1. Introduction to the One Health Framework
To address the escalating crisis of antimicrobial resistance (AMR), we must pivot away from a strictly clinical perspective and adopt a systemic, planetary lens. While healthcare-associated drivers—such as over-prescribing in hospitals—are well-documented, the most potent anthropogenic drivers of resistance often originate outside the medical facility. The One Health framework provides the necessary architecture to analyze these complex, transdisciplinary threats.
One Health is an integrated, unifying approach that aims to sustainably balance and optimize the health of people, animals, and ecosystems. It recognizes that the health of humans, domestic and wild animals, plants, and the wider environment (including ecosystems) is closely linked and interdependent.
From a public health consultancy standpoint, AMR is not merely a medical failure; it is a “planetary health issue” precipitated by environmental stresses and anthropogenic biodiversity loss. When ecosystems are degraded by industrial activity, the natural checks and balances that limit microbial dominance are stripped away, allowing resistant strains to flourish. To understand why human infections are becoming increasingly untreatable, we must first analyze the massive scale of drug use in global food production.
2. The Convergence of Agriculture and Pathogen Evolution
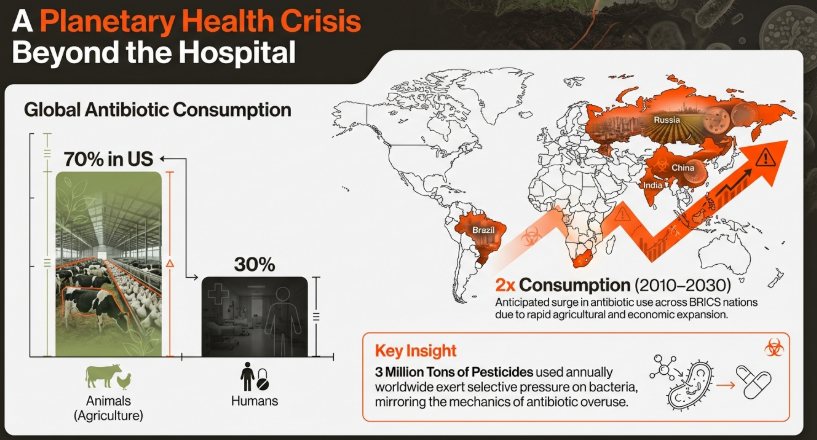
Antimicrobial resistance (AMR) has transitioned from a manageable clinical complication into a profound planetary health crisis that no longer respects the partitioning of hospital and field. According to 2019/2021 Global Burden of Disease data, drug-resistant bacteria were directly responsible for 1.14 million deaths and associated with 4.71 million deaths worldwide. If current trajectories hold, AMR is projected to directly cause 1.91 million deaths annually by 2050. While the historical focus has centered on clinical antibiotic stewardship, a “One Health” lens reveals that the health of soil, animals, and humans is inextricably linked, and that industrial food production has become a primary driver of pathogen evolution.

Table 1: Projected Increase in Antimicrobial Consumption (2010–2030)
| Region/Category | Projected Increase (%) | Strategic Metric |
|---|---|---|
| Global Agriculture | 67% | Driven by rising wealth and intensive protein demand. |
| BRICS Economies | 99% | Rapid industrialization of livestock production. |
| High-Income Countries | Baseline High | US livestock consume >70% of medically important antibiotics. |
The strategic focus must shift from “misuse of medicines” toward a deconstruction of environmental pollutants as primary selective pressures. Chemical inputs in industrial agriculture act as unseen catalysts, transforming vast landscapes into reservoirs for resistance. This transition in selection pressure suggests that even in the absence of clinical antibiotic exposure, the chemical-heavy infrastructure of global agriculture provides the precise stressors needed to cultivate multidrug-resistant (MDR) strains. Consequently, understanding the specific mechanisms through which chemical inputs alter the microbial landscape is critical to formulating effective policy.
The Scale and Purpose of Antimicrobials in Agriculture
The volume of antibiotics utilized in global agriculture is staggering, frequently eclipsing human medical consumption. In the United States, 70% of medically important antibiotics—drugs vital for treating human disease—are sold for use in livestock. This high-volume environment serves as a primary incubator for resistance.
The following table categorizes the primary agricultural uses of these agents:
| Use Type | Target Population | Primary Goal | Economic Benefit |
|---|---|---|---|
| Therapeutic | Sick animals | Treatment of diagnosed infections. | Essential for animal welfare and maintaining productivity. |
| Prophylactic | Healthy animals | Disease prevention in crowded or high-stress conditions. | High in intensive farming; prevents mass mortality in poor hygiene settings. |
| Growth Promotion | Healthy animals | Stimulating faster weight gain and feed efficiency. | Modest (<5% in high-income settings); benefits diminish as hygiene improves. |
As industrial farming techniques advance, the economic justification for growth promotion fades, yet its continued use at sub-therapeutic doses creates the ideal conditions for bacteria to develop “superpower” resistance traits.
The Colistin Alert: A Warning from the Front Lines
One of the most alarming narratives in planetary health is the story of Colistin. For doctors, Colistin is a “last-resort” drug—the final line of defense against highly aggressive Gram-negative bacteria. Because of its potential for kidney damage, it is used only when every other option has failed.
However, for years, Colistin was used routinely in Chinese pig farming as a growth promoter. In 2015, the MCR-1 gene—a piece of DNA that confers Colistin resistance—was found in 20% of sampled animals and 15% of raw meat. Most disturbing was the finding that 1% of sampled hospital patients already carried the gene.
The danger is that MCR-1 is “transferable” via plasmids (mobile genetic elements). This means resistance can leap between different species of bacteria, turning routine infections into untreatable clinical crises. This isn’t just a farm problem; it is a global security threat.
Direct and Food-Chain Pathways to Human Infection
Resistant bacteria do not remain confined to the farm; they migrate into human populations through two primary direct vectors:
• Direct Contact: This pathway poses an immediate risk to farmers, veterinarians, and slaughterhouse workers. Close proximity to livestock allows for the colonization of human hosts through physical touch or inhalation of farm-borne dust.
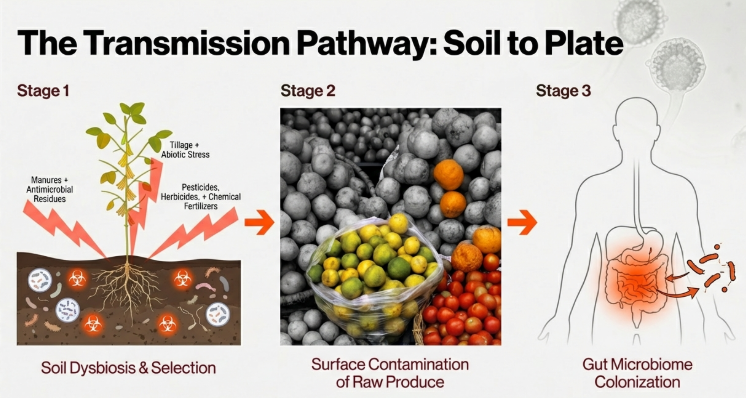
• The Food Chain: Resistant strains often contaminate animal products during slaughter and processing. Consumers are exposed through the handling of raw meat, consumption of undercooked products, or ingestion of unpasteurized dairy.
The “smoking gun” for this farm-to-human transmission was identified in a landmark Chinese study regarding the mcr-1 gene, which confers resistance to colistin—a last-resort antibiotic for humans. Researchers found the gene in 20% of animals tested and subsequently in 1% of hospital patients, providing definitive evidence that agricultural selection can directly compromise human clinical outcomes. Even without direct animal contact, resistant bacteria can still reach you via environmental hotspots.
Indirect Environmental Pathways: The “Hotspots”
Animal and industrial waste streams act as “evolutionary engines,” facilitating the emergence and global dissemination of resistance. These hotspots concentrate selective pressures far beyond natural background levels:
1. Animal Excretion: Between 75-90% of antibiotics consumed by livestock are excreted unmetabolized. When this manure is applied to crops as fertilizer, it introduces active pharmaceutical ingredients and resistant bacteria directly into the soil and human food crops.
2. Aquaculture: In fish farming, 70-80% of antibiotics are excreted directly into the water. These agents exert continuous selective pressure on aquatic microbiomes, allowing resistance to spread rapidly through water systems.
3. Manufacturing Waste: Effluent from pharmaceutical plants producing active ingredients often contains drug concentrations thousands or millions of times higher than sewage sites. These sites act as high-velocity evolutionary reservoirs where a single emergence event can rapidly escalate into a global health threat.
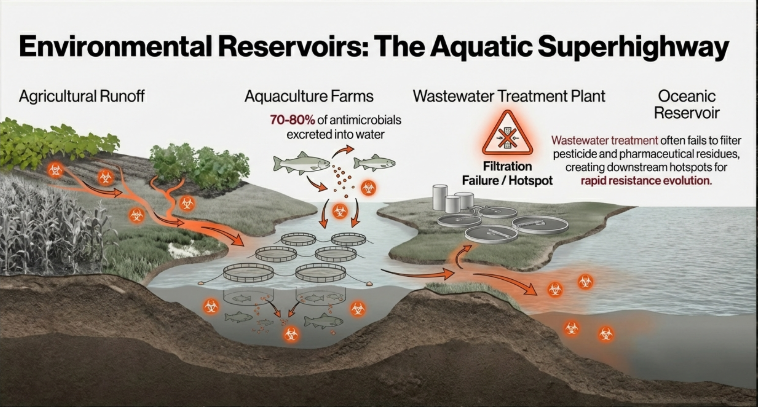
Crucially, the microbial community is shaped by more than just antibiotics; other industrial chemicals act as unseen catalysts for resistance.
The Environmental Nexus: Co-Selection and Cross-Resistance Drivers
A siloed policy approach focusing exclusively on clinical prescribing fails to account for the environment as a primary reservoir for resistance. Manufacturing discharge and agricultural runoff act as persistent reservoirs that circumvent traditional controls.
Impact of Non-Antibiotic Pollutants
The framework identifies environmental pollutants, specifically Organophosphate (OP) pesticides and Heavy Metals (HMs), as critical co-selection agents. These substances drive AMR through the following mechanisms:
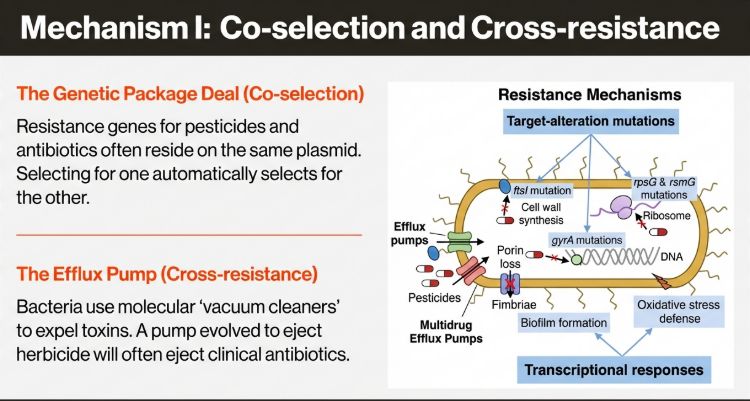
• Co-Selection and Cross-Resistance: Resistance genes for HMs and antibiotics frequently reside on the same mobile genetic elements. Exposure to metals inadvertently selects for antibiotic resistance via common efflux pumps.
