, Pandemrix, serves as a pivotal case study for the competitive and public-trust landscape. Pandemrix utilized the AS03 adjuvant system (squalene, alpha-tocopherol, and polysorbate 80). Following licensure, an increased incidence of narcolepsy (1.4–8 additional cases per 100,000) was observed in children and adolescents.
Analysis suggests that narcolepsy in these cases was likely an autoimmune reaction triggered by cross-reactivity between a specific T-cell epitope in the H1N1 virus antigen and hypocretin-producing cells in the hypothalamus. While the adjuvant increased the immune response, the event was also linked to specific HLA haplotype vulnerabilities in the host. This case emphasizes that safety is a function of the antigen-adjuvant-host triad, requiring sophisticated surveillance to distinguish between these factors.
7. Future Horizons: Nanotechnology and Systems Vaccinology
Emerging “antigen-agnostic” platforms and systems biology are revolutionizing pandemic preparedness.
7.1 The Future: Atomic Tuning and Nanotechnology
The final frontier is “Atomic Tuning,” where scientists manipulate Aluminum Hydroxide at the nanoparticle level. By adjusting the synthesis environment—specifically maintaining a pH between 5.0 and 7.1 (acidic to neutral)—researchers can “tune” three critical parameters:
• Crystallinity: The degree of atomic order.
• Hydroxyl Density: The number of chemical “sticky” groups on the surface.
• Hydration State: The amount of water trapped at the particle interface.
The Rational Design Principle: Research has produced an “aha!” moment: particles synthesized in these acidic/neutral conditions exhibit lower crystallinity and higher interfacial hydration. Crucially, these amorphous particles improve the thermal stability of the antigen proteins bound to them. Conversely, highly crystalline particles synthesized in basic conditions (pH 9.0) can actually destabilize the antigen. We are now moving toward a future where we can “rationally design” the adjuvant to perfectly preserve the antigen it carries.

The future of the field is defined by nine strategic perspectives:
1. Nanotechnology: Engineering microparticles for precision delivery and safety.
2. Synthetic/Biosynthetic Materials: Improving supply chain robustness and reducing batch heterogeneity.
3. Outer Membrane Vesicles (OMVs): Utilizing bacterial vesicles for low toxicity and high innate stimulation.
4. Targeted Adjuvant Systems: Formulations tailored for the “inflammaging” profile of the elderly or the specific needs of the immunocompromised.
5. Pandemic Preparedness: Rapid platforms like LNPs (used in SARS-CoV-2) for swift response.
6. Polysaccharide Adjuvants: Exploiting the biocompatibility of materials like chitosan.
7. Systems Vaccinology: Mapping “molecular fingerprints” and “innate immune signatures” to predict long-term protection (Howard et al., 2019).
8. Cancer Vaccines: Using combinations like AS01 to overcome tumor-induced immune evasion.
9. Data Sharing: Global initiatives to increase the reproducibility of adjuvant research.
8. The Big Picture of Vaccine Success
The integration of adjuvants into modern medicine is governed by rigorous Pharmacovigilance. Regulatory standards require safety databases of at least 3,000 subjects for new adjuvanted formulations to detect rare events—such as the 2009 Pandemrix case, where a specific H1N1 vaccine was linked to narcolepsy in certain genetic populations. This oversight ensures that “help” is always balanced with safety.

The Adjuvant’s Toolkit: 5 Primary Benefits
• Dose Sparing: Using smaller amounts of antigen or fewer total doses to achieve immunity, a vital strategy during global supply shortages.
• Broadening the Immune Response: Enhancing the body’s ability to recognize various related strains of a pathogen.
• Targeting Specific Populations: Optimizing the response for “low-responder” groups, such as the elderly, infants, or the immunocompromised.
• Modulating the Defense: Tuning the immune system to produce a specific type of protection, such as choosing between a humoral (antibody) response or a cell-mediated (killer T-cell) response.
• Inducing Long-term Memory: Promoting the production of memory T-cells that ensure a rapid defense if the pathogen is encountered again years later.
9. Conclusion: The Critical Balance of Efficacy and Safety
Adjuvants remain indispensable for modern vaccinology, providing the “signal” required to transform a simple protein into a potent immunogen. They allow for critical dose-sparing and the targeting of specific immune pathways (Th1/Th2/Th17). However, the molecular impact of these substances—from the physicochemical microenvironment to the MHC II peptide repertoire—must be meticulously characterized to meet stringent EU regulatory standards.

Ongoing toxicological research into “molecular fingerprints” is essential. By identifying the specific innate immune signatures associated with both efficacy and adverse events, the industry can move beyond the limitations of older adjuvant technologies toward a safer, data-driven era of vaccine development.
From Alexander Glenny’s “dirty” batches to the precision-tuned nanoparticles of today, adjuvants have fundamentally improved the safety and potency of global immunization, saving millions of lives by providing the immune system with a sophisticated and necessary “helping hand.”
Image Summary







Questions/Answers
1. How do different vaccine adjuvants physically trigger our immune system?
Vaccine adjuvants physically trigger the immune system through two primary categories of action: acting as delivery systems that provide physical structures for antigen presentation and acting as immune potentiators that bind to specific cellular receptors. These substances are essential because purified subunit antigens are often poorly immunogenic on their own.
Physical Delivery and Structural Mimicry
• Depot Formation and Sustained Release: Some adjuvants, like aluminum salts and certain emulsions, were historically believed to work primarily by creating an antigen depot at the injection site. This physical reservoir allows for the slow, sustained release of antigens, providing the immune system with prolonged exposure to the threat.
• Structural Mimicry: Virus-like particles (VLPs) and virosomes physically mimic the size, shape, and repetitive molecular organization of real viruses but lack infectious genetic material. Their highly ordered spatial structure allows them to effectively cross-link B-cell receptors (BCRs), which triggers strong B-cell activation even without T-cell help.
• Lymph Node Trafficking: Adjuvants in the 20–200 nm size range, such as VLPs and some nanoemulsions, are physically optimized to enter the lymphatic vessels via passive diffusion. Some synthetic delivery systems use “albumin-hitchhiking,” physically binding to endogenous albumin to be transported directly to the lymph nodes.
Inducing Cellular Stress and Damage (DAMPs)
• Aluminum Salts (Alum): Alum physically interacts with the immune system by forming porous aggregates that are phagocytosed by antigen-presenting cells (APCs). This process causes lysosomal destabilization and the release of cathepsin B into the cytosol, which physically triggers the NLRP3 inflammasome. Alum also induces necrotic cell death at the injection site, causing the release of damage-associated molecular patterns (DAMPs) such as host DNA and uric acid, which act as endogenous danger signals to recruit immune cells.
• Emulsions (MF59 and AS03): These oil-in-water emulsions trigger a transient release of ATP from muscle cells, which functions as a physical signal for immune cell recruitment. AS03 also induces endoplasmic reticulum (ER) stress, which activates the unfolded protein response (UPR) and is required for the production of high-avidity antibodies.
• Saponins (QS-21): Found in the AS01 system, QS-21 physically disrupts lysosomal membranes upon endocytosis, activating caspase-1 and promoting the production of pro-inflammatory cytokines like IL-1β and IL-18.
Direct Binding to Pattern Recognition Receptors (PRRs)
• TLR Agonists: Many modern adjuvants are synthetic ligands that physically bind to Pattern Recognition Receptors (PRRs), which the immune system uses to detect pathogens.
◦ TLR4 Agonists (MPL): Physically interact with surface or endosomal receptors to trigger MyD88 and TRIF signaling pathways, promoting Th1-type responses.
◦ TLR3 Agonists (Poly I:C): Physically mimic double-stranded viral RNA to activate endosomal TLR3 and cytosolic MDA5.
◦ TLR9 Agonists (CpG): These unmethylated DNA motifs physically target endosomal TLR9, inducing type I interferon production and strong cellular immunity.
• cGAS-STING Pathway: Some adjuvants, including those using manganese, physically activate the cGAS enzyme in the cytoplasm, which then stimulates STING to induce robust interferon responses and CD8+ T-cell activity.
