Antibody Therapy Offers Gentler Path to Stem Cell Transplantation
For decades, stem cell transplantation has offered hope to patients with bone marrow failure and blood disorders. But the standard conditioning—using irradiation or chemotherapy drugs like busulfan—comes at a steep price: severe tissue damage, infertility, heightened infection risk, and even secondary cancers.
For patients with Fanconi anemia (FA), a rare inherited DNA-repair disorder, these toxicities are especially devastating. Now, a groundbreaking phase 1b clinical trial shows that a gentler, antibody-based strategy may change the game.
Targeting Stem Cells with Precision
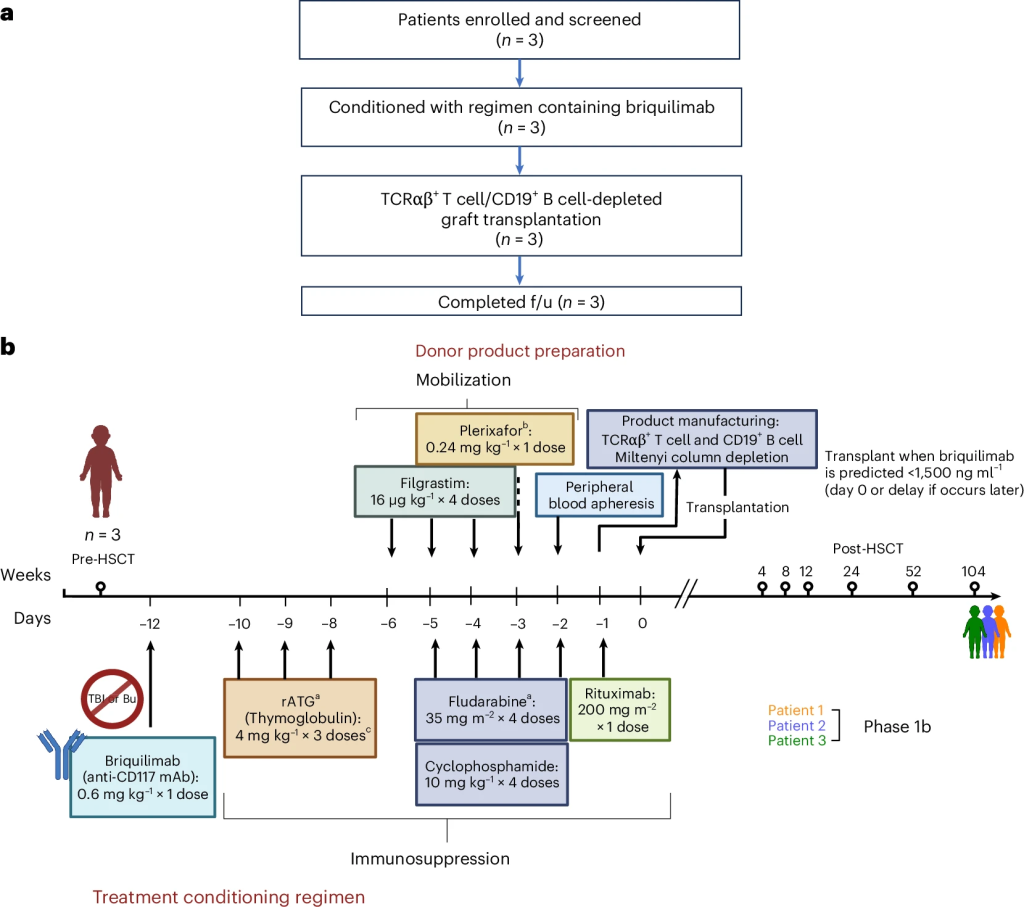
The new approach replaces genotoxic conditioning with briquilimab, an anti-CD117 antibody that selectively targets hematopoietic stem and progenitor cells. By clearing space in the bone marrow, briquilimab enables donor stem cells to engraft—without the collateral damage caused by irradiation or busulfan.
In the trial, three young FA patients received briquilimab alongside carefully calibrated immunosuppression (anti-thymocyte globulin, cyclophosphamide, fludarabine, and rituximab). They then underwent transplantation with haploidentical (half-matched family) donor stem cells that had been depleted of TCRαβ+ T cells and CD19+ B cells to reduce graft-versus-host disease (GvHD).
Results After Two Years
The outcomes were striking:
- Rapid engraftment: Neutrophil recovery in just 11–13 days.
- Durable donor chimerism: 99–100% donor cells maintained over 2 years.
- Minimal toxicity: Only mild mucositis; no veno-occlusive disease.
- No acute GvHD: Despite no pharmacological GvHD prophylaxis.
- Improved DNA repair: Chromosomal breakage defects in patient blood cells resolved after transplantation.
All three patients remain alive and well two years after treatment.
Why This Matters
This is the first trial to show irradiation- and busulfan-free conditioning can achieve successful, durable stem cell engraftment in Fanconi anemia.
- For patients: A safer transplant option with fewer long-term risks.
- For medicine: Proof-of-concept that targeted antibodies can replace genotoxic regimens.
- For the future: Potential application in other blood disorders and cancers where toxic conditioning remains a barrier.
A larger phase 2 trial is ongoing (NCT04784052), aiming to confirm safety and broaden applicability.
Conclusion
By harnessing the precision of antibodies, scientists are rewriting the rules of stem cell transplantation. For Fanconi anemia patients—and potentially many more—the era of safer, less toxic transplants may be just beginning.
Reference
Agarwal, R., Bertaina, A., Soco, C., Long-Boyle, J. R., Saini, G., Kunte, N., … & Czechowicz, A. (2025). Irradiation-and busulfan-free stem cell transplantation in Fanconi anemia using an anti-CD117 antibody: a phase 1b trial. Nature Medicine, 1-8. https://doi.org/10.1038/s41591-025-03817-1