• Element Correlation with β-lactamase: Statistical modeling identifies specific elements as the strongest predictors of resistance gene prevalence. Phosphorus (β0=3.935) and Iron (β0=90.85) show a significant positive correlation with β-lactamase genes. Conversely, elements such as Boron (B), Lithium (Li), Magnesium (Mg), and Sodium (Na) show negative correlations, suggesting that environmental policy must be hyper-targeted.
• Pesticide-Induced Resistance: Specific OPs like parathion exhibit a strong positive association with the prevalence of tetA and tetB genes, directly increasing the likelihood of tetracycline resistance in the absence of the drug itself.
Synthesize Manufacturing Risks
Active Pharmaceutical Ingredient (API) manufacturing waste represents a critical “hotspot” for AMR emergence. In major manufacturing hubs, antibiotic concentrations in local water courses have been recorded at 1,000 times higher than toxic levels for bacteria. This industrial pollution creates a massive selective pressure that breeds multidrug-resistant strains before they ever reach a clinical environment. Because these stressors act as negative externalities, national strategies must employ fiscal levers to force the internalization of these costs.
3. The Mechanistic Link: How Pesticides Drive Genetic Resistance
The abiotic stress induced by intensive crop management is not merely an ecological byproduct; it is a strategic driver of microbial adaptation. In 2021, global pesticide consumption reached approximately 3.54 million metric tons—a 96% increase since 1990. This volume of chemical flux represents a systemic failure in environmental partitioning, where “safe” environmental levels of residues actually serve as clinically relevant selective pressures.
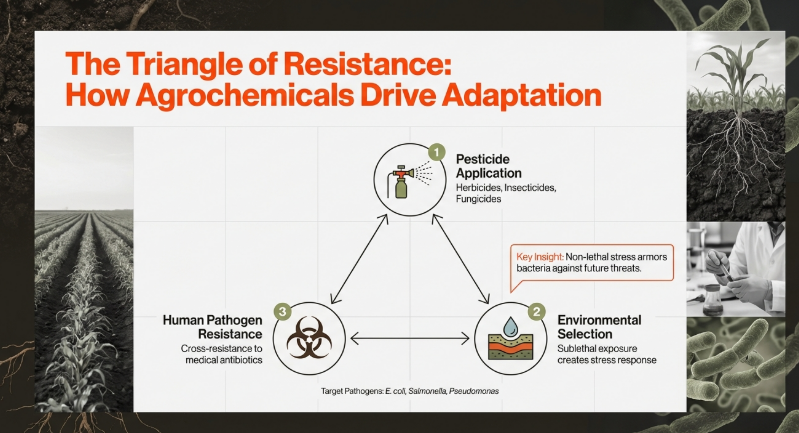
Microbial populations respond to pesticide exposure (herbicides, insecticides, and fungicides) through several unintended biological consequences:
• Efflux Pump Activation and Membrane Pore Closure: Pesticides trigger bacteria to overexpress efflux pumps. These pumps are not substance-specific; by evolving to expel toxic pesticides, bacteria simultaneously gain the ability to expel clinical antibiotics. Concurrently, bacteria decrease porin expression, closing the cellular “gates” to both toxins and treatments.
• Induction of Gene Mutations: Exposure to environmental pesticide residues stimulates the rsmG mutation. Research indicates that this specific genetic shift, occurring at concentrations common in agricultural runoff, confers resistance to streptomycin, a critical clinical antibiotic.
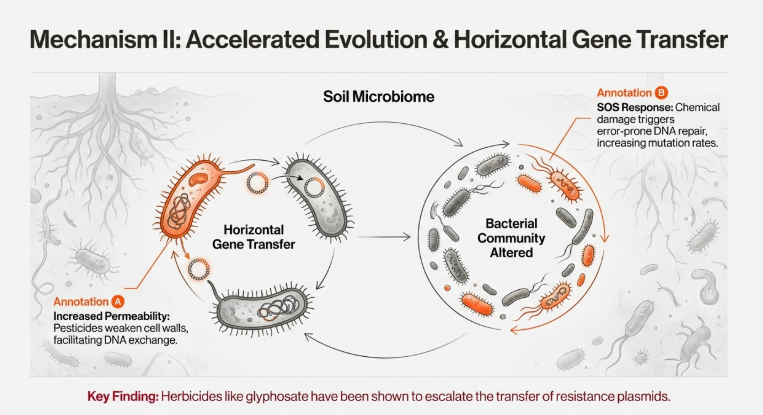
• Enhancement of Horizontal Gene Transfer (HGT): Pesticides increase bacterial cell membrane permeability. This facilitates the exchange of mobile genetic elements, such as plasmids, allowing antibiotic resistance genes (ARGs) to move rapidly across diverse bacterial taxa.
A primary policy concern is the “Cross-Resistance” phenomenon mediated by α–β hydrolase enzymes. Recent discoveries in Bacillus species show plasmid-encoded enzymes capable of degrading both Organophosphate (OP) pesticides and clinical antibiotics. This dual functionality effectively transforms a farmer’s field into a “bioreactor” for MDR strains, where selection for pesticide tolerance inadvertently selects for resistance to human medicine. These pesticide-driven risks are further compounded by the presence of persistent elemental contaminants that provide a cumulative selective landscape.
4. Elemental Co-Selection: Heavy Metals as Persistent Selective Pressures
Heavy metals (HMs) represent a persistent strategic challenge because, unlike antibiotics, they are non-degradable. They exert continuous selective pressure in aquatic and terrestrial ecosystems for decades. Resistance emerges through “co-selection,” where genes for metal resistance and ARGs are co-located on the same mobile genetic elements, meaning the presence of one pollutant maintains the resistance to both.
Metagenomic analysis identifies specific elements as statistically significant predictors of β-lactamase resistance genes. According to logistic regression models, the following elements act as primary drivers:
| Element | Correlated Resistance Genes | Impact on Resistance (Weighting) |
|---|---|---|
| Phosphorus (P) | β-lactamase genes | Strong predictor of prevalence (β0=3.935); promotes resistant bacterial growth. |
| Iron (Fe) | β-lactamase genes | Strongest predictor (β0=90.85); facilitates gene transfer and selective pressure. |
| Boron (B) | β-lactamase genes | Significant positive correlation with resistance gene frequency. |
| Lithium (Li) | β-lactamase genes | Enhances selective pressure in polluted aquatic ecosystems. |
| Magnesium (Mg) | β-lactamase genes | Linked to the proliferation of resistant microbial taxa. |
| Sodium (Na) | β-lactamase genes | Positively correlated with the presence of multidrug-resistant markers. |
This “co-resistance” logic indicates that heavy metal contamination acts as a long-term stabilizer for antibiotic resistance. Even if clinical antibiotic use is curtailed, elemental pollutants ensure that the microbial “memory” of resistance remains fixed in the environment. This chemical pressure converges with the massive biological waste streams generated by industrial livestock operations.
5. Industrial Livestock and the Environmental Waste Loop
The scale of antimicrobial flux in food production represents a systemic failure in agricultural partitioning, with global animal consumption often dwarfing human clinical use. In the United States, 70% of medically important antibiotics are sold for use in animals. This creates a “Threefold Risk” loop that directly impacts human mortality:
1. Direct Contact: Occupational transmission between livestock and agricultural workers.
2. Food Chain Transfer: Contamination of meat, dairy, and eggs. A 2023 study found that 8% of E. coli urinary tract infections in a U.S. population were traced directly to livestock sources, providing a “smoking gun” for animal-to-human transmission.
3. Environmental Excretion: Animals excrete 75–90% of consumed antibiotics unmetabolized.
The 2015 discovery of the mcr-1 gene in China marked a watershed moment; the gene, conferring resistance to the last-resort drug colistin, was found in 20% of animals and 1% of hospital patients in the same area. This strongly indicates that agricultural selection is the primary driver for the loss of last-defense clinical tools. While sub-therapeutic use for growth promotion offers only a marginal 5% productivity benefit in high-income countries, the biological trade-off is the creation of environmental “sinks” where resistance genes proliferate.
6. Environmental Reservoirs: Hotspots of Resistance and Global Targets
The strategic importance of monitoring aquatic ecosystems and manufacturing sites cannot be overstated. These sites serve as the ultimate “sinks” where chemical and biological drivers converge.
• Manufacturing Waste: API (Active Pharmaceutical Ingredient) concentrations near manufacturing plants have been found at levels 1,000 times higher than therapeutic thresholds. These “hotspots” provide concentrations exceeding what would be found in a patient’s bloodstream, accelerating the emergence of resistance.
• Aquatic Ecosystems: Metagenomic findings show a dominance of Proteobacteria (Gamma, Beta, and Alpha) in contaminated wetlands. These taxa are metabolically versatile; they do not just survive pollutants, they thrive on them, using pesticides and metals as catalysts for metabolic dominance.
Addressing these gaps requires strategic surveillance. Initiatives like the £265 million Fleming Fund are essential because you cannot regulate what you cannot measure. Furthermore, the O’Neill Review provides clear targets:
1. Global Reduction Targets: Adopting the “Denmark model” to limit use to 50mg/kg of livestock. Notably, Denmark’s agricultural productivity increased by 65% after reducing consumption, proving that the economic trade-off is a false dichotomy.
2. Restrictions on Critical Drugs: A global ban on antibiotics critical for human health (e.g., colistin, carbapenems).
3. Minimum Manufacturing Standards: Enforcing environmental criteria in Good Manufacturing Practice (GMP) guidelines to treat API discharge as industrial pollution.