Selective Antigen Presentation
Recent research indicates that adjuvants like MPLA and CpG physically impact the antigen processing stage. They can alter which specific regions of an antigen are targeted, favoring the presentation of low-affinity peptides on MHC II molecules, which can focus and fine-tune the resulting T-cell response.
2. What are the safety differences between traditional and new-age adjuvants?
Safety differences between traditional and new-age (novel) adjuvants center on their long-term track records, the intensity of their local and systemic reactogenicity, and specific rare adverse events associated with their mechanisms of action.
Traditional Adjuvants
Traditional adjuvants, primarily aluminum salts (alum) and Freund’s adjuvants, are characterized by decades of use and a safety profile largely established through empirical development.
• Aluminum Salts: Alum has been the most widely used adjuvant since the 1920s and is regarded as having an excellent safety track record. Side effects are typically mild and transient, consisting of local pain, erythema, and occasional granuloma or sterile abscess formation at the injection site. While some debate exists regarding its potential neurotoxicity, no certain evidence links alum at recommended doses to neurodegenerative diseases.
• Freund’s Adjuvants: These are highly reactogenic and largely considered too toxic for human use in their complete form (CFA). CFA can induce ulcerative necrosis, painful local inflammation, and sterile abscesses. Incomplete Freund’s Adjuvant (IFA) was used in human flu vaccines in the 1950s but was hampered by severe side effects; a WHO survey noted sterile abscesses in 40,000 out of one million immunized individuals.
New-Age Adjuvants
New-age adjuvants include oil-in-water emulsions (MF59, AS03), Adjuvant Systems (AS01, AS04), and TLR agonists (CpG 1018). These are generally well-tolerated but are often more reactogenic than alum.
• Reactogenicity and Systemic Effects: Emulsions like MF59 and AS03 are associated with a higher risk of transient local pain and fatigue compared to unadjuvanted or alum-adjuvanted vaccines. Systemic reactions can include fever, malaise, and headache.
• Specific Rare Risks (Narcolepsy): A notable safety concern arose with the AS03-adjuvanted pandemic H1N1 vaccine (Pandemrix), which was associated with rare cases of narcolepsy in children and adolescents in specific regions like Sweden and Finland. However, subsequent studies suggested this might have been caused by a specific viral nucleoprotein rather than the AS03 adjuvant itself.
• Toxicity Management: Modern adjuvant design allows for the neutralization of inherent toxicities. For example, the saponin QS-21 used in AS01 has a potent hemolytic effect (lysing red blood cells), but this is abrogated by encapsulating it in liposomes with cholesterol, making it safe for human use.
• Targeted Safety: Synthetic TLR agonists like GLA-SE and GLA-AF are designed for high potency with low toxicity and limited side effects compared to parental microbial molecules like LPS.
Common and Evolving Concerns
Both traditional and new-age adjuvants have been linked to ASIA syndrome (autoimmune/inflammatory syndrome induced by adjuvants) in genetically susceptible individuals. This syndrome can manifest as myalgia, chronic fatigue, and sleep disturbances following exposure to adjuvants like alum, silicone, or mineral oil. However, some studies argue that these associations are spurious and that the clinical benefits of vaccination far outweigh these rare risks.
To ensure continued safety, new-age adjuvants undergo rigorous postmarket safety monitoring (Phase IV) to evaluate rare adverse reactions that might not appear in initial clinical trials.
3. How do Alum and emulsions trigger cell stress signals?
Alum and oil-in-water emulsions trigger the immune system by inducing specific cell stress signals and tissue damage, rather than relying solely on the direct activation of pathogen-recognition receptors (PRRs). These signals, often referred to as damage-associated molecular patterns (DAMPs) or “danger signals,” create an immunocompetent environment that recruits and activates immune cells.
Alum (Aluminum Salts)
Alum induces cell stress and damage through several physical and biochemical pathways:
• Lysosomal Destabilization: When antigen-presenting cells (APCs) phagocytose alum-antigen complexes, the crystalline particles cause lysosomal rupture. This destabilization releases the protease cathepsin B into the cytosol, which physically triggers the NLRP3 inflammasome.
• Release of DAMPs: Alum causes local cell death at the injection site, resulting in the release of endogenous danger signals such as uric acid and host cell DNA. Uric acid, in particular, acts as a potent signal for the recruitment of inflammatory dendritic cells.
• Membrane Perturbation: Alum interacts with the lipids of dendritic cell (DC) membranes without being absorbed. This interaction causes lipid reordering and “lipid sorting,” which activates the Syk and PI3K signaling pathways, eventually leading to enhanced antigen uptake and MHC class II expression.
Emulsion Adjuvants (AS03 and MF59)
Emulsions trigger stress signals primarily through metabolic disruptions and cell death pathways:
• Endoplasmic Reticulum (ER) Stress (AS03): AS03 induces a rapid downregulation of genes involved in lipid metabolism in draining lymph nodes. This leads to ER stress and activates the unfolded protein response (UPR) pathway. This specific stress response, mediated by the sensor kinase IRE1α, is required for the production of high-avidity antibodies and somatic hypermutation.
• Transient ATP Release (MF59): Intramuscular injection of MF59 triggers the transient extracellular release of ATP from muscle cells. This ATP functions as a physical stress signal that is essential for the recruitment of innate immune cells and the subsequent adaptive response; neutralizing this ATP with apyrase has been shown to reduce the adjuvant’s effectiveness.
• Necroptosis and RIPK3 Signaling: Squalene-based emulsions like MF59 induce RIPK3-dependent necroptosis (a form of programmed inflammatory cell death) in lymph node macrophages. This process is critical for stimulating antigen-specific CD8+ T-cell responses in various tissues.
While both categories of adjuvants induce localized inflammation, they use distinct stress “codes”: Alum relies heavily on lysosomal damage and uric acid release, while emulsions like AS03 and MF59 utilize ER stress and ATP signaling to mobilize the immune system.
4. How do adjuvants like MPLA fine-tune T-cell responses?
Adjuvants like monophosphoryl lipid A (MPLA) fine-tune T-cell responses by selectively activating specific innate signaling pathways and physically altering how antigens are processed and presented to the adaptive immune system. As a detoxified derivative of lipopolysaccharide (LPS), MPLA acts as a potent Toll-like receptor 4 (TLR4) agonist.
Selective Signaling and Th1 Polarization
Unlike parental LPS, which triggers both the MyD88 and TRIF pathways, MPLA preferentially activates the TRIF-biased signaling pathway. This biased signaling induces a specific cytokine profile characterized by the production of interleukin-12 (IL-12) and interferon-gamma (IFN-γ), which are the primary drivers for polarizing naive T cells into Th1-type effector cells. This fine-tuning is essential for generating the cell-mediated immunity required to combat intracellular pathogens and tumors.
Altering the MHC II Peptide Repertoire
Recent research indicates that MPLA fine-tunes the specificity of the immune response by influencing antigen processing within antigen-presenting cells (APCs).
• Narrowing the Target: In the presence of MPLA, APCs tend to present a narrower repertoire of peptide sequences on their surface MHC II molecules.
• Low-Affinity Selection: MPLA shifts the presentation toward low-affinity peptides for MHC II, which typically require less concentration to activate CD4+ T cells compared to high-affinity peptides.
• Focusing the Response: This selective presentation complexes with T-cell receptors (TCRs) to provide stronger signals, thereby focusing the T-cell response on specific protein sites that might otherwise be ignored.
Clonal Selection and TCR Affinity
Adjuvants like MPLA can modulate the quality of the T-cell repertoire by altering the selection thresholds for T-cell clones. By providing a tailored innate stimulus, they encourage the expansion of T-cell clones with higher TCR affinity and improved functional capacity.
Synergistic Adjuvant Systems
MPLA is often used in combination with other substances to further refine the T-cell message:
• AS04: By adsorbing MPLA onto aluminum salts, the adjuvant prolongs the local cellular responses initiated by TLR4 activation, extending the window for T-cell priming.
