Introduction: The Genetic Engineer’s Dilemma
Imagine you are a genetic engineer, and your job is to add a new, handwritten page into a specific book. The challenge is that you have a massive library with millions of identical books, and after your attempt, you don’t know which ones successfully received the new page.

How would you find the right ones without opening every single book? This is the core problem in molecular cloning: finding the few bacteria that have correctly taken up a new piece of DNA.

Blue-white screening is a clever, color-coded “magic trick” that solves this problem.

It’s a powerful technique that allows scientists to instantly spot the bacteria containing the modified DNA just by looking at the color of their colonies on a petri dish.

This document will break down the science behind this simple and powerful method, explaining step-by-step how it works.

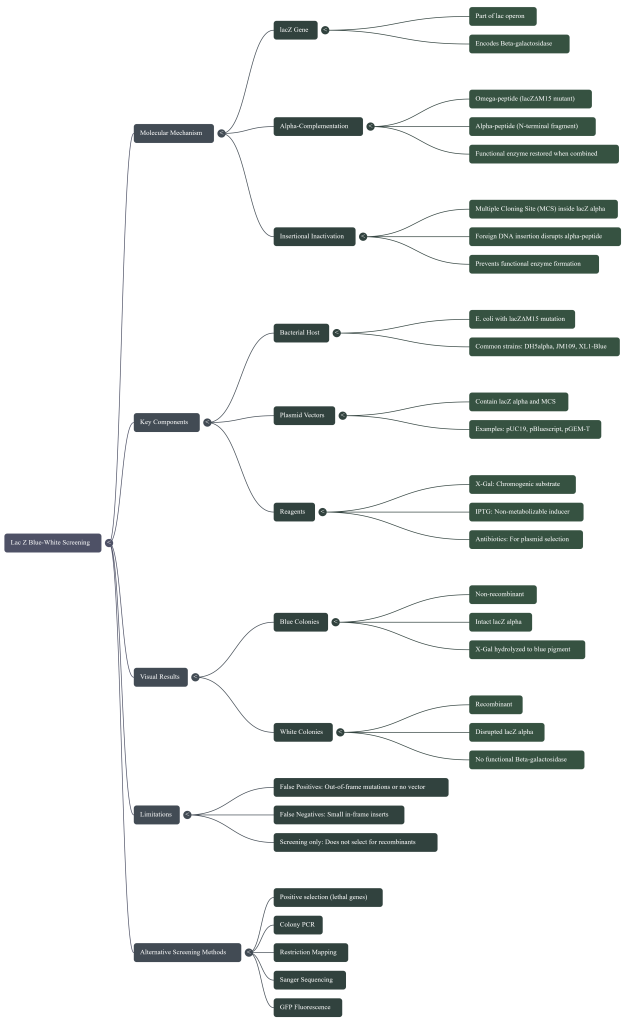
1. Meet the Key Players: The Cast of Characters in Our Genetic Story
To understand this genetic trick, we first need to meet the main characters involved in the process. Each plays a critical role in the final color change.
• The Bacteria (E. coli): Our tiny, living factory where the experiment takes place.
• The Plasmid: A small, circular instruction manual (DNA) that we can add new pages (genes) to.
• The lacZ gene: A specific recipe in the instruction manual that tells the factory how to make a special enzyme.
• β-galactosidase: The “chef” enzyme that is built by following the lacZ recipe. Its main job is to break down sugars, allowing the bacteria to use lactose as an energy source.
• X-gal: A special, colorless sugar-like ingredient. When our “chef” (β-galactosidase) “cooks” (cleaves) it, X-gal is broken down into a compound that spontaneously dimerizes to form 5,5′-dibromo-4,4′-dichloro-indigo—an insoluble, bright blue pigment.
• IPTG: The “on switch” or inducer. It’s a molecular mimic (an analog) of lactose. Its job is to bind to the system’s repressor, telling the factory to start reading the lacZ recipe at full speed.

Now that we’ve met the players, let’s look at the clever bit of genetic teamwork that makes the color change possible.
2. The Secret Handshake: Understanding α-Complementation
The magic of blue-white screening hinges on a principle called α-complementation. Think of it as a “two-part key” system needed to start the blue-color reaction. Both parts must be present and fit together perfectly for the key to work.
1. The Bacteria’s Half: The special E. coli used in the lab has a broken lacZ gene, specifically a version called lacZΔM15. This means the bacteria can only produce a defective, non-functional piece of the β-galactosidase enzyme (called the ω-peptide). This is like having only one half of the key.
2. The Plasmid’s Half: The engineered plasmid that scientists use contains the missing piece of the lacZ gene, a small fragment called lacZα. This piece produces the other half of the key (the α-peptide).