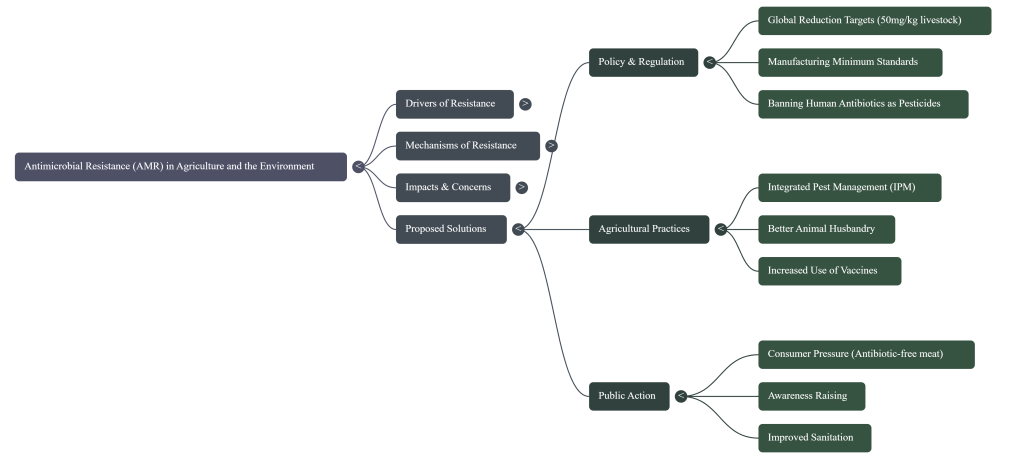
7. Solutions and the Path Forward

Mitigating the threat of AMR requires bold policy shifts focused on reducing unnecessary pharmaceutical use and improving environmental stewardship. The following action items are essential for a sustainable future:
Action Items for a Healthy Planet
• Establish Global Targets: Countries should adopt limits on antibiotic use in livestock, modeled after the Danish system of 50mg/kg. This must be supported by a robust surveillance system like DANMAP, because you cannot manage what you do not measure.
Minimum Standards for API Discharge
To address manufacturing-source resistance, the following stakeholder-specific responsibilities are mandated:
• Regulators: Must update Good Manufacturing Practice (GMP) guidelines to include environmental discharge limits for antibiotics. Statutory risk assessments must consider resistance potential, not just direct toxicity.
• Pharmaceutical Buyers (Public & Private): Must integrate environmental waste management into procurement criteria. Access to lucrative markets should be contingent on “Resistance-Safe” certification from suppliers.
• Manufacturers: Obligated to implement advanced waste treatment technologies to ensure antibiotic concentrations in effluent do not exceed scientifically established no-effect levels (typically 10% of the maximum limit).
Surveillance and Data Transparency
Radically improved surveillance is the “So What?” of this entire framework. Without transparent, granular data on usage patterns and discharge levels, targets cannot be legally mandated or enforced. Improved data collection provides the evidence required to hold industries and sovereign nations accountable for the negative externalities they generate.
• Restrict Human “Last-Resort” Drugs: Implement an immediate ban on the agricultural use of antibiotics critical to human medicine, such as Colistin.
• Mandate Manufacturing Standards: Establish minimum international standards for pharmaceutical waste management to prevent factory effluent from serving as an evolutionary engine for new superbugs.

Ultimately, systemic change is driven by public demand. By advocating for “responsible use” labeling and transparency, consumers can leverage their purchasing power to force a shift in the global food system, ensuring that our most vital medicines remain effective for future generations.
The Danish Success Story: A Blueprint for Change
The situation is urgent, but Denmark provides a practical blueprint for reform. Since the 1990s, Denmark has proven that you can de-link pharmaceutical profits from agricultural productivity.
Using a rigorous surveillance system known as DANMAP, Denmark banned antibiotic growth promoters and transitioned to a high-transparency model. The result was a 50% reduction in antimicrobial use in livestock. Remarkably, Danish agricultural productivity grew by 65% during this period, far outstripping the EU average of 25%.
The “Denmark Method” focuses on:
• DANMAP Surveillance: Rigorous data collection and mapping of resistance trends.
• Biosecurity: Better farm hygiene and selection of sites with good water exchange.
• Vaccines: Using immunizations to prevent disease before it requires therapy.
• Regulatory Oversight: Strict mandatory reporting of all prescriptions.
Fiscal Levers: Internalizing Negative Externalities through Market Correction Mechanisms
In economic theory, antimicrobial use in agriculture constitutes a classic “negative externality.” Individual producer incentives (e.g., rapid weight gain) impose massive, uncompensated costs on global society. To correct this market failure, this framework proposes a Market Correction Mechanism based on Pigouvian taxation.
Taxation Strategy for Negative Externalities
Taxation should be levied at the factory gate or point of manufacture. This minimizes the number of taxable entities and reduces the risk of black-market circumvention.
Strategic Objectives of the Taxation Model:
1. Demand Destruction for Non-Essential Use: Increasing the cost relative to the product price discourages the commercially unjustifiable practice of growth promotion.
2. Tiered Risk-Based Taxation: Higher tax brackets must be applied to drugs “critically important” for human health. Specifically, last-resort agents such as colistin must be subject to punitive tax rates to reflect their clinical value, particularly following the discovery of the plasmid-mediated mcr-1 gene.
3. Inducing Behavioral Shifts: Elevated costs incentivize the adoption of rapid diagnostics and higher husbandry standards as a lower-cost alternative to chemical reliance.
While taxation reduces the demand for harmful practices, it must be strategically paired with subsidies to lower the barrier to entry for regenerative and sustainable production systems.
Fiscal Incentives: Subsidizing the Transition to Regenerative Systems
The transition away from antibiotic reliance involves significant “switching costs.” Subsidies must be framed as strategic investments in positive externalities, effectively paying for the public health and environmental security that intensive systems currently exploit.
Strategic Subsidy Allocation
The framework identifies three priority areas for fiscal support:
1. Vaccine Development and Commercialization: Proper vaccination can halve antibiotic consumption. Subsidies should prioritize non-injectable delivery mechanisms (e.g., feed-based vaccines) for smaller livestock and aquaculture.
2. Rapid Point-of-Care Diagnostics: Transitioning from empirical to targeted treatment is essential. Subsidies must lower the cost of diagnostic hardware, ensuring that treatment is only applied following confirmed bacterial infection.
3. Infrastructure and Biosecurity Upgrades: Financial support is required for farms to transition to “Scandinavian-style” hygiene standards, reducing the physiological stress on animals that necessitates prophylactic antibiotic use.
The Productivity Myth
Opponents of stewardship often cite food security risks. However, the Danish precedent establishes a scalable baseline: between 1992 and 2012, Denmark achieved a 65% increase in agricultural productivity despite an aggressive ban on growth promoters and a 51% reduction in per-unit antibiotic use. This confirms that low-antibiotic systems are not only compatible with food security but are more resilient and commercially competitive in the long term.
8. Conclusion: Navigating the Post-Antibiotic Era
Antimicrobial resistance is a definitive “Planetary Health” issue that transcends clinical stewardship. To protect the efficacy of modern medicine, we must implement a fundamental shift in agricultural chemical management and industrial waste oversight. The successful phase-out of Chlorofluorocarbons (CFCs) under the Montreal Protocol serves as a historic precedent for global “Planetary Health” interventions; just as the world united to save the ozone layer, a harmonized global effort can stabilize the microbial landscape.

Success depends on transitioning to “responsible use” labeling, adopting vaccines and rapid diagnostics as pesticide/antibiotic alternatives, and treating environmental contamination as a primary driver of pathogen evolution. By addressing these unseen catalysts, we can secure the future of global food systems and human health.
Image Summary

Questions/Answers
1. How do non-antibiotic farm chemicals like pesticides promote drug resistance?
Non-antibiotic farm chemicals, particularly pesticides (including herbicides, fungicides, and insecticides), promote drug resistance through several indirect mechanisms that exert selective pressure on microbial communities. These chemicals act as environmental stressors that trigger adaptive responses in bacteria, often conferring resistance to medically important antibiotics even in the absence of the antibiotics themselves.
1. Co-selection Mechanisms
The primary way non-antibiotic chemicals drive resistance is through co-selection, which occurs via three main processes:
• Cross-resistance: This happens when a single molecular mechanism provides resistance to both a pesticide and an antibiotic. For example, efflux pumps—proteins that bacteria use to expel toxic substances—can be activated by pesticides to pump out antibiotics like ciprofloxacin and tetracycline.
• Co-resistance: Multiple resistance genes, such as those for pesticide degradation and antibiotic resistance, are physically linked on the same piece of DNA, such as a plasmid. When a pesticide selects for the degradation gene, the antibiotic resistance gene is “co-selected” and maintained in the population.
• Co-regulation: Exposure to one chemical agent can trigger a coordinated transcriptional or translational response that includes the expression of resistance genes for other agents.
2. Upregulation of Efflux Pumps
Many pesticides induce the overexpression of multidrug efflux pumps. Groundbreaking studies have shown that sub-lethal exposure to common herbicides like glyphosate, dicamba, and 2,4-D induces the AcrAB-TolC efflux pump system in E. coli and Salmonella. This system effectively expels antibiotics, including carbapenems and fluoroquinolones, out of the bacterial cell.
3. Increased Horizontal Gene Transfer (HGT)
Pesticides facilitate the rapid spread of resistance by accelerating the rate at which bacteria exchange DNA through conjugation, transformation, or transduction. These chemicals can:
• Increase cell membrane permeability, making it easier for foreign DNA (like plasmids carrying resistance genes) to enter the cell.
• Induce stress responses (such as the SOS response) and the production of reactive oxygen species (ROS), which stimulate the mobilization of genetic elements.
4. Genetic Mutations and Structural Changes
Exposure to pesticides can stimulate de novo mutations and phenotypic changes that hinder antibiotic effectiveness.
• Target Modification: Long-term exposure to pesticide mixtures has been shown to induce mutations in genes like rpsG (related to streptomycin resistance) or gyrA (related to ciprofloxacin resistance).
• Reduced Permeability: Pesticides can cause bacteria to decrease the synthesis of outer membrane porins, effectively closing the “doors” through which antibiotics enter the cell.
• Biofilm Formation: Pesticides can induce a holistic stress response that promotes biofilm formation, a protective layer that allows bacteria to better withstand antibiotic challenges.
5. Impact of Specific Chemicals
• Herbicides (e.g., Glyphosate, Dicamba): These are the most widely used agrochemicals and have been shown to increase resistance to antibiotics like ampicillin, ciprofloxacin, and kanamycin.
• Insecticides (e.g., Imidacloprid, Chlorpyrifos): Experimental studies have linked imidacloprid to significant increases in resistance to carbapenems, aminoglycosides, and cephalosporins. Chlorpyrifos has also been shown to increase the abundance of antibiotic resistance genes (ARGs) in soil.
• Fungicides (e.g., Azoles): The massive use of azole fungicides in agriculture has directly contributed to the emergence of triazole-resistant Aspergillus fumigatus, a pathogen responsible for human aspergillosis.