• AS01: In this system, MPLA is co-formulated with the saponin QS-21 in liposomes, creating a synergy that induces polyfunctional CD4+ T cells,. These cells are particularly effective because they can simultaneously secrete multiple cytokines, such as IL-2 and IFN-γ, which are critical for overcoming age-related immune senescence.
5. How do nanoparticles help deliver antigens to lymph nodes?
Nanoparticles facilitate the delivery of antigens to lymph nodes through several distinct physical and biological mechanisms, serving as both carriers and specialized targeting systems.
Trafficking via Passive Diffusion
Nanoparticles of specific sizes are physically optimized to bypass the need for active cellular transport.
• Size Optimization: Particles in the 20–100 nm (or up to 200 nm) range are small enough to enter the afferent lymphatic vessels via passive diffusion. If particles are too small (under 5 nm), they tend to enter the capillaries, while those that are too large (over 200 nm) are often physically blocked by the interstitial matrix.
• Surface Charge and Hydrophilicity: Because the interstitial matrix is primarily composed of negatively charged fibers, nanoparticles engineered with a net negative charge move more efficiently toward lymph nodes. Additionally, modifying the surface with polyethylene glycol (PEGylation) increases hydrophilicity, which significantly enhances the accumulation of the vaccine payload in the draining lymph nodes.
Active Targeting and Retention
Specific nanoparticle designs ensure that antigens do not just reach the lymph node but are delivered to the correct immune cells.
• Targeting Follicular Dendritic Cells (FDCs): Nanoparticles between 50 and 100 nm are physically sized to be preferentially retained by FDCs within the lymph node follicles. This retention is critical for the long-term affinity maturation of B cells.
• Structural Shuttling: Nanoparticles displaying glycosylated antigens can engage with mannose-binding lectins (MBL), which act as a molecular shuttle to rapidly transport the particles to the germinal centers of the lymph nodes.
• B-Cell Activation: Due to their highly ordered and repetitive spatial structure, virus-like particles (VLPs) can cross-link B-cell receptors (BCRs) more effectively than soluble antigens, triggering robust activation once they arrive in the node.
Cell-Mediated Transport (The “APC Train”)
Many adjuvanted nanoparticles work by recruiting the body’s own cells to act as delivery vehicles.
• Leukocyte Recruitment: Adjuvants like MF59 and AS03 trigger the release of chemokines (such as CCL2 and CXCL8) at the injection site.
• Migration to Nodes: These chemokines recruit monocytes, neutrophils, and dendritic cells (DCs), which take up the antigen-nanoparticle complex. These “loaded” antigen-presenting cells (APCs) then physically migrate through the lymphatic system into the draining lymph nodes, where they prime T cells.
Albumin-Hitchhiking
Some synthetic nanoparticles use a specialized chemical strategy to “hitch a ride” on endogenous proteins.
• By attaching lipophilic tails (such as DSPE) to the antigen, the nanoparticles can physically bind to endogenous albumin in the interstitial fluid.
• Since albumin naturally circulates through the lymphatic system, it acts as a carrier, transporting the vaccine components directly into the lymph nodes.
Bypassing Physical Barriers
Specialized formulations like nanoemulsions utilize physical effects to reach the lymphatic system.
• Occlusive Effect: Nanoemulsions provide an occlusive layer on the skin that increases local hydration and maceration, physically disrupting the stratum corneum.
• Penetration Enhancers: Surfactants within the nanoemulsion act as penetration enhancers, facilitating the uptake of outer membrane vesicles by epidermal and transfollicular routes to reach the dermis, where they are captured by migratory DCs.
6. How do liposomes make new-age adjuvants like QS-21 safer?
Liposomes make new-age adjuvants like QS-21 safer primarily by encapsulating the potent immunostimulant to neutralize its inherent toxicities while facilitating targeted delivery to immune cells.
Neutralization of Hemolytic Effects
The saponin QS-21, while highly effective, is known to have a potent hemolytic effect, meaning it can cause the lysis of red blood cells and induce significant cell death if administered in a free form. When formulated within liposomes, this hemolytic activity is completely abrogated, preventing the destruction of red blood cells and associated tissue damage at the injection site.
Quenching Reactogenicity with Cholesterol
Liposomes used in systems like AS01 include cholesterol, which plays a critical role in safety by binding directly to QS-21. This binding effectively “quenches” or quells the inherent reactogenicity of the saponin, significantly reducing the risk of both local and systemic adverse reactions that might otherwise occur. By sequestering the molecule within the phospholipid bilayer, the liposome ensures the immune system is stimulated without the harsh side effects associated with raw saponin extracts.
Targeted and Controlled Delivery
Liposomes function as a sophisticated delivery vehicle that ensures the vaccine components reach the correct cellular targets safely:
• APC Targeting: The liposomal formulation facilitates the entry of QS-21 into cells through cholesterol-dependent endocytosis, specifically targeting it to antigen-presenting cells (APCs) like macrophages and dendritic cells.
• Synergistic Safety: By co-delivering multiple immunostimulants (such as MPL and QS-21) to the same cell population, liposomes allow these components to work synergistically. This synergy means that a lower, safer dose of each component can be used to achieve a robust immune response that would otherwise require higher, more toxic doses if used individually.
• Localized Action: The particulate nature of the liposome helps localize the immune signal to the injection site and the draining lymph nodes, preventing the potent chemicals from spreading systemically and causing widespread inflammation.
7. What are the common symptoms of ASIA syndrome?
ASIA syndrome (Autoimmune/Inflammatory Syndrome Induced by Adjuvants) manifests through a variety of immune-mediated disorders that typically occur in genetically susceptible individuals following exposure to adjuvants such as aluminum salts, silicone, or mineral oil.
According to the diagnostic framework described in the sources, the common symptoms and clinical manifestations are categorized into major and minor criteria:
Common Physical Symptoms (Major Criteria)
The most frequently reported clinical manifestations used to aid in the diagnosis of ASIA syndrome include:
• Musculoskeletal pain: Specifically myalgia (muscle pain), myositis (muscle inflammation), arthralgia (joint pain), and arthritis.
• Neurological and cognitive issues: Including memory loss, demyelination, and sleep disturbances.
• Systemic effects: Such as chronic fatigue, pyrexia (fever), and dry mouth.
Clinical and Laboratory Findings (Minor Criteria)
In addition to physical symptoms, the syndrome is associated with several clinical markers:
• Autoantibodies: The appearance of autoantibodies or specific antibodies directed against the triggering adjuvant (e.g., 48.2% of patients in one study were ANA positive).
• Specific Genetic Markers: The presence of particular HLA patterns, such as HLA DRB1 and HLA DQB1.
• Evolution of Autoimmune Diseases: The progression into defined conditions like multiple sclerosis, systemic sclerosis, Sjögren syndrome, or undifferentiated connective tissue disease (UCTD).
The sources note that symptoms often improve once the triggering adjuvant is removed, although the association between these symptoms and adjuvanted vaccines remains a subject of ongoing debate and is considered by some researchers to be spurious or the result of random events rather than a direct causal relationship.
8. How do adjuvants like CpG and MPLA fine-tune which antigens are presented?
Adjuvants like CpG and MPLA (which act as pattern recognition receptor agonists) fine-tune the immune response by altering the repertoire and affinity of the peptides presented to T cells by antigen-presenting cells (APCs).
Shifting Peptide Selection and Affinity
Recent research involving H. pylori protein antigens demonstrates that the presence of these adjuvants significantly changes which protein sites are reactive with T cells.
• Low-Affinity Selection: When APCs are exposed to antigens in the presence of CpG or MPLA, they are more likely to present peptide antigens with low affinity for MHC II molecules.
• Repertoire Narrowing: These adjuvants cause APCs to present a lower number of different peptide sequences. In contrast, unadjuvanted vaccines result in a broader peptide repertoire, primarily favoring those with high affinity for MHC II.
Focusing the T-Cell Response
This selection process acts to focus the CD4+ T-cell response on a narrower array of specific protein sites.