When the plasmid is successfully taken up by the bacteria, both halves of the key are present. The α-peptide from the plasmid fits together with the ω-peptide from the bacteria. They “complement” each other, assembling a complete, functional β-galactosidase enzyme. This functional enzyme is now ready to act on the X-gal ingredient and turn the colony blue.
This two-part key system is the secret, and scientists cleverly exploit it to see if their experiment was a success.
3. The Experiment: How a New Gene Creates a White Colony
The primary goal of a cloning experiment is to insert a new gene—the “gene of interest”—into the plasmid. To make blue-white screening possible, scientists designed the plasmid so that the spot where new genes are inserted (called the Multiple Cloning Site, or MCS) is located directly inside the lacZα gene—the part that makes the plasmid’s half of the key.

This clever design relies on a principle called insertional inactivation: if the new gene is successfully inserted, it physically disrupts the lacZα gene, “inactivating” it. This leads to two possible outcomes, which are compared in the table below.


| Outcome | Scenario 1: Failure (Blue Colony) | Scenario 2: Success! (White Colony) |
| What Happened? | The plasmid fails to take up the new gene and simply closes back on itself (recircularizes). | The new gene is successfully inserted into the plasmid’s MCS. |
| Effect on the lacZα Gene | The lacZα gene remains intact and its instructions are readable. | The insertion disrupts the lacZα gene’s instructions. It’s like ripping the recipe for the key in half to add a new page. |
| Resulting Enzyme | A perfect α-peptide is made. α-complementation succeeds, and a fully functional β-galactosidase enzyme is formed. | The plasmid’s half of the key (the α-peptide) cannot be made correctly. α-complementation fails, and no functional β-galactosidase is formed. |
| Interaction with X-gal | The functional enzyme breaks down X-gal, producing the blue pigment. | The non-functional enzyme cannot break down X-gal. |
| Final Colony Color | BLUE | WHITE |

With this understanding, we can now see how a scientist interprets the results on their petri dish.
4. Reading the Plate: White is Right!
After performing the experiment and letting the bacteria grow on an agar plate containing X-gal and IPTG, a scientist will see a mix of blue and white bacterial colonies. The interpretation is simple and direct:
• The Blue Colonies are the failures. They contain plasmids without the new gene. The lacZα gene is intact, a functional enzyme is made, and the colony turns blue.
• The White Colonies are the potential successes. They contain plasmids where the lacZα gene has been disrupted. This disruption strongly indicates that a new gene has been inserted.
The white colonies are the “special books” the scientist was looking for, easily identified by their color without having to check each one individually.
5. A Quick Word of Caution
Blue-white screening is an excellent screening tool, but it is not completely foolproof. Scientists must be aware of a few key limitations that can sometimes lead to misleading results.

• False Positives (a white colony with no insert): Sometimes, a white colony appears even if the desired gene wasn’t inserted. This can happen if a random mutation breaks the lacZα gene, or if the plasmid simply re-ligated incorrectly after being cut, disrupting the gene by mistake.
• Rare False Negatives (a blue colony with an insert): Very rarely, a blue colony might contain an inserted gene. This can happen if the new gene is small and inserted perfectly “in-frame” with the original lacZα recipe, creating a fusion protein that, by chance, can still function as an enzyme.

Because of these possibilities, blue-white screening is used as a first step. Scientists always use a second method—such as colony PCR, restriction enzyme analysis, or DNA sequencing—to “double-check” the white colonies and confirm they contain exactly the right insert.

Conclusion: A Colorful Shortcut to Discovery
Blue-white screening is a powerful visual method that allows scientists to rapidly distinguish between bacteria containing recombinant plasmids (with a gene inserted) and those with non-recombinant ones. By cleverly disrupting a color-producing pathway, this technique provides an immediate signal of a successful cloning event. This seemingly simple trick saves enormous amounts of time and effort, making it a fundamental and indispensable tool that helps drive genetic research forward.

Image Summary
References
https://en.wikipedia.org/wiki/Blue–white_screen
https://www.thermofisher.com/np/en/home/brands/thermo-scientific/molecular-biology/molecular-biology-learning-center/molecular-biology-resource-library/spotlight-articles/5-ways-screen-recombinant-clones.html
https://www.sigmaaldrich.com/NP/en/technical-documents/technical-article/genomics/cloning-and-expression/blue-white-screening?srsltid=AfmBOoodPguw2yIE-wP0p7GSh42SJQDkFJZNzdckso5xOAi-L1BZT1BB
https://bio.libretexts.org/Bookshelves/Biochemistry/Book%3A_Biochemistry_Free_and_Easy_(Ahern_and_Rajagopal)/09%3A_Techniques/9.14%3A_Lac_Z_Blue-White_Screening
https://blog.addgene.org/plasmids-101-blue-white-screening
https://www.goldbio.com/blogs/articles/blue-white-screening?srsltid=AfmBOooHvBFmbsZ7qRZRzAYyV0LSlBe0Dl9yCQpnOmlpCOQaDIcAn9WI







Pingback: The Genetic Engineer’s Toolkit: A Beginner’s Guide to Cloning Vectors - Aneknowledge.com
Pingback: Understanding the Types of Cloning: A Comprehensive Guide for Beginners - Aneknowledge.com