Finally, these chemicals can deplete the microbial biodiversity of healthy soils. When general microbial diversity declines, competition for resources is reduced, creating empty ecological niches that allow drug-resistant “superbugs” to proliferate and spread through the food chain.
2. How does climate change accelerate the global spread of antimicrobial resistance?
Climate change acts as a powerful force multiplier that accelerates the emergence and spread of antimicrobial resistance (AMR) through several distinct environmental and biological mechanisms.
1. Temperature-Driven Proliferation and Mutation
Rising global temperatures directly influence the behavior and prevalence of bacterial pathogens. Higher ambient temperatures increase the rate of bacterial proliferation, which naturally leads to an increased risk of infection. Research indicates that a 1°C increase in average temperature is associated with a 5% increased risk of salmonellosis, a 7% increase for shigellosis, and a 15% increase for typhoid.
Beyond simple growth, warmer temperatures serve as a biological stressor that can increase bacterial mutation rates and promote horizontal gene transfer (HGT), the primary method by which resistance genes move between different species of bacteria. Studies in the U.S. and China have already established a correlation between higher local temperatures and increased resistance in clinical pathogens, including significant rises in carbapenem-resistant K. pneumoniae and P. aeruginosa.
2. Extreme Weather and Environmental Dispersal
Climate change increases the frequency and intensity of extreme weather events, such as hurricanes, heavy precipitation, and subsequent flooding, which serve as major vehicles for the waterborne transmission of resistant bacteria and their genes.
• Flooding and Runoff: Floodwaters can carry resistant bacteria from diverse sources—including compromised sewer systems and agricultural fields treated with animal manure—into coastal sediments and urban soils.
• Persistent Contamination: Following events like Hurricane Harvey, elevated loads of human fecal bacteria and antibiotic resistance genes (ARGs) remained in coastal sediments for several months.
• Marine Pathogens: Warmer, wetter conditions and changes in salinity levels facilitate the proliferation of marine pathogens like Vibrio spp., which have been found to carry numerous ARGs and mobile genetic elements following major storm surges.
3. Impacts on Food Systems and Agricultural Feedbacks
The environmental stresses caused by climate change—specifically heat and drought—threaten the stability and productivity of global food systems. As yields from traditional monoculture crops decline, there is a risk of a “feedback loop” where agricultural practices are further intensified.
• Chemical Stressors: To maintain productivity in a changing climate, there may be an increased reliance on chemical fertilizers, herbicides, and pesticides.
• Induced Resistance: These chemical inputs act as abiotic stressors that deplete soil microbial biodiversity and select for resistance traits in the remaining bacterial populations, creating niches where “superbugs” can flourish without competition.
In summary, the combination of warmer ambient conditions and increased environmental volatility provides an ideal breeding ground for drug-resistant bacteria while simultaneously compromising the infrastructure and natural ecosystems that might otherwise limit their spread.
3. Explain the role of pharmaceutical manufacturing waste in creating resistance hotspots.
Pharmaceutical manufacturing waste creates resistance hotspots by introducing exceptionally high concentrations of active pharmaceutical ingredients (APIs) into localized environments, providing a massive selection pressure for the evolution and spread of antibiotic resistance.
The specific role of this waste in creating these hotspots is detailed below:
1. Extreme Selection Pressure
Unlike the antibiotics excreted by humans and animals, which are limited by metabolism and dosage, emissions from drug manufacturing can reach levels that are thousands or even millions of times higher than those found in municipal sewage. In some industrially polluted sites, the concentration of antibiotics in the water or sediment can actually exceed therapeutic levels found in the blood of a patient taking the drug. This creates an environment where only bacteria with high-level resistance can survive, effectively eliminating sensitive strains and promoting the dominance of resistant genotypes.
2. Spawning Ground for New Resistance Genes
Manufacturing sites act as “spawning grounds” for resistance evolution. These hotspots foster the emergence of previously unknown antibiotic resistance genes (ARGs). Because these environments often harbour dense, complex bacterial communities in close contact with high antibiotic levels, they facilitate the stepwise evolution of resistance:
• Mobilization: Moving immobile chromosomal genes to mobile elements like plasmids.
• Horizontal Gene Transfer (HGT): The rapid exchange of these genes between different bacterial species.
• Selection: The high chemical stress ensures that rare genotypes with newly acquired resistance are selected for and fixed in the population rather than disappearing.
3. Geographic and Economic Drivers
A significant portion of global antibiotic production occurs in low- and middle-income countries (LMICs), particularly China and India, due to lower production costs. In these regions, insufficient waste management and erratic discharge patterns have been reported, leading to excessive emissions into local waterways. These communities bear the health burden of polluted water, which serves as a reservoir for resistant bacteria that can ultimately spread globally through travel and trade.
4. Global Ramifications
Experts emphasize that it only takes one evolutionary event in one setting for a new, successful resistance genotype to emerge. Once a “superbug” is created in a manufacturing hotspot, its global spread is often irreversible. For this reason, many stakeholders consider the risks of high-level industrial antibiotic pollution to be unacceptable, urging the implementation of minimum standards for waste management to eliminate these hotspots.
4. How does glyphosate exposure affect the activity of bacterial efflux pumps?
Exposure to glyphosate affects bacterial efflux pumps primarily by inducing their upregulation and increasing their activity as an adaptive response to chemical stress.
The specific ways glyphosate impacts these systems include:
• Induction of the AcrAB-TolC System: Sub-lethal doses of glyphosate have been shown to trigger the AcrAB-TolC efflux pump system in bacteria such as Escherichia coli and Salmonella enterica. This system is a major multi-drug transporter that expels toxic compounds from the cell.
• Mechanism of Adaptive Resistance: This upregulation is an adaptive phenotypic response to the presence of environmental signals (the herbicide), allowing the bacteria to survive toxic stressors.
• Promotion of Cross-Resistance: Because these pumps are not specific to glyphosate, their activation leads to cross-resistance toward medically important antibiotics. By hyper-expressing these pumps, bacteria can effectively export various drugs, including ciprofloxacin, ampicillin, kanamycin, tetracycline, and chloramphenicol, thereby reducing their intracellular concentration and effectiveness.
• Evidence through Inhibition: The role of efflux pumps in this process is confirmed by studies showing that when these systems are chemically or genetically inhibited, the tolerance to antibiotics induced by glyphosate significantly decreases.
Additionally, glyphosate-induced stress can lead to de novo mutations in genes that regulate these pumps or target-alteration mutations that further enhance a bacterium’s resistant phenotype. These effects can be cumulative; when glyphosate is combined with other herbicides or stressors like salicylic acid, the magnitude of the efflux response can increase further.
5. Can triazole fungicides in agriculture lead to drug-resistant human fungal infections?
Yes, triazole fungicides used in agriculture can lead to drug-resistant human fungal infections, specifically those caused by Aspergillus fumigatus, the primary pathogen responsible for aspergillosis.
The Link Between Agriculture and Human Infection
Triazole fungicides used in plant production are structurally similar (chemically homologous) to the medical triazoles used to treat human fungal infections. Because both agricultural and medical azoles share the same mechanism of action—inhibiting the fungal enzyme lanosterol 14 alpha-demethylase (Cyp51A) to disrupt cell membrane synthesis—wide-scale agricultural use can select for resistance traits that provide cross-resistance to life-saving medical drugs.
Key evidence supporting this link includes:
• Infections in “Azole-Naïve” Patients: Approximately 50% of patients with invasive aspergillosis caused by triazole-resistant strains have never been treated with medical azoles before, strongly indicating they acquired the resistant fungus directly from an environment altered by agricultural chemicals.
• Shared Genetic Mutations: Specific resistance mechanisms, such as the TR34/L98H and TR46/Y121F/T289A mutations, have been identified in both environmental soil samples from treated agricultural fields and clinical samples from patients across Europe, Asia, and Africa.
• Environmental Persistence: Azoles are highly stable and can persist in soil and water for many months (with half-lives exceeding 300 days in some cases), providing a long-term selective pressure for resistant fungal strains.
Impact on Human Health
Aspergillosis is a serious condition with high morbidity and mortality, especially in immunocompromised individuals. When these infections are caused by environmental triazole-resistant strains, standard medical treatments such as itraconazole, voriconazole, and posaconazole can fail. In cases of acute invasive aspergillosis involving these environmentally derived resistant strains, mortality rates as high as 88% have been reported.
Geographic Hotspots
The problem is most prevalent in regions with heavy agricultural fungicide application.
• The Netherlands: Standard practices, such as dipping every tulip bulb in fungicide, have been linked to some of the highest rates of triazole resistance in A. fumigatus (approaching 7%).
• Europe: Nearly 50% of the acreage for cereal and grapevine production in Europe is treated with azoles annually, compared to less than 5% in the U.S., leading to higher concern in European clinical settings.
6. Do pesticides increase the rate of horizontal gene transfer between bacteria?
Yes, several types of pesticides—including herbicides, fungicides, and insecticides—have been shown to increase the rate of horizontal gene transfer (HGT) between bacteria. This process is a primary mechanism by which antibiotic resistance genes (ARGs) spread through microbial communities in the environment.
Pesticides facilitate this increase through several biological and physical mechanisms:
1. Increased Membrane Permeability
Exposure to pesticides can stress bacterial cells and lead to increased cell membrane permeability. This physical change makes it easier for foreign DNA, such as plasmids carrying resistance genes, to enter the bacterial cell. For example, the herbicide glyphosate has been shown to escalate the horizontal transfer of conjugative plasmids by upregulating genes related to membrane permeability.
2. Induction of Stress Responses
Pesticides act as environmental stressors that trigger adaptive responses in bacteria.
• SOS Response: Herbicide exposure can cause oxidative damage to DNA and the accumulation of reactive oxygen species (ROS), which induces the SOS response. This stress response is a known driver for the mobilization and transfer of genetic elements.
• Conjugation Genes: Specific fungicides like azoxystrobin, mancozeb, and carbendazim have been observed to enhance the expression of genes directly involved in conjugation (the “mating” process between bacteria), thereby facilitating the transfer of multi-drug resistance plasmids.
3. Co-selection and Mobile Genetic Elements
Pesticides can increase the prevalence of mobile genetic elements (MGEs), such as integrons and transposons, which are responsible for capturing and moving ARGs.
• Herbicides: Research indicates that typical application levels of herbicides like dicamba, glyphosate, and glufosinate can elevate the relative abundance of MGEs and ARGs in soil bacterial communities regardless of changes in the overall community composition.
• Organophosphates: Contamination of soil and water with organophosphate pesticides has also been linked to the horizontal transfer of ARGs via plasmids and transposons.