• Enhanced Activation: Despite their lower molecular interaction strength, these low-affinity complexes can provide stronger signals to T-cell receptors, promoting more effective activation.
• Lower Antigen Requirements: It has been observed that significantly less peptide is needed to activate T cells when low-affinity peptides are presented compared to responses elicited by high-affinity peptides.
Influencing Intracellular Processing
Crucially, this fine-tuning is not caused by differences in which antigens the APCs initially take up. Instead, adjuvants like CpG and MPLA physically influence the internal antigen processing steps. This suggests that pattern recognition receptor signaling impacts how the cell breaks down and selects specific fragments of a protein for display, thereby refining the specificity and quality of the resulting adaptive immune response.
9. How do AS01 liposomes reduce the reactogenicity of QS-21?
AS01 liposomes reduce the reactogenicity of the saponin component QS-21 primarily by sequestering it within a cholesterol-containing phospholipid bilayer, which neutralizes its inherent toxicity.
According to the sources, these liposomes mitigate adverse effects through the following mechanisms:
Binding and Quenching with Cholesterol
The most critical factor in reducing reactogenicity is the inclusion of cholesterol in the liposomal formulation. Cholesterol binds directly to QS-21, a process that effectively “quenches” or quells the saponin’s natural reactivity. This prevents the molecule from interacting destructively with other cellular membranes in the body.
Abrogation of Hemolytic Effects
In its free form, QS-21 is known to have a potent hemolytic effect, meaning it causes the lysis (destruction) of red blood cells and significant local cell death. When encapsulated within AS01 liposomes, this hemolytic activity is completely abrogated, preventing tissue damage at the site of injection.
Targeted Delivery to APCs
Liposomes act as a sophisticated delivery vehicle that facilitates the entry of QS-21 into antigen-presenting cells (APCs), such as dendritic cells and macrophages, through cholesterol-dependent endocytosis. By ensuring the adjuvant is internalized by the correct immune cells before it can trigger broader inflammatory responses, the liposomes localize the immune signal and reduce both local and systemic reactogenicity.
Synergistic Dose Reduction
The liposomal structure allows for the co-delivery of QS-21 and MPL (monophosphoryl lipid A) to the same cell populations. This co-delivery creates a synergy of innate activation that is far more potent than either component used alone. Because of this synergy, a lower and thus better-tolerated dose of each immunostimulant can be used to achieve a robust protective immune response that would otherwise require higher, more toxic concentrations.
10. How do adjuvants like CpG and MPLA focus T-cell targets?
Adjuvants like CpG and MPLA (monophosphoryl lipid A) focus T-cell targets by directly activating Pattern Recognition Receptors (PRRs) on antigen-presenting cells (APCs) to alter the specificity and quality of the resulting immune response. Research involving H. pylori antigens demonstrates that these adjuvants physically influence intracellular antigen processing rather than just changing how much antigen is initially taken up by the cell.
Key mechanisms for focusing these targets include:
• Peptide Repertoire Narrowing: In the presence of CpG or MPLA, APCs tend to present a narrower repertoire of peptide sequences on their surface MHC II molecules. Without these adjuvants, vaccines typically result in a broader, less focused array of presented peptides.
• Low-Affinity Selection: These adjuvants shift the selection process to favor peptide antigens with low affinity for MHC II molecules. While unadjuvanted vaccines favor high-affinity peptides, the low-affinity complexes selected by CpG and MPLA can actually provide stronger signals to T-cell receptors (TCRs), promoting more effective activation.
• Lower Antigen Requirements: Under the influence of these adjuvants, significantly less peptide is required to activate CD4+ T cells compared to the amounts needed when high-affinity peptides are presented in unadjuvanted conditions.
• Clonal Selection and TCR Affinity: By providing tailored innate stimuli, these adjuvants modulate the quality of the T-cell repertoire, encouraging the expansion of T-cell clones with higher TCR affinity and focused specificity.
• Th1 Polarization: MPLA specifically targets TLR4 via biased TRIF signaling to induce IL-12 and IFN-γ, which focuses the immune system on generating Th1-type effector cells necessary for combating intracellular pathogens. Similarly, CpG targets TLR9 to produce a proinflammatory environment that focuses the response toward Th1 cells and cytotoxic T lymphocytes (CTLs).
11. How do albumin-hitchhiking nanoparticles target lymph nodes?
Albumin-hitchhiking is a delivery strategy where vaccine components target lymph nodes by binding to endogenous albumin proteins that naturally circulate through the lymphatic system. To facilitate this process, antigens or nanoparticles are engineered with lipophilic tails, such as 1,2-distearoyl-sn-glycero-3-phosphoethanolamine (DSPE), or specialized dyes like Evans blue derivatives. Once these modified components are administered, they physically bind to albumin in the interstitial fluid, which then functions as a carrier to transport the vaccine payload directly into the draining lymph nodes.
This approach is highly effective because it results in a heavy accumulation of vaccine components within the nodes, which can increase T-cell production by up to 30-fold and significantly enhance anti-tumor immunity. Furthermore, albumin can be utilized as a mucosal vaccine chaperone; for instance, when paired with an adjuvant like CpG, this strategy has been shown to promote the generation of lung-resident memory T cells.
12. Does nanoparticle size affect long-term B-cell memory?
Nanoparticle size significantly affects long-term B-cell memory by determining how efficiently antigens are transported to and retained within the lymph nodes.
The sources identify the following size-dependent factors as critical to humoral immunity and memory:
Follicular Dendritic Cell (FDC) Retention
• Optimal Size Range (50–100 nm): Nanoparticles designed within this specific range are preferentially retained by FDCs within the lymph node follicles.
• Significance for Memory: This retention is described as “critical” for the long-term affinity maturation of B cells and the generation of durable antibody responses. This environment allows B cells to undergo multiple rounds of selection, leading to high-affinity memory B cells and long-lived plasma cells (LLPCs).
Lymphatic Trafficking and Diffusion
• Passive Diffusion (20–100 nm): Particles in this size range are physically optimized to enter afferent lymphatic vessels via passive diffusion.
• Size Constraints: If particles are too small (under 5 nm), they tend to enter the blood capillaries rather than the lymphatic system. Conversely, if they are too large (over 200 nm), they are often physically blocked by the interstitial matrix or filtered out before reaching the necessary immunological inducer sites.
B-Cell Receptor (BCR) Activation
• Multivalent Display: Nanoparticles (such as virus-like particles) with highly ordered and repetitive spatial structures effectively cross-link BCRs. This structural mimicry of real viruses can trigger robust B-cell activation and proliferation even without T-cell help.
• Durable Responses: Specific formulations, such as the TLR7/8 agonist 3M-052 delivered via nanoparticles, have been shown to induce high magnitudes of germinal center responses and LLPCs that persist in the bone marrow for at least 70 weeks in non-human primates.
Indirect Effects of Larger Aggregates
While individual nanoparticles are essential for trafficking, some adjuvants like alum form larger, porous aggregates (up to 20 µm) that act differently. Rather than diffusing passively, these larger structures may rely on recruitment of migratory dendritic cells to carry the antigen to the lymph nodes, or they may utilize “hitchhiking” mechanisms where antigens bind to endogenous proteins like albumin to gain entry to the lymphatic system.
13. Can nanoemulsions deliver vaccines through the skin?
Nanoemulsions are capable of delivering vaccines through the skin by facilitating the penetration of antigens across the stratum corneum, the skin’s primary barrier. While the skin is recognized as an excellent vaccination site due to its complex network of antigen-presenting cells (APCs), most vaccines struggle to cross the skin barrier without physical or chemical disruption. Nanoemulsions overcome this hurdle through several physical and biological mechanisms:
• Occlusive Effect and Skin Maceration: Nanoemulsions provide an occlusive layer on the skin that increases local hydration. This hydration can induce skin maceration, which helps break down the “impermeable” properties of the stratum corneum.
• Penetration Enhancers: These formulations contain high amounts of surfactants (such as Labrasol® and Plurol® oleique) that act as penetration enhancers. These chemicals alter lipid and polar pathways in the skin, disrupting its structure to facilitate antigen transport.