4. Role of Metal-Based Pesticides
Metals like copper, which are frequently used as agricultural fungicides and bactericides, also promote HGT. Copper ions can generate intracellular ROS, which increases membrane permeability and boosts the transfer efficiency of resistance-associated genes between different genera of bacteria.
In summary, pesticides accelerate the spread of drug resistance not just by killing sensitive bacteria, but by actively promoting the genetic machinery that allows bacteria to share resistance traits with one another.
7. Does climate-driven agricultural intensification create new superbug niches?
Yes, climate-driven agricultural intensification creates new niches for “superbugs” (multidrug-resistant bacteria) through a cycle of environmental stress, chemical intervention, and the resulting loss of microbial biodiversity.
The mechanisms through which this occurs include:
1. The Intensification Feedback Loop
Climate change increases the frequency of extreme weather events, such as severe droughts and rising temperatures, which threaten the stability and productivity of traditional monoculture food systems. As yields decline, there is an increased risk of an agricultural “feedback loop” where practices are further intensified to maintain output. This intensification often involves a higher reliance on chemical fertilizers, herbicides, and pesticides.
2. Creation of “Empty” Ecological Niches
The heavy use of these chemicals acts as a potent abiotic stressor that significantly depletes soil microbial biodiversity. When general microbial diversity is lost, the natural competition for resources among species is drastically reduced. This depletion of the microbial ecosystem creates empty ecological niches where drug-resistant bacteria can flourish. Without diverse, healthy microbial communities to act as a barrier, these “superbugs” are able to proliferate and spread through the food chain more effectively.
3. Accelerated Proliferation and Spread
Climate-driven environmental changes further support these new niches:
• Temperature-Driven Mutation: Higher ambient temperatures increase the rate of bacterial proliferation and can increase mutation rates, facilitating the emergence of new resistance traits.
• Dispersal via Flooding: Heavy precipitation and subsequent flooding act as major vehicles for the waterborne transmission of resistant bacteria and their genes, carrying them from agricultural fields and waste streams into new environments like urban soils and coastal sediments.
In summary, the combination of intensive chemical inputs (to combat climate-driven crop failure) and warmer, more volatile conditions provides an ideal environment for drug-resistant pathogens to occupy niches previously filled by a more diverse array of sensitive microbial species.
8. Can heat stress increase the rate of bacterial mutations?
Yes, heat stress can increase the rate of bacterial mutations. Rising temperatures act as a powerful biological stressor that can trigger several genetic and physiological changes in bacteria:
• Increased Mutation Rates: Heat stress is a proposed mechanism for the emergence of antimicrobial resistance (AMR) because it can increase the rate of de novo mutations within bacterial populations.
• Linked Resistance Mechanisms: The processes by which bacteria develop temperature resistance and antibiotic resistance are often intimately linked. Exposure to thermal stress can therefore select for traits that also confer resistance to medically important drugs.
• Adaptive Transcriptional Responses: Research on E. coli has shown that resistant mutants often exhibit higher expression of heat shock defense genes (such as ibpAB and hslVU) when navigating environmental challenges. These general stress responses can provide a survival advantage against multiple concurrent threats, including antibiotics.
• Stimulated Horizontal Gene Transfer (HGT): Beyond individual mutations, warmer temperatures can promote the rate of horizontal gene transfer, allowing resistance genes to spread more rapidly across different bacterial strains and species.
Furthermore, higher temperatures increase the overall rate of bacterial proliferation, which not only leads to a higher risk of infection but also creates more opportunities for spontaneous mutations to occur and become fixed within a population.
9. How do extreme weather events like floods spread resistance genes?
Extreme weather events, particularly heavy precipitation and subsequent flooding, act as powerful vehicles for the global spread and waterborne transmission of antimicrobial resistance (AMR) by dispersing resistant bacteria and their genes across vast distances and into new environments.
The mechanisms through which floods spread resistance genes include:
1. Mingling of Diverse Resistance Sources
Flooding provides unique opportunities for the mingling and dispersal of bacteria and antibiotic resistance genes (ARGs) from normally separated sources. Flood waters can carry resistant bacteria from:
• Agricultural Fields: Manure used as fertilizer, which often contains high loads of ARGs and antibiotic residues from livestock, can be washed into waterways.
• Sewer Systems: Inundated municipal and hospital sewer systems can overflow, releasing human fecal bacteria and clinical resistance factors into the environment.
• Waste Streams: Runoff from industrial and agricultural waste streams provides a constant supply of genetic material for environmental dispersal.
2. Persistent Contamination of Sediments and Soils
Research indicates that the impact of flood-driven ARG spread is not just immediate but can lead to long-term environmental alterations:
• Coastal Sediments: Following Hurricane Harvey, researchers found significantly elevated loads of human fecal bacteria (E. coli, Enterococcus spp., and Salmonella spp.) and ARGs conferring resistance to beta-lactams in coastal sediments, which remained present for several months.
• Urban Soils: Flooding can cause transient increases in ARGs in urban areas, such as tet(E) (tetracycline resistance) and blaCMY-2 (beta-lactam resistance), as well as the mobile genetic element intl1 (class 1 integron-integrase), which facilitates the acquisition of new resistance traits.
3. Proliferation of Waterborne Pathogens
Warmer and wetter ambient conditions following extreme weather events create ideal breeding grounds for drug-resistant pathogens. For instance, storm surges and changes in salinity levels following events like Hurricane Ian have been linked to spikes in marine pathogens like Vibrio parahaemolyticus and Vibrio vulnificus, which were found to carry numerous ARGs and mobile genetic elements.
4. Compromised Infrastructure
In many regions, particularly low- and middle-income countries, inferior infrastructure for managing human and animal waste streams makes these areas particularly vulnerable to weather-driven AMR spread. When these systems are overwhelmed by floods, the emission of resistant fecal bacteria and residual antibiotics into the surrounding environment increases significantly.
10. How do concentrations in waste compare to human therapeutic levels?
In certain environments, particularly those contaminated by pharmaceutical manufacturing waste, antibiotic concentrations can reach levels that are far higher than the therapeutic levels found in the blood of a human patient taking the drug.
The comparison between these waste concentrations and therapeutic levels can be broken down into several key findings from the sources:
1. Exceeding Human Blood Levels
In industrially polluted areas where active pharmaceutical ingredients (APIs) are discharged, the concentration of antibiotics in the wastewater or surrounding effluent can exceed the therapeutic concentrations expected in a patient’s bloodstream. Unlike antibiotics reached through human or animal excretion—which are limited by the doses used and the body’s metabolism—manufacturing waste carries untreated high levels of the direct active ingredient.
2. Orders of Magnitude Above Normal Sewage
Concentrations in manufacturing waste hotspots can be many thousands or even millions of times higher than the concentrations found in standard municipal sewage. While most environmental concentrations are low (often in the nanogram per liter range), industrially polluted sites can exceed selective concentrations by orders of magnitude.
3. Comparison to Minimal Inhibitory Concentrations (MICs)
• Excreted Waste: Typical antibiotic concentrations from human and animal use in environments like rivers or treated sewage are generally low—often much lower than the Minimal Inhibitory Concentrations (MICs) required to kill bacteria.
• Manufacturing Waste: In contrast, surface waters polluted by drug manufacturing waste frequently show antibiotic concentrations that fall at the high end of, or exceed, typical MIC ranges (which often range from 10 to 10,000 µg/l).
4. Impact of Extreme Concentrations
The presence of such high concentrations creates extreme selection pressure. While lower concentrations in municipal waste may select for resistance more subtly, these industrial hotspots eliminate all but the most resistant bacteria. This makes these areas “spawning grounds” for the evolution of new resistance genes, as rare genotypes are rapidly selected for and fixed in the population by the intense chemical stress.
11. What structural similarities cause cross-resistance between agricultural and medical azoles?
The cross-resistance between agricultural and medical azoles is primarily caused by their chemical homology and their identical biological target within fungal cells.
The following factors explain how these structural similarities drive resistance:
1. Shared Molecular Mechanism
All azoles, whether used to protect crops or treat human infections, work through the same mechanism: they inhibit the fungal enzyme lanosterol 14 alpha-demethylase (encoded by the Cyp51A gene). This enzyme is essential for synthesizing ergosterol, a critical component of fungal cell membranes. Because agricultural and medical azoles target the exact same site, a mutation that protects a fungus from one often provides protection against the other.
2. Structural Homology
While the specific active ingredients may differ, many agricultural fungicides are chemically homologous to the triazoles used in human and veterinary medicine. Key examples of agricultural azoles with structural similarities to medical drugs include:
• Propiconazole
• Bromuconazole
• Tebuconazole
• Epoxiconazole
• Difenoconazole
3. Common Resistance Mutations
Because of these structural similarities, exposure to azoles in the environment (such as in tulip production or cereal farming) selects for specific genetic mutations in the Cyp51A gene. Two major mechanisms have been identified in both environmental soil samples and clinical samples from human patients:
• TR34/L98H: A 34-bp tandem repeat in the promoter region combined with a leucine-to-histidine substitution.
• TR46/Y121F/T289A: A similar repeat and mutation pattern that confers high-level resistance.
4. Clinical Consequences
This shared structure means that patients can acquire triazole-resistant Aspergillus fumigatus (the cause of aspergillosis) directly from the environment without ever having been treated with medical azoles. Approximately 50% of patients with invasive triazole-resistant aspergillosis are “azole-naïve,” meaning their infection was likely pre-selected for resistance by agricultural chemicals before it ever entered their body. In these cases, standard medical treatments like itraconazole, voriconazole, and posaconazole often fail, leading to reported mortality rates as high as 88%.
12. Could reducing pesticide use lower the risk of drug-resistant aspergillosis?
Yes, reducing the use of specific pesticides—specifically triazole fungicides—could significantly lower the risk of drug-resistant aspergillosis in humans.
The connection between agricultural practices and this human health threat is established through several factors detailed in the sources:
1. Mechanism of Cross-Resistance
Triazole fungicides used in crop production are chemically homologous (structurally similar) to the medical triazoles used to treat human infections caused by Aspergillus fumigatus. Because both agricultural and medical azoles share the same mechanism of action—inhibiting the fungal enzyme 14α-demethylase—the widespread use of these chemicals in the environment selects for resistance traits that provide cross-resistance to life-saving human drugs.
2. Environmental Acquisition of Resistance
Evidence suggests that many humans acquire drug-resistant aspergillosis directly from the environment rather than through clinical exposure.
• “Azole-Naïve” Patients: Approximately 50% of patients with invasive aspergillosis caused by triazole-resistant strains have never been treated with medical azoles before, indicating they were infected by resistant fungi already present in the environment due to agricultural selection.
• High Mortality: For these patients, mortality rates can reach 88% because standard medical treatments like itraconazole and voriconazole often fail.
• Stability in Soil: Azoles are highly stable and can remain active in soil and water for hundreds of days, providing a persistent selective pressure that favors the survival of resistant strains.
3. Geographic Evidence
The correlation between fungicide use and human resistance is strongest in regions with intensive application. For example, in the Netherlands, where practices like dipping every tulip bulb in fungicide are standard, resistance rates in A. fumigatus are approaching 7%. In contrast, the U.S. treats less than 5% of its cereal and grapevine acreage with azoles, compared to nearly 50% in Europe, which correlates with higher clinical concern in European settings.
4. Impact of Reduced Use
The sources suggest that the “irrational” or excessive use of these fungicides is a primary driver of the current crisis. Proposed actions to lower the risk of resistant aspergillosis include:
• Targeted Bans: Banning certain azoles from non-food crop production (such as tulip farming) and prohibiting the agricultural use of any new classes of clinical antifungals developed in the future.
• Integrated Pest Management (IPM): Adopting IPM strategies that use fungicides only as a last resort when other methods fail, thereby reducing the environmental selective pressure on fungal populations.
• Prudent Stewardship: Implementing stricter regulations on the dosage and timing of applications to prevent the accumulation of residues that foster resistance.
By lowering the environmental load of these chemicals, the selective pressure on Aspergillus species is reduced, which can help preserve the efficacy of medical triazoles as vital tools for treating human infections.
13. How do copper and zinc fungicides contribute to bacterial resistance?
Copper and zinc are among the most widely used inorganic antimicrobials in agriculture, utilized as both fungicides and bactericides to protect crops and as growth-promoting additives in animal feed. They contribute to bacterial resistance primarily through co-selection mechanisms and by accelerating the spread of resistance genes in the environment.
1. Co-selection Mechanisms
The presence of copper and zinc exerts selective pressure on bacteria, leading to the development of antibiotic resistance through three key processes:
• Co-resistance: This occurs when genes conferring resistance to metals and those for antibiotics are physically linked on the same piece of DNA, such as a plasmid or integron. When a metal like copper selects for a metal-resistant strain, the linked antibiotic resistance genes (ARGs) are “co-selected” and maintained. A documented example is in Enterococcus faecium, where copper resistance (tcrB) is genetically linked to macrolide (erm(B)) and glycopeptide (vanA) resistance on the same plasmid.
• Cross-resistance: A single molecular mechanism can provide resistance to both the metal and multiple antibiotics. For instance, multidrug efflux pumps can be activated to expel both metal ions and medically important drugs like ciprofloxacin and chloramphenicol from the bacterial cell.
• Co-regulation: Exposure to metal ions can trigger coordinated transcriptional responses that activate the expression of multiple separate resistance genes, including those for antibiotics.
2. Stimulation of Horizontal Gene Transfer (HGT)
Metals contribute to the rapid dissemination of resistance traits by increasing the efficiency of DNA exchange between bacteria:
• Increased Membrane Permeability: Copper ions, in particular, can generate intracellular reactive oxygen species (ROS), which stress the cell and increase the permeability of the bacterial membrane. This physical change makes it easier for foreign DNA, such as plasmids carrying ARGs, to enter the cell.
• Enhanced Conjugation: Metal stress can upregulate genes involved in conjugation (the “mating” process between bacteria), boosting the transfer efficiency of resistance genes across different genera of bacteria.
3. Environmental Persistence
Unlike organic antibiotics, which can undergo photodegradation or be deactivated in soil, metals are highly durable and do not degrade. This environmental stability creates a large temporal selective window, meaning copper and zinc can exert constant selective pressure for many years. This long-term presence facilitates the stabilization and fixation of novel resistance genotypes within environmental microbial communities.
4. Impact through the Food Chain
The use of these metals in industrial agriculture creates a feedback loop that impacts human health:
• Accumulation in Manure: Copper and zinc added to animal feed are often excreted non-metabolized and accumulate in manure. When this manure is used as fertilizer, it introduces both metal residues and ARGs into crop soils.
• Superbug Niches: Studies in Chinese swine farms have shown that high levels of zinc and copper in manure are strongly associated with increased loads of multidrug resistance genes and the mobile genetic element intI1 in soil and aquatic sediments. This pressure can lead to the emergence of “superbugs” like MRSA in livestock, which carry resistance traits that can eventually be transmitted to humans through the food chain.
14. Can household pesticide exposure select for drug resistance in cities?
Yes, household pesticide exposure can select for drug resistance in cities. While often associated with large-scale agriculture, the use of pesticides and related biocides in urban settings is a significant and often overlooked driver of antimicrobial resistance (AMR).
1. Urban and Household Exposure Pathways
In cities, resistant microbes are selected through several household-related channels:
• Pest Control and Home Gardening: Pesticides, including insecticides, herbicides, and fungicides, are used extensively for household pest control and private gardening. Residues from these products persist in urban soil and can leach into groundwater.
• Household Cleaning Products: Many common household items, such as soaps, floor cleaners, and detergents, contain “antibacterial” biocides like triclosan or quaternary ammonium compounds (QACs), such as benzalkonium chloride.
• Municipal Waste Systems: Low-level exposure from household chemical use selects for resistant bacteria in wastewater and municipal systems. These systems often serve as “hotspots” where pesticides, biocides, and excreted antibiotics converge, creating a complex chemical environment that fosters resistance.
2. Mechanisms of Resistance in Urban Settings
Pesticides and household biocides do not have to be present at lethal doses to drive resistance. Instead, they act as environmental stressors that trigger several biological responses in bacteria:
• Co-selection and Cross-resistance: Bacteria often develop mechanisms, such as multidrug efflux pumps, to expel toxic household chemicals. These same pumps can inadvertently grant the bacteria resistance to medically important antibiotics.
• Stress-Induced Adaptations: Exposure to these chemicals at sub-lethal levels induces stress responses that promote horizontal gene transfer (HGT), increased mutation rates, and the formation of biofilms.
• Horizontal Gene Transfer: Chemicals in urban wastewater can increase the permeability of bacterial cell membranes, making it easier for them to share and acquire antibiotic resistance genes (ARGs).
3. Impact on Pathogens
This urban selective pressure can affect pathogens that are highly relevant to human health. For example:
• ESBL-producing E. coli: Household use of pesticides leads to low-level exposure that selects for resistant microbes, including Extended-Spectrum Beta-Lactamase (ESBL)-producing E. coli, which are a major cause of severe human infections like urinary tract ailments and sepsis.
• Triazole-resistant Aspergillus: The use of triazole fungicides for non-agricultural purposes, such as in home gardening or wood preservation, has raised concerns about the emergence of drug-resistant human fungal infections like aspergillosis.
Because AMR transcends borders through global trade and environmental connectivity, the selection of resistant “superbugs” in urban household environments poses a threat that can spread to both rural and clinical settings.
15. How does reduced soil biodiversity enable superbugs to thrive?
Reduced soil biodiversity enables superbugs to thrive primarily by removing the natural biological barriers that normally limit the proliferation of resistant bacteria. Healthy soil is an incredibly complex ecosystem, often containing upwards of 50,000 microbial species that compete for limited resources. When this diversity is compromised, superbugs find an environment much more conducive to their growth.
The mechanisms through which reduced biodiversity supports superbugs include:
• Lack of Competition: In healthy, diverse soils, a vast array of microorganisms engage in a “never-ending competition for resources,” which acts as a natural check against any single strain becoming dominant. Intensive agricultural practices—such as heavy tillage and the overuse of chemical fertilizers, pesticides, and herbicides—deplete this general microbial population. This reduction in diversity removes the competitors that would normally out-compete “superbugs” for nutrients and space.
• Creation of “Empty” Ecological Niches: The depletion of soil microbiota creates vacant ecological niches. Without a diverse community to fill these gaps and act as a barrier to the accumulation of resistance genes, drug-resistant bacteria are able to occupy these empty spaces and proliferate without hindrance.
• Reduced Natural Resistance Barriers: Research has shown that selection for antimicrobial resistance is significantly reduced when a bacterium is embedded in a natural, diverse microbial community. A stable, diverse environmental microbiome serves as a buffer that makes it much harder for new resistance traits to be selected for and fixed within a population.
• Feedback from Chemical Stressors: The very practices that reduce biodiversity often simultaneously act as selective pressures for resistance. For example, intensive soil management and chemical inputs act as abiotic stressors that kill sensitive natural microbes while providing an environment that favors the survival of mutants carrying resistance genes.
In summary, extractive and intensive agricultural practices create a “vicious cycle” where they simultaneously weaken the natural microbial competition and select for resistance traits, effectively transforming once-healthy soil into a “spawning ground” or niche where superbugs can flourish and eventually enter the human food chain.
16. Can regenerative farming practices help reverse herbicide-driven resistance?
Regenerative farming practices can help mitigate and potentially reverse herbicide-driven resistance by removing the chemical stressors that select for resistance and by restoring the natural microbial competition that limits the spread of “superbugs.”
1. Removal of Selective Pressure
The primary way regenerative agriculture addresses herbicide-driven resistance is by reducing or eliminating the use of chemical inputs like glyphosate, dicamba, and 2,4-D. These herbicides act as abiotic stressors that induce adaptive resistance mechanisms in bacteria, such as the upregulation of multidrug efflux pumps.
Evidence suggests that this type of adaptive resistance is often reversible:
• Return to Baseline: When the environmental signal (the herbicide) is removed, bacterial antibiotic resistance generally returns to its original levels.
• Fitness Costs: Most resistance traits carry a metabolic “fitness cost.” In the absence of herbicide exposure, resistant strains are often outcompeted by sensitive strains that do not have to expend energy maintaining resistance machinery.
2. Restoring Soil Biodiversity
Industrial practices like intensive tillage and monocropping deplete soil microbial diversity, creating “empty” ecological niches where drug-resistant bacteria can flourish without competition.
Regenerative practices—such as no or minimal tillage, cover cropping, and polyculture—restore this biodiversity. A diverse soil microbiome acts as a natural biological barrier:
• Competitive Exclusion: In a healthy ecosystem, upwards of 50,000 microbial species engage in constant competition for resources, which prevents any single resistant strain from becoming dominant.
• Reduced Resistance Accumulation: Research shows that the selection and fixation of antimicrobial resistance are significantly reduced when bacteria are embedded in a stable, natural microbial community.
3. Lower Abundance of Resistance Genes
Studies specifically comparing different farming systems have found that organic and regenerative practices are associated with a lower abundance of antibiotic resistance genes (ARGs) in the soil. By using practices like integrated pest management (IPM), which uses chemical pesticides only as a last resort, farmers can drastically reduce the environmental load of chemicals that drive cross-resistance to human medicine.