• Bypassing the Stratum Corneum: Antigens in a nanoemulsion can bypass the outer layers of the skin via the pilosebaceous apparatus (hair follicle pathway). Immunohistochemistry analysis has confirmed that bacterial vesicles in nanoemulsions can reach the dermis and epithelial cells surrounding hair follicles within four hours of application.
• Structural Deformability: The “liposome-like” structure of certain antigens, such as outer membrane vesicles, may offer elastic properties that allow them to squeeze through the intracellular sealing lipids of the stratum corneum when delivered in a nanoemulsion vehicle.
In experimental studies, the topical application of outer membrane antigens from Salmonella enterica in a nanoemulsion induced a clear specific antibody response and significantly increased IFN-γ levels, whereas simple ointments or aqueous suspensions were ineffective. However, the delivery efficiency is limited by the size and nature of the payload; while small vesicles (20–100 nm) penetrate effectively, larger or non-deformable nanoparticles (around 230 nm) may still be hampered by their size and fail to reach the necessary immunological inducer sites.
14. How does cholesterol specifically help reduce adjuvant reactogenicity?
Cholesterol helps reduce adjuvant reactogenicity, specifically in systems like AS01, by binding to the saponin component QS-21 to sequester it within a liposomal structure. This is a vital safety mechanism because free QS-21 possesses a potent and adverse hemolytic effect, meaning it can cause the lysis of red blood cells and induce localized cell death if administered in an unencapsulated form. By incorporating cholesterol into the liposome’s phospholipid bilayer, the hemolytic activity of the saponin is completely abrogated, preventing tissue damage at the site of injection.
This specific interaction is described as “quenching” the reactogenicity of the adjuvant, which significantly lowers the risk of both local and systemic adverse reactions. Furthermore, this formulation strategy facilitates the safe delivery of the vaccine components to antigen-presenting cells (APCs) via cholesterol-dependent endocytosis. Once internalized by these cells, the saponin can then trigger necessary innate immune responses—such as the activation of the NLRP3 inflammasome and the release of cytokines like IL-1β and IL-18—without the toxicity associated with its free form. This cholesterol-mediated stabilization also enables the synergistic co-delivery of immunostimulants like MPL and QS-21 to the same cell populations, allowing for a robust immune response at lower, better-tolerated doses.
15. Do these liposomal systems improve vaccine effectiveness in the elderly?
Liposomal adjuvant systems significantly improve vaccine effectiveness in the elderly by overcoming the physiological phenomenon of immunosenescence, which typically reduces the immune response to natural infections and standard vaccinations.
Overcoming Immunosenescence
In individuals over 40–50 years of age, immune responses to traditional vaccines begin to decline. Liposomal systems, most notably AS01, have shown a “remarkable ability” to overcome this age-related decline.
• High Efficacy: The AS01-adjuvanted recombinant varicella-zoster (shingles) vaccine, Shingrix, demonstrated an exceptionally high efficacy of 97.2% in populations aged 50 years and older.
• Virosomal Efficacy: Other liposome-like platforms, such as virosomes used in the Inflexal V influenza vaccine, have also demonstrated good efficacy specifically in elderly cohorts.
Mechanisms of Improvement
Liposomal systems improve effectiveness in older populations through targeted delivery and synergistic activation of the immune system:
• Synergistic Innate Activation: The AS01 system uses a liposomal delivery vehicle to co-deliver two different immunostimulants (MPLA and the saponin QS-21) to the same immune cell populations. This co-delivery creates a synergy of innate activation that is more potent than the individual components used alone.
• Robust T-Cell Induction: This synergy is particularly effective at inducing polyfunctional CD4+ T cells and high-quality antibody responses, which are critical for providing protection in older adults whose immune systems may otherwise respond poorly to purified subunit antigens.
• Mitochondrial Support: Recent research suggests that potent adjuvants like AS01 may help maintain mitochondrial integrity, which is essential to support vaccine-induced immunity in aging individuals.
Comparison with Traditional Adjuvants
While traditional adjuvants like alum primarily stimulate humoral (antibody) responses and can have limited efficacy in older populations, liposomal systems and other new-age adjuvants (such as the TLR9 agonist CpG 1018) are designed to induce the robust cell-mediated immunity often missing in the elderly. For example, vaccines formulated with newer adjuvants can often achieve superior antibody responses and higher rates of seroconversion compared to alum-adjuvanted or non-adjuvanted versions in this demographic.
16. What genetic factors make someone more susceptible to ASIA?
Susceptibility to ASIA syndrome (Autoimmune/Inflammatory Syndrome Induced by Adjuvants) is primarily determined by a predisposing genetic background that interacts with external triggers like aluminum salts, silicone, or infectious agents.
The following genetic and hereditary factors have been identified as contributors to this susceptibility:
• Specific HLA Antigens: The most common autoimmune background identified in ASIA patients is the simultaneous presence of the HLA-DRB1 antigen and the PTPN22 gene. Other specific genetic patterns used as clinical markers for the syndrome include HLA DQB1.
• Biological Sex: There is a higher reported incidence rate among female individuals, suggesting that sex-linked genetic or hormonal factors may influence susceptibility.
• Familial History: Individuals with a family history of autoimmune diseases are at a significantly higher risk of developing the syndrome following exposure to adjuvants.
• Personal Medical History: A higher rate of ASIA is observed in patients who have previously been diagnosed with an autoimmune disease.
• Association with Polygenic Disorders: The syndrome frequently manifests in individuals with a genetic predisposition toward polygenic autoimmune diseases, most notably Sjögren’s syndrome and undifferentiated connective tissue disease (UCTD).
While these genetic factors provide a “prime” for the immune system, the syndrome typically only manifests after the individual is exposed to an adjuvant that triggers the immune-mediated disorder.
17. How do low-affinity peptides provide stronger T-cell signals?
Adjuvants like MPLA and CpG provide stronger T-cell signals by physically altering the intracellular processing of antigens within antigen-presenting cells (APCs). This shift in processing favors the selection and presentation of a specific type of target that would otherwise be ignored.
The specific mechanisms include:
• Selective Presentation of Low-Affinity Peptides: In the presence of pattern recognition receptor (PRR) agonists like MPLA or CpG, APCs preferentially present peptide antigens with low affinity for MHC II molecules. In contrast, unadjuvanted vaccines typically favor the presentation of high-affinity peptides.
• Lower Activation Thresholds: Research indicates that the threshold for T-cell activation is influenced by these adjuvants. Despite their lower interaction strength with the MHC molecule, the complexes formed by low-affinity peptides provide stronger signals to T-cell receptors (TCRs), which more effectively promote their activation.
• Reduced Antigen Requirements: Because these low-affinity complexes deliver more potent signals, significantly less peptide is required to activate CD4+ T cells than is needed for responses elicited by high-affinity peptides.
• Focusing the Immune Response: These adjuvants cause APCs to present a narrower repertoire of peptide sequences. By restricting the number of different antigens displayed, the adjuvants focus the T-cell response on a few specific protein sites, preventing the immune response from becoming diluted across a broad array of targets.
This “fine-tuning” of the MHC II peptide repertoire is not a result of how much antigen the cell initially takes up, but rather a direct consequence of how the PRR signaling impacts the internal breakdown and selection of protein fragments for display.
18. Does MPLA also affect CD8+ T-cell antigen presentation?
Monophosphoryl lipid A (MPLA) does affect CD8+ T-cell (cytotoxic T-lymphocyte) responses, though its impact is often most significant when it is formulated within specific adjuvant systems like AS01 or AS02. While purified subunit vaccines typically struggle to induce robust cellular immunity, MPLA-based adjuvants have been shown to facilitate the cross-presentation of antigens required to trigger these responses.
Mechanisms for CD8+ T-Cell Induction
• Synergy in Adjuvant Systems: In the AS01 system, MPLA is combined with the saponin QS-21 in a liposomal formulation. This combination acts synergistically to increase the release of cytokines like IL-12 and IL-18, which promote the maturation of dendritic cells and the subsequent induction of Th1-type responses and CTLs.
• Selective Signaling: MPLA preferentially activates the TRIF-biased signaling pathway of the TLR4 receptor. Research indicates that while this promotes Th1 polarization, it also influences the differentiation quality of the resulting CD8+ T-cell pool.
• Clonal Expansion and Phenotype: Primary immunization using MPLA as an adjuvant has been observed to drive the clonal expansion of CD8+ T cells. However, these cells tend to exhibit a terminally differentiated effector phenotype, whereas parental molecules like LPS more effectively promote the development of long-term memory precursor cells.
Limitations and Challenges
• Magnitude of Response: Despite its ability to stimulate CTLs in animal models, subunit vaccines delivered with MPLA typically do not induce the high magnitude of antigen-specific CD8+ T-cell responses seen with live viral vaccines like yellow fever or smallpox.
• Human Translation: While MPLA-based adjuvants (such as AS01, AS02, and AS04) activate monocyte and myeloid dendritic cells that can present to Th1 cells, it remains unclear in humans exactly how much these specific cell populations contribute to effective cross-priming of CD8+ T cells in vivo.
• Comparison to Other Agonists: In some studies, MPLA-based formulations like AS01B were found to be more effective at inducing antigen-specific CTL responses than emulsion-based systems like AS02A, demonstrating that the delivery vehicle (liposome vs. emulsion) significantly alters the T-cell “message”.
19. Can nanoparticles be used as mucosal vaccine chaperones?
Nanoparticles can be used as mucosal vaccine chaperones to enhance the delivery, stability, and immunogenicity of antigens administered via mucosal routes, such as the nose or mouth. Because most pathogens enter the body through mucosal surfaces, these chaperones are critical for inducing local protective immunity that traditional injectable vaccines may fail to provide.
Specific Nanoparticle Chaperone Strategies
• Albumin-Hitchhiking: Antigens or nanoparticles can be engineered with lipophilic tails that physically bind to endogenous albumin. Albumin acts as a chaperone, transporting the vaccine payload through the lymphatic system to draining lymph nodes. This strategy has been shown to increase T-cell production up to 30-fold and is particularly effective at generating lung-resident memory T cells when paired with adjuvants like CpG.
• Chitosan Nanoparticles: Chitosan is a natural biopolymer with bioadhesive properties that allow it to remain in contact with mucosal surfaces for extended periods. It facilitates the transport of antigens across epithelial barriers by modulating “tight junctions” and reducing mucociliary clearance, which improves the chances of antigen uptake by antigen-presenting cells (APCs).
• Outer Membrane Vesicles (OMVs): These are “liposome-like” bacterial vesicles that are emerging as promising mucosal delivery vehicles. They are effective at bypassing the stratum corneum or mucosal barriers to reach the dermis or basal epithelial layers, where they trigger both innate and adaptive immunity.
Roles of Mucosal Chaperones
• Cargo Protection: Nanoparticles like lipid nanoparticles (LNPs) or biodegradable polymers provide a physical shield for sensitive payloads, such as mRNA or DNA. This prevents enzymatic degradation by RNA or DNA enzymes in the extracellular environment, maintaining the vaccine’s bioactivity.
• Bypassing Harsh Environments: For oral vaccines, micro- and nanoparticles can be engineered to withstand the low pH of the gastrointestinal tract. These chaperones can target antigens specifically to M cells in the intestine to facilitate stable delivery and immune activation.
• Facilitating Cross-Presentation: Some chaperones are designed to facilitate endosomal escape, allowing the antigen to enter the cell’s cytosol. This is essential for inducing the CD8+ T-cell (cytotoxic) responses necessary to combat viral infections and tumors at mucosal sites.
Prime-Pull Vaccination
Nanoparticles are also instrumental in “prime-pull” strategies, where a systemic (injectable) prime is followed by a mucosal “pull” or boost. Specialized delivery systems, such as cationic liposomes or pH-responsive nanoparticles, can help retain primed B and T cells at mucosal tissues to establish long-term, site-specific memory.
20. How do adjuvants help vaccines work in aging immune systems?
Adjuvants are a critical tool for overcoming immunosenescence, the physiological decline of the immune system in older adults that reduces the effectiveness of natural infection responses and standard vaccinations. Because the elderly often show poor responsiveness to purified subunit antigens, the sources indicate that adjuvants represent a valid tool to enhance the magnitude, breadth, and durability of their immune protection.
Overcoming Immunosenescence with Targeted Adjuvants
Specific adjuvant platforms have been engineered to address the weakened immune state of aging populations:
• MF59: This squalene-based oil-in-water emulsion was first licensed in Europe for use in influenza vaccines (Fluad) specifically for adults older than 65 years of age. It increases the effectiveness of vaccines by inducing higher antibody titers and stimulating polyfunctional cytokine-producing CD4+ T cells.
• AS01: Found in the Shingrix shingles vaccine, this adjuvant has shown a “remarkable ability” to overcome immunosenescence, achieving 97.2% efficacy in adults aged 50 and older. It works through a unique combination of a TLR4 ligand (MPL) and a saponin (QS-21) that synergistically triggers an early interferon-gamma (IFN-γ) response and high-quality T-cell activation.
• CpG 1018: Research suggests that this TLR9 agonist is significantly more effective in older adults than traditional aluminum salts, facilitating a robust Th1-type immune response that is often missing in aging immune systems.
• Virosomes: These virus-like particles have also demonstrated good efficacy and high tolerability, specifically within elderly cohorts.
Mechanisms of Enhancement in the Elderly
The sources highlight several specific ways these “new-age” adjuvants assist the aging immune system:
• Inducing Polyfunctional T Cells: Potent adjuvant systems like AS01 induce T cells that can simultaneously secrete multiple cytokines (such as IL-2, IFN-γ, and TNF), which are necessary for providing protection in populations with poor immunological responsiveness.
• Naive B-Cell Stimulation: Adjuvants like AS03 can help overcome the “original antigenic sin”—where the body preferentially reacts to old virus strains rather than new vaccine antigens—by specifically enhancing the stimulation of naive B cells.
• Recruitment and APC Activation: Emulsion adjuvants create an “immunocompetent environment” at the injection site, recruiting monocytes and macrophages that differentiate into activated dendritic cells, which then migrate to lymph nodes to more effectively prime the remaining T-cell pool,.
• Mitochondrial Support: Emerging research suggests that maintaining mitochondrial integrity is a major challenge for aging cells, and potent adjuvants may support the immune response by bolstering this cellular function.
Dose Sparing and Accessibility
In addition to increasing potency, adjuvants like AS03 allow for antigen sparing, enabling the elderly to achieve protective antibody levels with significantly lower amounts of antigen. This is particularly valuable during pandemics when vaccine supplies are limited, and a rapid, robust immune response is required to protect vulnerable, aging demographics.
21. Can liposomes improve safety by reducing local tissue damage?
Liposomes improve safety and reduce local tissue damage by encapsulating potent immunostimulants, which neutralizes their inherent toxicity and prevents them from interacting destructively with non-target cells. This protective mechanism is most notably observed in the AS01 adjuvant system, which is used in vaccines for shingles and malaria.
Neutralization of Toxic Effects
• Abrogation of Hemolysis: The saponin QS-21, a key component of AS01, possesses a potent hemolytic effect in its free form, meaning it can rupture red blood cells and cause significant localized cell death. When QS-21 is encapsulated within a liposomal structure, this hemolytic activity is completely abrogated, preventing the destruction of red blood cells and associated tissue damage at the injection site.
• Cholesterol Quenching: Liposomes used in these systems are formulated with cholesterol, which plays a vital role in safety by binding directly to the saponin. This binding effectively “quenches” the reactogenicity of the adjuvant, significantly lowering the risk of local inflammation and pain that would otherwise occur if the raw extract were administered alone.
Targeted Delivery to APCs
• Restricting Systemic Spread: Liposomes function as sophisticated delivery vehicles that ensure vaccine components are localized to the injection site and the draining lymph nodes. By preventing potent chemicals from spreading systemically, liposomes reduce the likelihood of widespread inflammatory responses and systemic reactogenicity.