4. Integration of Circular Ecosystems
Regenerative systems often incorporate livestock in a circular ecosystem (e.g., silvopasture), which reduces the need for the high-density animal farming practices that require heavy antibiotic use. By avoiding the accumulation of concentrated animal wastes and chemical residues, these systems prevent the “vicious cycle” where agricultural runoff constantly re-introduces ARGs and selective agents into the environment.
In summary, while some genetic mutations induced by pesticides may be permanent, a shift toward regenerative agriculture can stop the emergence of new resistance and allow existing resistant populations to dwindle as natural ecological balances are restored.
17. Do flooding events increase the risk of antibiotic resistance transfer?
Yes, flooding events significantly increase the risk of antibiotic resistance transfer and spread. Flooding acts as a powerful vehicle for the environmental dispersal of resistant bacteria and their associated antibiotic resistance genes (ARGs) across vast distances and into previously uncontaminated areas.
The mechanisms through which flooding facilitates this spread include:
• Waterborne Transmission and Dispersal: Heavy precipitation and subsequent flooding serve as major routes for the waterborne transmission of bacteria and ARGs. These events facilitate the physical movement of resistant microbes through waterways, often carrying them from concentrated sources into broader environmental reservoirs.
• Mingling of Diverse Sources: Floodwaters provide unique opportunities for the mingling of bacteria and ARGs from normally isolated sources. This includes carrying runoff from agricultural fields (which may contain manure with high loads of ARGs and antibiotic residues) and causing sewer systems to overflow, releasing human fecal bacteria and clinical resistance factors into the wider environment.
• Contamination of Sediments and Soils: Research following major storms like Hurricane Harvey has shown that flooding increases levels of ARGs (such as those conferring resistance to beta-lactams and tetracyclines) and loads of human fecal bacteria in coastal sediments and urban soils.
• Persistence in the Environment: The risk is not merely transient; elevated loads of resistant bacteria and ARGs have been found to persist in coastal sediments for several months following a major flood event.
• Proliferation of Pathogenic “Superbugs”: The warmer and wetter conditions associated with flooding create ideal breeding grounds for certain pathogens. For instance, storm surges have been linked to spikes in marine pathogens like Vibrio spp., which were found to carry numerous ARGs and mobile genetic elements following extreme weather events.
• Increased Genetic Exchange: Environmental stressors brought on by extreme weather, such as changes in salinity or temperature, can stimulate horizontal gene transfer (HGT). This allows resistance genes to move more rapidly between different bacterial species, facilitating the emergence of new resistant genotypes.
In summary, flooding events act as a “force multiplier” for the AMR crisis by simultaneously broadening the geographic range of resistant bacteria and providing new opportunities for different microbial communities to exchange resistance traits.
18. Explain the link between temperature resistance and antimicrobial resistance.
The link between temperature resistance and antimicrobial resistance (AMR) is established through shared evolutionary pathways, genetic mechanisms, and coordinated biological stress responses. Rising global temperatures act as a potent environmental stressor that not only accelerates the growth of pathogens but also directly influences the genetic processes that drive resistance.
1. Shared Evolutionary and Genetic Mechanisms
The biological processes by which bacteria develop temperature resistance and AMR are intimately linked. High temperatures serve as a stimulus for genetic change in several ways:
• Increased Mutation Rates: Heat stress is a biological stressor that can increase the rate of de novo mutations within bacterial populations. These mutations can lead to the emergence of resistant phenotypes that are then selected for in the environment.
• Accelerated Horizontal Gene Transfer (HGT): Warmer conditions promote the frequency of conjugation, allowing bacteria to share antibiotic resistance genes (ARGs) more rapidly across species boundaries.
• Thermal Stability and Protein Interaction: Some antimicrobials, such as zinc, work by forming thermally stable complexes with bacterial proteins (e.g., PsaA in S. pneumoniae), which increases oxidative stress and contributes to the antibacterial effect. This suggests that a bacterium’s ability to navigate thermal challenges is fundamentally tied to its ability to survive chemical stressors.
2. Linked Transcriptional Stress Responses
Research into the gene expression profiles of resistant bacteria reveals that mechanisms for surviving heat shock and antibiotics are often activated simultaneously.
• Upregulation of Heat Shock Defense Genes: Resistant mutants (such as those resistant to ampicillin and ciprofloxacin) have been observed to exhibit higher expression of heat shock defense genes, including ibpAB, hslVU, patZ, and ygcP, when navigating environmental challenges.
• General Stress Adaptation: These transcriptional responses provide a holistic defense mechanism. Mutants that evolve under chemical stress (like pesticide or antibiotic exposure) often adopt generalist resistance traits that include increased biofilm formation and heat shock proteins, which grant them a survival advantage against multiple concurrent threats.
3. Temperature as a Driver of Clinical Resistance
Epidemiological data supports a direct correlation between ambient temperature and the prevalence of drug-resistant infections.
• Risk Multiplication: Every 1°C increase in average annual temperature has been associated with significant rises in resistant pathogens, including a 4.7% to 10.7% increase in carbapenem-resistant K. pneumoniae and a 6% increase in carbapenem-resistant P. aeruginosa.
• Resistance Distribution: Studies in the U.S. found that a 10°C increase in local temperature correlated with specific increases in AMR for E. coli (4.2%), K. pneumoniae (2.2%), and Staphylococcus aureus (2.7%).
4. Environmental Transmission Dynamics
Temperature is a critical abiotic determinant for the survival and transmission of bacteria in the environment. Warmer conditions facilitate the proliferation of pathogens in soil and water, providing a larger reservoir for resistant bacteria to persist and eventually infect human hosts. This is particularly evident following extreme weather events, where warmer, wetter conditions following floods provide an ideal breeding ground for resistant marine pathogens like Vibrio spp., which have been found to carry numerous mobile genetic elements and ARGs.
19. How do heat shock genes provide an advantage against antibiotics?
Heat shock defense genes provide an advantage against antibiotics by serving as a holistic transcriptional defense mechanism that helps bacteria survive the physiological stress and oxidative damage caused by antimicrobial drugs. Research has shown that resistant mutants—particularly those selected under combined exposure to pesticides and antibiotics—exhibit significantly higher expression of heat shock defense genes, such as ibpAB, hslVU, patZ, and ygcP, when navigating environmental challenges.
These genes contribute to bacterial survival in several specific ways:
• General Stress Adaptation: Heat shock proteins are part of a broader, generalist stress response that provides a survival advantage against multiple concurrent threats, allowing “superbugs” to flourish in ecological niches where sensitive bacteria would perish.
• Oxidative Stress Defense: Antibiotic exposure often triggers the production of reactive oxygen species (ROS); the upregulation of heat shock genes helps the cell maintain its integrity and navigate this oxidative stress.
• Enhanced Tolerance to Medically Important Drugs: Mutants with increased expression of genes like ibpA and ibpB have demonstrated significantly higher resistance to frontline antibiotics, including ampicillin and ciprofloxacin.
• Linkage to Temperature Resistance: The mechanisms for developing temperature resistance and antimicrobial resistance are often intimately linked, meaning that exposure to thermal stress can select for traits that inadvertently protect bacteria from medically important antibiotics.
In summary, by hyper-expressing these defense genes, bacteria become more resilient to the cellular damage caused by antibiotics, effectively diversifying their evolutionary trajectory toward higher and more robust resistance levels.
20. Can rising temperatures actually increase the rate of bacterial mutations?
Yes, rising temperatures can increase the rate of bacterial mutations, serving as a powerful biological stressor that triggers genetic and physiological changes.
The following mechanisms explain the link between heat stress and increased mutation rates:
• Heat Stress as a Driver of Genetic Change: Rising temperatures act as a biological stimulus that can increase the rate of de novo mutations within bacterial populations. These spontaneous mutations can lead to the emergence of resistant phenotypes that are then fixed through environmental selection.
• Linked Resistance Mechanisms: The processes by which bacteria develop temperature resistance and antimicrobial resistance (AMR) are often intimately linked. Consequently, exposure to thermal stress can select for traits that inadvertently confer resistance to medically important drugs.
• Increased Proliferation and Opportunity: Higher ambient temperatures promote the overall rate of bacterial proliferation. This increased growth not only raises the risk of infection but also creates significantly more opportunities for spontaneous mutations to occur and spread within a population.
• Stimulated Horizontal Gene Transfer (HGT): Beyond individual point mutations, warmer conditions also promote the frequency of horizontal gene transfer, allowing resistance genes to be shared more rapidly across species boundaries.
Epidemiological evidence supports these biological observations, showing a clear correlation between higher local temperatures and increased resistance in clinical pathogens, such as E. coli, K. pneumoniae, and S. aureus. This suggests that the acquisition of AMR and rising global temperatures are intimately connected through these genetic pathways.
21. Could vaccines for livestock replace some antibiotic use?
Yes, evidence from the sources indicates that vaccines for livestock could replace a significant portion of current antibiotic use by preventing infections before they require treatment.
Potential for Reduction
• Significant Decreases in Use: Research suggests that antibiotic consumption in agriculture could be roughly halved if farm animals were properly vaccinated. In some instances, vaccination programs might even allow antibiotic use to be entirely eliminated.
• Case Studies:
◦ Norwegian Salmon: The use of antimicrobials in Norway’s aquaculture fell by 99% between 1987 and 2013, a success attributed in large part to the development and use of effective vaccines.
◦ Danish Pig Farming: Mass immunization experiments in animal herds have led to significant decreases in antibiotic reliance.
• Replacing Growth Promoters: In one study, vaccinated pigs gained more weight and provided a higher yield than non-vaccinated controls, suggesting vaccines could potentially decrease the need for antibiotics used strictly for growth promotion.
Advantages Over Antibiotics
• No Resistance Issues: Bacteria do not develop resistance to vaccines in the same way they do to antibiotics, making them a more sustainable long-term tool for disease management.
• Prophylactic and Therapeutic Benefit: Successful vaccination performs the same role as prophylactic antibiotic use (prevention) while simultaneously reducing the eventual need for therapeutic use (treatment).
Obstacles to Implementation
Despite their potential, several factors currently limit the widespread adoption of vaccines in livestock:
• High Costs: Prevailing prices are often seen as an impediment, particularly for the mass vaccination of smaller animals like poultry.
• Delivery Challenges: While individual injections may be affordable for large animals, they are prohibitively expensive for small animals or fish. There is an urgent need for more vaccines that can be administered via feed or bath mechanisms.
• Lack of Commercial Incentives: While the science for many important vaccines exists, some have not been commercialized because the current market does not provide enough incentive compared to the low cost of antibiotics.