• Facilitating Safe Uptake: The liposomal formulation facilitates the entry of the adjuvant into antigen-presenting cells (APCs) through cholesterol-dependent endocytosis. This ensures that the immune system is stimulated internally by the correct cell populations (like macrophages and dendritic cells) without triggering the harsh external side effects associated with raw immunostimulants.
Synergistic Dose Reduction
• Lowering Toxicity Thresholds: Liposomal systems allow for the synergistic co-delivery of multiple components, such as MPL (monophosphoryl lipid A) and QS-21. Because these components work together to create a more potent innate signal, a lower, safer dose of each can be used to achieve the same protective effect, further minimizing the potential for local tissue irritation or damage.
In summary, the particulate nature and chemical composition of liposomes—particularly the inclusion of cholesterol—act as a buffer that shields the host from the aggressive properties of individual adjuvant molecules while focusing their power on the intended immune targets.
22. How do virosomes differ from VLPs in vaccine design?
Virosomes and Virus-Like Particles (VLPs) are both advanced nanoparticle delivery systems that mimic the structure of natural viruses to enhance vaccine immunogenicity without being infectious or replicative. While they share several characteristics, they differ significantly in their composition, structural properties, and how they are manufactured.
Composition and Structural Properties
• Virosomes: These are composed of reconstituted viral envelopes consisting of membrane lipids (such as phospholipids) and viral glycoproteins, like hemagglutinin (HA) and neuraminidase (NA), but they lack the internal nucleocapsid and genetic material of the source virus. A defining feature of virosomes is their fluid phospholipid bilayer, which mimics the native viral structure.
• VLPs: These are typically formed by structural viral proteins, such as capsid or envelope proteins, that have self-assembly properties. Unlike virosomes, VLPs are characterized by a highly repetitive and rigid protein-based structure. They can be further categorized as non-enveloped (composed only of pathogen components) or enveloped (consisting of the host cell membrane combined with the target antigen).
Manufacturing Processes
• Virosomes: These are produced by dissolving the envelope of a native virus with a detergent, followed by the complete removal of the viral genetic material and non-membranous proteins. The viral membrane lipids and glycoproteins are then reassembled into particles typically ranging from 100 to 200 nm.
• VLPs: These are manufactured using recombinant DNA technology in various expression systems, such as E. coli, yeast, or mammalian cells, where the capsid proteins spontaneously self-assemble. They can also be produced using cell-free in vitro assembly systems.
Antigen Loading and Interaction
• Virosomes: Due to their fluid lipid bilayer, virosomes are highly effective at adsorbing antigens onto their surface or within their lumen through hydrophobic lipid interactions. This fluidity is considered an advantage in vaccine design because it facilitates better interactions with host cell receptors compared to more rigid structures.
• VLPs: Antigens are typically incorporated into VLPs through chemical coupling or genetic fusion to the structural proteins. While VLPs are excellent at cross-linking B-cell receptors due to their repetitive geometry, they are considered to have more limited movement than virosomes because of their rigid protein framework.
Mechanism of Immune Activation
• Virosomes: They act as a specialized delivery system that can move antigens into the cytosol of antigen-presenting cells (APCs) via receptor-mediated endocytosis and membrane fusion. This allows the antigens to be processed through both the MHC I and MHC II pathways, inducing both humoral and cytotoxic T-lymphocyte (CTL) responses.
• VLPs: These particles are highly immunogenic due to their self-adjuvanting properties, often inducing direct B-cell activation and proliferation. Like virosomes, they are also capable of inducing antigen cross-presentation to CD8+ T cells.
23. Are there virosome-based vaccines currently licensed for human use?
Yes, several virosome-based vaccines have been licensed for human use, primarily in Europe, for protection against influenza and hepatitis A.
The common licensed brands include:
• Influenza: Virosome-based flu vaccines include Inflexal V, Invivac, Isiflu, Viroflu, and Nasalflu.
• Hepatitis A: The primary brands for hepatitis A are Epaxal and Epaxal Junior.
These vaccines have been authorized for use in more than 45 countries and have been administered to over 10 million people. For instance, Inflexal V is recognized for its efficacy across all age groups, including healthy and immunocompromised children, adults, and the elderly.
However, it is important to note that while these vaccines have a long history of use and high safety profiles, one source indicates that virosomes may no longer be licensed for use in humans in certain contexts or jurisdictions.
Beyond these established vaccines, several other virosome-based formulations are currently undergoing clinical investigation for conditions such as HIV, HPV, respiratory syncytial virus (RSV), and malaria. HIV virosomal vaccines, for example, have shown acceptable outcomes in phase I clinical trials.
24. How do virosomes facilitate antigen cross-presentation to CD8+ T cells?
Virosomes facilitate antigen cross-presentation to CD8+ T cells by mimicking the natural infection process of a virus, allowing them to deliver antigens directly into the cytosol of antigen-presenting cells (APCs).
Receptor-Mediated Endocytosis
The process begins when viral glycoproteins on the virosome surface, such as hemagglutinin (HA) and sialic acid, interact with receptors on the surface of APCs. This interaction induces the cell to internalize the virosome through receptor-mediated endocytosis.
Endosomal Fusion and Cytosolic Release
Once the virosome is trapped within an endosome, the environment undergoes acidification. This low pH triggers a conformational change in the HA protein, which facilitates membrane fusion between the virosomal envelope and the endosomal membrane. This fusion allows the antigenic cargo contained within the virosome lumen to escape the endosome and enter the cytosol.
MHC Class I Pathway Activation
By delivering antigens directly into the cytosol, virosomes allow the proteins to be processed via the MHC class I pathway rather than being restricted to endosomal degradation and the MHC class II pathway. Once in the cytosol, the antigens are processed and presented on the cell surface in complex with MHC I molecules.
Induction of CTL Responses
This mechanism is critical for the induction of cytotoxic T-lymphocyte (CTL) responses, as it provides the necessary signals to activate and proliferate antigen-specific CD8+ T cells. Because they naturally deliver antigens into the cytosolic processing stream, virosomes are highly effective at inducing robust cellular immunity comparable to that of a natural viral infection.
25. Discuss about Depot effect (sustained release), in the larger context of Mechanisms of Action.
The depot effect, characterized by the formation of a localized site at the point of injection that facilitates the sustained release of vaccine antigens, is historically one of the oldest and most widely recognized mechanisms of action (MoA) for adjuvants. While its primary role has been challenged in recent years for some licensed adjuvants, it remains a critical design feature for many modern delivery systems and emerging platforms.
Historical Context and Theoretical Foundation
Traditionally, adjuvants were broadly classified as either immune potentiators (directly stimulating innate cells) or delivery systems (acting as passive vehicles). The depot effect was the hallmark of delivery systems, operating on the principle that trapping antigens and releasing them slowly ensures constant stimulation of the immune system, leading to higher antibody titers.
• Aluminum Salts (Alum): For decades, alum was believed to owe its adjuvanticity to this MoA. It was thought that soluble antigens adsorbed onto the adjuvant surface were sequestered and slowly released over 2–3 weeks.
• Water-in-Oil Emulsions: Adjuvants like Incomplete Freund’s Adjuvant (IFA) and Montanide rely heavily on forming an oily deposit that provides continuous antigen release, thereby extending the antigen’s biological lifespan at the injection site.
Challenges to the Traditional Depot Theory
Recent immunobiological research has demonstrated that for several major licensed adjuvants, the depot effect may be dispensable or secondary to direct innate immune activation.
• Alum Re-evaluation: Multiple studies show that if an “antigen-alum depot” is surgically removed shortly after immunization (ranging from 2 hours to 14 days), the resulting immune response remains unaltered. This suggests that alum’s primary MoA is likely the direct activation of the NLRP3 inflammasome and the recruitment of immune cells via localized inflammation.
• Squalene-based Emulsions (MF59 and AS03): Initially suspected of forming depots, these oil-in-water emulsions are now known to have short half-lives (e.g., 42 hours for MF59) and do not rely on sustained release at the injection site. Their efficacy instead stems from creating a transient “immunocompetent environment” that promotes rapid cell recruitment and antigen transport to lymph nodes.
Depot Effect in the Modern MoA Framework
Despite these challenges, sustained release is still considered a key design principle for specific objectives in vaccine engineering.
• Enhanced Germinal Center (GC) Maturation: Controlled release is critical for single-dose vaccines and for promoting long-term affinity maturation. By allowing B cells in germinal centers to undergo multiple rounds of selection against a persisting antigen source, sustain-release systems (like injectable hydrogels or osmotic pumps) can produce larger quantities of high-affinity antibodies.
• Prolonging Bioavailability: Modern delivery systems use sustained release to maintain sufficient levels of antigens available to antigen-presenting cells (APCs) over time. For example, biodegradable polymer particles (PLGA or polyanhydride) can be engineered to release subunit antigens over a period of days to months.
• Formation of “Immune Niches”: Some delivery systems go beyond simple release by creating an inflammatory niche at the injection site. This niche not only retains antigens but also recruits and infiltrates innate immune cells, providing local inflammatory cues that significantly amplify the adaptive response.
Adjuvants Utilizing the Depot Effect
| Adjuvant Type | Depot Characteristics |
|---|---|
| IFA / Montanide | Classic oily deposit for continuous antigen release. |
| PLGA / Polymers | Encapsulates antigens in solid particles for release over weeks/months. |
| Injectable Hydrogels | Tunable grids that allow for “pulsed” or sustained delivery to optimize GC reactions. |
| Liposomes | Certain cationic formulations (like CAF01) induce long-lasting depots required for their specific immune profile. |
| AS04 | While based on MPLA, the presence of alum is thought to prolong the immune response by stabilizing the components at the site. |
References
Apostólico, J. D. S., Lunardelli, V. A. S., Coirada, F. C., Boscardin, S. B., & Rosa, D. S. (2016). Adjuvants: Classification, Modus Operandi, and Licensing. In Journal of Immunology Research (Vol. 2016). Hindawi Limited. https://doi.org/10.1155/2016/1459394
Awate, S., Babiuk, L. A., & Mutwiri, G. (2013). Mechanisms of action of adjuvants. In Frontiers in Immunology (Vol. 4, Number MAY). https://doi.org/10.3389/fimmu.2013.00114
Christensen, S. M., Kaltenpoth, M., Vogel, H., & Vannette, R. L. (2026). Streptomyces anthophorae sp. nov. and Streptomyces nidicola sp. nov., novel actinobacteria isolated from a solitary bee. International Journal of Systematic and Evolutionary Microbiology, 76(1). https://doi.org/10.1099/ijsem.0.007029
Coffman, R. L., Sher, A., & Seder, R. A. (2010). Vaccine adjuvants: Putting innate immunity to work. In Immunity (Vol. 33, Number 4, pp. 492–503). https://doi.org/10.1016/j.immuni.2010.10.002
Cui, Y., Ho, M., Hu, Y., & Shi, Y. (2024). Vaccine adjuvants: current status, research and development, licensing, and future opportunities. In Journal of Materials Chemistry B (Vol. 12, Number 17, pp. 4118–4137). Royal Society of Chemistry. https://doi.org/10.1039/d3tb02861e
Facciolà, A., Visalli, G., Laganà, A., & Di Pietro, A. (2022). An Overview of Vaccine Adjuvants: Current Evidence and Future Perspectives. In Vaccines (Vol. 10, Number 5). MDPI. https://doi.org/10.3390/vaccines10050819
Gupta, A., & Chaphalkar, S. R. (2015). Vaccine adjuvants: The current necessity of life. In Shiraz E Medical Journal (Vol. 16, Number 7). Shriaz University of Medical Sciences. https://doi.org/10.17795/semj28061
Hauser, M. I., Muscatello, D. J., Soh, A. C. Y., Dwyer, D. E., & Turner, R. M. (2019). An indirect comparison meta-analysis of AS03 and MF59 adjuvants in pandemic influenza A(H1N1)pdm09 vaccines. In Vaccine (Vol. 37, Number 31, pp. 4246–4255). Elsevier Ltd. https://doi.org/10.1016/j.vaccine.2019.06.039
Klug, B., Celis, P., Ruepp, R., & Robertson, J. S. (2015). EU regulatory guidelines for the clinical evaluation of adjuvants. In Clinical Research and Regulatory Affairs (Vol. 32, Number 2, pp. 57–62). Taylor and Francis Ltd. https://doi.org/10.3109/10601333.2015.1001899
Lavelle, E. C., & McEntee, C. P. (2024). Vaccine adjuvants: Tailoring innate recognition to send the right message. In Immunity (Vol. 57, Number 4, pp. 772–789). Cell Press. https://doi.org/10.1016/j.immuni.2024.03.015
Li, B., Zhang, J., He, T., Yuan, H., Wu, H., Wang, P., & Wu, C. (2024). PRR adjuvants restrain high stability peptides presentation on APCs. ELife, 13. https://doi.org/10.7554/elife.99173
Marciani, D. J. (2003). Vaccine adjuvants: role and mechanisms of action in vaccine immunogenicity. Therapeutic Focus, 8(20).
Prajapati, P., & Mourya, A. (2025). Vaccine adjuvants: Insights into development, present and future perspective. International Journal of Pharmaceutical Chemistry and Analysis, 12(2), 121–130. https://doi.org/10.18231/j.ijpca.v.12.i.2.7
Pulendran, B., S. Arunachalam, P., & O’Hagan, D. T. (2021). Emerging concepts in the science of vaccine adjuvants. In Nature Reviews Drug Discovery (Vol. 20, Number 6, pp. 454–475). Nature Research. https://doi.org/10.1038/s41573-021-00163-y
Rinee, K. C., Ilavsky, J., Kuzmenco, I., Zuo, X., & Xu, A. Y. (2026). Tuning Antigen–Adjuvant Interactions by Modulating the Physicochemical Properties of Aluminum Hydroxide Nanoparticles for Improved Antigen Stability. Colloids and Interfaces, 10(1), 16. https://doi.org/10.3390/colloids10010016
Shi, S., Zhu, H., Xia, X., Liang, Z., Ma, X., & Sun, B. (2019). Vaccine adjuvants: Understanding the structure and mechanism of adjuvanticity. In Vaccine (Vol. 37, Number 24, pp. 3167–3178). Elsevier Ltd. https://doi.org/10.1016/j.vaccine.2019.04.055
Sun, Y., Gruber, M., & Matsumoto, M. (2012). Overview of global regulatory toxicology requirements for vaccines and adjuvants. Journal of Pharmacological and Toxicological Methods, 65(2), 49–57. https://doi.org/10.1016/j.vascn.2012.01.002
Tamayo, I., Gamazo, C., de Souza Rebouças, J., & Irache, J. M. (2017). Topical immunization using a nanoemulsion containing bacterial membrane antigens. Journal of Drug Delivery Science and Technology, 42, 207–214. https://doi.org/10.1016/j.jddst.2017.02.009
Verma, S. K., Mahajan, P., Singh, N. K., Gupta, A., Aggarwal, R., Rappuoli, R., & Johri, A. K. (2023). New-age vaccine adjuvants, their development, and future perspective. In Frontiers in Immunology (Vol. 14). Frontiers Media S.A. https://doi.org/10.3389/fimmu.2023.1043109
Xing, J., Zhao, X., Li, X., Fang, R., Sun, M., Zhang, Y., & Song, N. (2025). The recent advances in vaccine adjuvants. In Frontiers in Immunology (Vol. 16). Frontiers Media SA. https://doi.org/10.3389/fimmu.2025.1557415
Zhao, T., Cai, Y., Jiang, Y., He, X., Wei, Y., Yu, Y., & Tian, X. (2023). Vaccine adjuvants: mechanisms and platforms. In Signal Transduction and Targeted Therapy (Vol. 8, Number 1). Springer Nature. https://doi.org/10.1038/s41392-023-01557-7