Policy and Economic Solutions
The sources suggest that the market for vaccines would be strengthened if the economic conditions changed—for example, through taxes on antibiotics or direct subsidies for vaccination. International bodies have identified increased vaccine uptake as a key pillar in strategies to decrease the global reliance on antibiotics.
22. How effectively can animal vaccines reduce agricultural antibiotic consumption?
Evidence from the sources indicates that animal vaccines are a highly effective tool for reducing agricultural antibiotic consumption, with the potential to roughly halve global use in farm animals and, in some specific cases, eliminate it entirely,.
The effectiveness of vaccination is demonstrated through several key mechanisms and successful real-world applications:
1. Demonstrable Reductions in Antibiotic Use
• Norwegian Salmon Industry: One of the most striking examples is Norway’s aquaculture sector, where antimicrobial use fell by 99% between 1987 and 2013. This massive reduction occurred while industry output grew 20-fold and is directly attributed to the development and use of effective vaccines alongside better hygiene.
• Danish Pig Farming: Experiments involving the mass immunization of pig herds have led to significant decreases in antibiotic reliance. One specific study reported that mass vaccination more than halved antibiotic use for pigs in a Danish herd.
2. Mechanisms of Reduction
Vaccines reduce antibiotic consumption by addressing the three primary reasons drugs are administered to livestock:
• Replacing Prophylaxis: Successful vaccination essentially performs the same preventive task as prophylactic antibiotic use without contributing to antimicrobial resistance (AMR).
• Reducing Therapeutic Need: By preventing initial infections, vaccines drastically lower the subsequent need for therapeutic antibiotic treatments for sick animals.
• Alternative to Growth Promotion: Some evidence suggests that vaccinated animals (such as pigs) can gain more weight and provide higher yields than non-vaccinated controls, potentially decreasing the perceived need for antibiotics used strictly for growth promotion.
3. Barriers to Implementation
Despite their effectiveness, several obstacles limit the widespread adoption of vaccines:
• High Costs: Prevailing prices are often cited as a major impediment, particularly for the mass vaccination of smaller animals like poultry,.
• Delivery Challenges: While individual injections are feasible for large livestock, they are prohibitively expensive for fish or small animals,. There is a critical need for more vaccines that can be administered via feed or bath mechanisms,.
• Economic Incentives: Many promising vaccines exist in the science phase but have not been commercialized because the low cost of antibiotics currently undermines the market incentive for vaccine development,.
4. Policy and Economic Solutions
To maximize the effectiveness of vaccines, the sources suggest that economic conditions must change to favor immunization over antibiotic use. Proposed strategies include:
• Taxing Antibiotics: Implementing taxes on antibiotic use would make vaccines a more attractive and cost-competitive alternative for farmers,.
• Direct Subsidies: Because society as a whole benefits from reduced antibiotic use, providing subsidies or support loans for farmers to transition to vaccination programs is recommended,.
• Innovation Funding: Using global innovation funds to support the research and development of new animal vaccines, which are generally cheaper to trial than human vaccines, could bring more options to market.
23. What are the biggest cost barriers to mass livestock vaccination?
The biggest cost barriers to mass livestock vaccination involve the high market price of the vaccines themselves, the prohibitive expense of individual administration for small animals, and a lack of financial incentives compared to cheap antibiotic alternatives.
According to the sources, the following factors are the primary economic impediments to widespread use:
• High Prevailing Prices: The current market price of vaccines is frequently cited as a major barrier, particularly for the mass vaccination of smaller livestock like poultry.
• Administration and Delivery Costs: While individual injections may be financially manageable for large animals, the cost to administer them is prohibitively expensive for small animals and fish. There is an urgent need for more vaccines that can be delivered via lower-cost methods like feed or bath mechanisms to make mass immunization viable.
• Comparison to Low-Cost Alternatives: Farmers often perceive vaccine prices as too high relative to the low cost of existing alternatives, namely antibiotics. Currently, the agricultural sector does not fully utilize available vaccines because antibiotics remain a more affordable short-term solution for disease management.
• Lack of Commercial Incentives: Although the science for many important vaccines exists, some have not been commercialized because the current market does not provide enough incentive for pharmaceutical companies to invest in them. This results in a weak supply of products that match farmers’ specific needs.
• Switching and Transitional Costs: Moving away from antibiotic reliance toward vaccination often requires farmers to incur “switching costs,” such as investing in new animal housing to reduce infection spread or more resilient livestock breeds. These costs can be substantial enough that many smaller farms may struggle to remain profitable without subsidies or support loans.
The sources suggest that interventions such as taxes on antibiotics or direct subsidies for vaccines could help shift these economics, making vaccination a more attractive and competitive option for producers.
24. Could taxes on antibiotics incentivize a shift toward animal vaccines?
Yes, according to the sources, taxes on antibiotics could act as a powerful economic lever to incentivize a shift toward animal vaccines by making antimicrobials more expensive and alternatives more commercially attractive to farmers.
Economic Logic of Taxation
From an economic perspective, the use of antibiotics in agriculture creates negative externalities, meaning the individual user (the farmer) does not bear the full cost that their action—increasing drug resistance—imposes on society.
• Internalizing Costs: A tax aims to “internalize” these societal costs by adding them to the purchase price of the drug.
• Aligning Incentives: By making antibiotics more expensive, the tax aligns the farmer’s financial incentives with the interests of public health, discouraging growth promotion and unnecessary prophylactic use.
Strengthening the Vaccine Market
The sources highlight that current standard animal vaccines are often underutilized because they are perceived as too expensive compared to cheap, readily available antibiotics. Implementing a tax would change this dynamic:
• Increased Attractiveness: As the price of antibiotics rises, vaccines and other alternatives (such as improved animal husbandry and diagnostics) become more attractive to farmers.
• Market Stimulation: Increased demand from farmers would create a stronger market, which in turn would encourage pharmaceutical companies to commercialize promising vaccines currently in late-stage development and invest in new ones.
• Funding Research: Taxes on antibiotics could potentially be used to fund the research and development of new animal vaccines, which is generally less expensive than developing human vaccines due to lower trial costs.
Implementation and Potential Challenges
While the sources support taxation as a viable strategy, they also note several practical considerations:
• Tax Level: Because antibiotics are currently very cheap, the tax might need to be high relative to the base cost of the product to effectively change behavior.
• Point of Levy: To minimize the risk of farmers circumventing the tax through black markets or counterfeit drugs, it is recommended that the tax be levied as early as possible in the supply chain, such as when the product leaves the factory.
• Targeted Taxation: Policy-makers could choose to tax antibiotics most important for human medicine more heavily to specifically discourage their use in livestock.
• Support for Small Farmers: Some governments may need to pair taxes with subsidies or support loans to help smaller farmers manage the upfront “switching costs” involved in transitioning to new, less antimicrobial-reliant farming practices.
In summary, by shifting the economic balance away from cheap chemical reliance, taxation can drive a “One Health” transition toward vaccination and improved hygiene as more sustainable methods for managing livestock disease.
References
Ahmad, E., Qasim, K., & Bibi, T. (2025). Antimicrobial Resistance In Extended Spectrum Beta Lactamase Producing Escherichia Coli Triggered By Pesticides. Pakistan Journal of Medical & Cardiological Review, 4(4). https://pakjmcr.com/index.php/1/about
Ahmad, M. F., Ahmad, F. A., Alsayegh, A. A., Zeyaullah, M., AlShahrani, A. M., Muzammil, K., Saati, A. A., Wahab, S., Elbendary, E. Y., Kambal, N., Abdelrahman, M. H., & Hussain, S. (2024). Pesticides impacts on human health and the environment with their mechanisms of action and possible countermeasures. In Heliyon (Vol. 10, Number 7). Elsevier Ltd. https://doi.org/10.1016/j.heliyon.2024.e29128
Baig, M. I. R., Kadu, P., Bawane, P., Nakhate, K. T., Yele, S., Ojha, S., & Goyal, S. N. (2023). Mechanisms of emerging resistance associated with non-antibiotic antimicrobial agents: a state-of-the-art review. In Journal of Antibiotics (Vol. 76, Number 11, pp. 629–641). Springer Nature. https://doi.org/10.1038/s41429-023-00649-4
Curutiu, C., Lazar, V., & Chifiriuc, M. C. (2017). Pesticides and antimicrobial resistance: from environmental compartments to animal and human infections. In New Pesticides and Soil Sensors (pp. 373–392). Elsevier. https://doi.org/10.1016/B978-0-12-804299-1.00011-4
Daram, N., Moreno Morales, J., Baryamujura, J., Akinwotu, O., Wadanamby, R., & Van Dongen, M. B. (2021). Herbicides and their impact on antimicrobial resistance in the context of food and food safety. The Journal of Food Diagnostics. www.amr-insights.eu/about-us/ambassadors
Graham, M. E., Wilson, B. A., Ramkumar, D., Rosencranz, H., & Ramkumar, J. (2025). Unseen Drivers of Antimicrobial Resistance: The Role of Industrial Agriculture and Climate Change in This Global Health Crisis. Challenges, 16(2), 22. https://doi.org/10.3390/challe16020022
Larsson, D. G. J., & Flach, C. F. (2022). Antibiotic resistance in the environment. In Nature Reviews Microbiology (Vol. 20, Number 5, pp. 257–269). Nature Research. https://doi.org/10.1038/s41579-021-00649-x
Miller, S. A., Ferreira, J. P., & Lejeune, J. T. (2022). Antimicrobial Use and Resistance in Plant Agriculture: A One Health Perspective. Agriculture (Switzerland), 12(2). https://doi.org/10.3390/agriculture12020289
Murray, L. M., Hayes, A., Snape, J., Kasprzyk-Hordern, B., Gaze, W. H., & Murray, A. K. (2024). Co-selection for antibiotic resistance by environmental contaminants. Npj Antimicrobials and Resistance, 2(1). https://doi.org/10.1038/s44259-024-00026-7
Pasha S A, R., B N, N., Ismail, W., Gowri, R., & Natarajan, A. (2024). Experiment to Demonstrate Pesticide-Induced Antimicrobial Resistance (AMR): An Emerging Health Threat. Cureus. https://doi.org/10.7759/cureus.54243
Xing, Y., Wu, S., & Men, Y. (2020). Exposure to Environmental Levels of Pesticides Stimulates and Diversifies Evolution in Escherichia coli toward Higher Antibiotic Resistance. Environmental Science and Technology, 54(14), 8770–8778. https://doi.org/10.1021/acs.est.0c01155
Yasemi, M., Jalali, A., Asadzadeh, M., & Komijani, M. (2025). Organophosphate pesticides and their potential in the change of microbial population and frequency of antibiotic resistance genes in aquatic environments. Chemosphere, 376. https://doi.org/10.1016/j.chemosphere.2025.144296






